The presence of both sympathetic activation-mediated triggers and parasympathetic activation-mediated substrates are required to initiate and maintain some forms of atrial fibrillation (AF). AF predominantly precipitated by parasympathetic stimulation is known as vagally-mediated AF (VM-AF). The role of novel drugs and molecular targeted gene therapy that modulate the autonomic nervous system are therapeutic options in this unique population with VM-AF. Here, we review the role of the sympatho-vagal balance in the genesis of AF and consider drug therapy for VM-AF.
In accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Statement, literature search was conducted using the keywords “vagal”, “vagal nerve”, “vagus”, “vagus nerve”, and “atrial fibrillation”. Retrieved citations were first screened independently by 2 reviewers for inclusion and exclusion criteria.
A total of 14 studies and 3 practice guidelines from 1986-2017 were included. Only two clinical investigations evaluated the effectiveness of disopyramide and sotalol in human subjects with VM-AF. The potential role of antiarrhythmic drugs has been studied in animal models.
Growing evidence suggests that the autonomic nervous system is integral in the development of VM-AF. Novel medications and genetic targets are undergoing investigation with promising results.
Atrial fibrillation (AF) in patients without underlying structural heart disease can be precipitated by parasympathetic stimulation. Vagally-mediated AF (VM-AF) was initially described in 1978 by Coumel et al 1. The mechanism was thought to be parasympathetic stimulation-induced inhomogeneous shortening of the atrial effective refractory period (AERP) and slowing of the sinus rate 1. This may be accompanied by sympathetic stimulation and subsequent triggered activity. Several reports suggest that high parasympathetic tone was involved in causing lone paroxysmal AF 2-7.
Patients with VM-AF are usually young males with paroxysmal episodes that typically occur at night or following food intake. The prevalence of VM-AF is unclear. In selected patients with paroxysmal AF undergoing pulmonary vein isolation have a prevalence of VM-AF up to 27% 8. In contrast, “adrenergic AF” is AF initiated predominantly by sympathetic stimulation associated with exercise and emotional stress 9.
The innervation of the heart includes local parasympathetic and sympathetic ganglionated plexi Figure 1. They are closely collocated at the tissue and cellular level for intrinsic and extrinsic cardiac innervation 10. In an experimental and animal model, the presence of sympathetic activation-mediated triggers and parasympathetic activation-mediated substrates are required to initiate and maintain AF 11,12. In a human study, vagal and adrenergic components were involved in initiation of paroxysmal AF and this was followed by vagal predominance 13.
Surgical or percutaneous interventions, novel drugs, and molecular-targeted gene therapy that modulate the autonomic nervous system could be potential therapeutic options, but the number of published studies is limited, and the majority are observational and small-scale. The present review considers current evidence about the role of the vagus nerve activation in the genesis of AF and eligible drug therapies for VM-AF.
Definition of Vagally-Mediated AF
In animal models, VM-AF was defined as AF caused by stimulation of the vagus nerve with observed atrioventricular block, asystolic periods, sinus bradycardia and an increase in heart rate variability 14,15. In humans, VM-AF was defined as paroxysmal AF that occurs with predominant vagal activation, such as, during sleep, after eating a big meal, or in relationship to other recognized vagal triggers, usually preceded by bradycardia 8,15,16. Patients may have no underlying heart disease or other systemic disorders that could explain the AF 17.
The current study was conducted in accordance with the recent Preferred Reporting Items for Systematic Reviews and Meta-Analyses Statement (18, 19). Relevant articles were obtained from a search of EMBASE and MEDLINE databases from inception through 2020. We performed an initial search on February 8, 2020 including the terms “vagal”, “vagal nerve”, “vagus”, “vagus nerve”, and “atrial fibrillation”. We performed a second search on April 26, 2020 using the term “parasympathetic”, “parasympathetic stimulation”, and "atrial fibrillation” (full search strategy and search terms in Figure 2) then the first and second search databases were combined. Only full articles in English were included. The search strategy was done according to published recommendations. To find additional eligible studies, review articles were also screened. A manual search for additional pertinent studies using references from retrieved articles was also completed.
Figure 2. Search methodology and selection process.
Study eligibility of drug therapy for VM-AF was independently determined by two investigators (PR and JK) and differences were resolved by mutual consensus. The eligibility criteria included the following:
(1) Experimental studies, cohort studies (prospective or retrospective), case-control studies, cross-sectional studies, and randomized control trials reporting the use of antiarrhythmic drugs (AADs) on VM-AF were included. Studies were excluded if intervention, such as ablation, was performed on subjects.
(2) Studies describing the definition of VM-AF or induction protocol of the vagus nerve stimulation, medication name, dose of medication, and outcome were also included in the review.
A standardized data collection form was used to obtain the following information from each study: medication name; name of first author; year of publication; subject model; induction of VM-AF; diagnostic criteria of VM-AF; medication dose; proposed drug mechanisms; and outcome.
Two investigators (PR and JK) independently performed data extraction to ensure accuracy. Any data discrepancy was resolved by reviewing the primary data from the original articles.
Details of the selection flowchart are summarized in Figure 2. Our initial search on February 8, 2020 yielded 2,133 potentially relevant articles (1,498 articles from EMBASE and 635 articles from MEDLINE). The second search on April 26, 2020 further identified 343 potentially relevant articles (224 articles from EMBASE and 119 articles from MEDLINE). After the exclusion of 996 duplicated articles, 1,480 articles underwent title and abstract review. Of the total, 1,273 articles were excluded at this stage as it was conducted in non-VM-AF. This left 207 abstracts for full-length article review; 126 studies were further excluded as these studies did not evaluate pharmacological management or they did not report any AAD use. An additional 36 articles were excluded as they were review articles that referred to the original articles; 28 abstracts were excluded as full articles were not available.
A total of 14 studies 17,20-32 and 3 practice guidelines 16,33,34 from 1986-2017 were included Table 1. Only two clinical investigations evaluated the effectiveness of AADs in human subjects with VM-AF 17,30. Clinical characteristics of patients with vagal-mediated and adrenergic-mediated AF in the included studies are summarized in Table 2.
Table 1. Summary characteristics of included studies or reports of drug therapy for vagally-mediated atrial fibrillation
|
Antiarrhythmic Class/Therapy
|
Medication
|
First author, year
|
Experimental Model/Guideline
|
Vagal AF induction
Patient’s characteristic /remarks
|
Medication dose
|
Proposed mechanism
|
Outcome
|
|
Class IA
|
Cibenzoline
|
Miyakoshi, 2009
|
Human
|
AF at nighttime, at rest, after taking meals, and terminated spontaneously within 24 h, without underlying heart or endocrine disorders
|
200-300mg/day
|
- M2 muscarinic receptor blocker
- Class IA antiarrhythmic effect
|
AF was completely eliminated in 24/32 patients.
|
|
Disopyramide
|
Fuster, 2006
|
Guideline
|
Expert comments in ACC/AHA/ESC 2006 guidelines
|
N/A
|
- M2 muscarinic receptor blocker
- Direct IKAch blocker
- Class IA antiarrhythmic effect
|
N/A
|
|
January, 2014
|
Guideline
|
Expert comments in 2014 AHA/ACC/HRS guideline
|
N/A
|
N/A
|
|
Kirchhof, 2016
|
Guideline
|
Expert comments in 2016 ESC Guidelines
|
N/A
|
N/A
|
|
Class IB
|
Lidocaine
|
David, 1990
|
Animal, canine
|
Vagal nerve stimulation
|
2 -3 mg/kg
|
- Mechanism is unclear
|
Terminated AF in 10/10 episodes
|
|
Class IC
|
Flecainide
|
Wang, 1992
|
Animal, canine
|
Vagal nerve stimulation
|
1 mg/kg
|
- Slowed atrial conduction
- Tachycardia-dependent increase AERP
- Increased refractory period
|
Terminated AF in 16/16 dogs. AERP was increased and conduction velocity was reduced.
|
|
Fuster, 2006
|
Guideline
|
Expert comments in ACC/AHA/ESC 2006 guidelines
|
N/A
|
N/A
|
|
Pilsicainide
|
Hayashi, 1998 (21)(Hayashi et al., 1998)21(21)
|
Animal, canine
|
Vagal nerve stimulation
|
1.0 mg/kg
|
- Prolong intraarial conduction time
- Reduce Vmax
- M2 muscarinic receptor blocker
|
Terminated AF in 6/6 dogs. Increased in AERP and intraatrial conduction time. Wavelength index slightly reduced.
|
|
Class II
|
Sotalol
|
Yesil, 1999
|
Human
|
Paroxysmal AF with vagal stimulus without any heart or other significant lung, liver, kidney, and thyroid disease
|
80–120 mg/day for 2-4 days
|
- Prolong action potential and AERP (Class II with class III antiarrhythmic effect)
|
5/14 patients converted to sinus rhythm. In non-converted patients, ventricular rate was significantly reduced
|
|
Class III
|
Bretylium tosylate
|
Goldberger, 1986
|
Animal, canine
|
Vagal nerve stimulation
|
2.5–5 mg/kg
|
- Mechanism is unclear
|
Terminated AF in 5/5 dogs, total of 31/31 episodes.
|
|
MS-551
|
Hayashi, 1998
|
Animal, canine
|
Vagal nerve stimulation
|
0.5–1.0 mg/kg
|
- Inhibits IK and IK1
- Prolong action potential duration and AERP.
- M2 muscarinic receptor blocker
|
Terminated AF in 6/8 dogs. Increased in AERP, wavelength index significantly increased.
|
|
Nibentan
|
Fedorov, 2000
|
Animal, canine
|
Vagal nerve stimulation
|
0.063–0.250 mg/kg
|
- Inhibits IK
- M2 muscarinic receptor blocker
- Inhibit IKACh
- Prolong AERP
|
Terminated AF in 6/8 dogs. Prevent AF induction in 9/10 dogs.
Increased AERP with and without vagal stimulation, and, reduced the number of simultaneously occurring reentrant wavelets
|
|
Tedisamil
|
Fischbach, 2001
|
Animal, canine
|
Vagal nerve stimulation
|
0.1-1 mg/kg
|
- Inhibit IKACh
- Prolong AERP
|
Terminated AF in 11/11 episodes and could not be reinduced within 30 minutes. AF cycle length was prolonged.
|
|
Selective IKAch inhibitor
|
Tertiapin
|
Hashimoto, 2006
|
Animal, canine
|
Animal, canine
|
4, 12, and 41 nmol/kg
|
- Selective IKAch inhibitor
|
Terminate 5/5 dogs at 12 nmol/kg
Prolong AERP
|
|
NIP-151
|
Hashimoto, 2008
|
Animal, canine
|
Animal, canine
|
5-15 µg/kg/min
|
N/A
|
|
NTC-801
|
Machida, 2011
|
Animal, pig
|
Vagal nerve stimulation
|
0.3, 1, and 3 µg/kg/min
|
Terminated AF in 8/8 dogs
|
|
DNA vectors expressing
(Gαictp)
|
Gαictp/ Gαoctp
|
Aistrup, 2011
|
Animal, canine
|
Vagal nerve stimulation
|
Gαictp minigene 1 mg
|
- Competitively bind M2 muscarinic receptor
- Preventing degradation of the Gαi to Gαi/o
- Prolonged and reduced dispersion of AERP
|
Vagal-induce AERP shortening, AF inducibility, mean AF duration was significantly decreased
|
|
Gαictp
|
Lou, 2018
|
Animal, canine
|
Vagal nerve stimulation
|
500 μl of recombinant adenovirus
|
|
Epicardial Botulinum Toxin Injection
|
Botulinum toxin
|
Oh, 2011
|
Animal, canine
|
Vagal nerve stimulation
|
50 units
|
- Reduced dispersion of AERP
|
Tempolary suppression of VM-AF inducibility for 1 week
|
AERP: Atrial effective refractory period, AF: atrial fibrillation, AV: Atrioventricular, CTP: C-terminal peptide, IKAch: Acetylcholine-activated K+ current, IK: Delayed rectifier potassium current, IK1: Inward rectifier potassium current
Drugs being investigated in humans Table 1
According to relevant literature, antiarrhythmic effectiveness of disopyramide and cibenzoline were evaluated in humans 17. In the first study, Miyakoshi et al 17 investigated the role of cibenzoline and disopyramide in 20 and 14 VM-AF patients, respectively. Medications were found successful to eliminate VM-AF episodes in 75% of patients. There was also a significant decrease in vagal tone index (high frequency component of heart rate variability). No significant difference of vagal tone index was seen between cibenzoline and disopyramide, but anticholinergic side-effects were observed more frequently in patients receiving disopyramide compared to cibenzoline (14% versus 0%, respectively) 17. Because cibenzoline is only available in Japan and Europe and the U.S. Food and Drugs Administration has not approved it, only disopyramide, a class IA AAD, is suggested in treatment of VM-AF given its prominent vagolytic pharmacological effect according to the AHA/ACC/HRS 2014 guidelines 33 and ESC 2016 guidelines 34 for the management of patients with AF.
Drugs being investigated in experimental animal models Table 1
Class I antiarrhythmic drugs
Potential effects of class IB AADs in VM-AF was studied in a canine model by David et al 29. The study reported 100% effectiveness in pharmacologically converting VM-AF to sinus rhythm with lidocaine 29. Wang et al 31 investigated the role of flecainide, a class IC AAD, in 16 canines. Flecainide terminated VM-AF episodes in all 16 dogs by a use (tachycardia)-dependent increase in atrial effective refractory period (AERP) (70±9%, 79±4%, and 92±7% at cycle length of 250, 200, and 150 ms, respectively; p<0.001). Flecainide also increased the reentry wavelength (31.0±10.1%, 28.5±6.4%, and 28.3±8.9% at cycle length of 250, 200, and 150 ms, respectively; p<0.05) and decreased the number of functional reentry circuits resulting in reduced propensity for VM-AF (p<0.001; raw data not reported) 31.
According to these data, Flecainide, via its Class IC AAD effect, was recommended as a second treatment option for VM-AF in ACC/AHA/ESC 2006 Guidelines for the Management of Patients with Atrial Fibrillation 16.
In 1998, Hayashi et al 21 studied the effectiveness of pilsicainide for AF termination of VM-AF in 6 dogs. Pilsicainide was 100% effective; it increased AERP (100±30 ms to 143± 28 ms; p<0.05) and wavelength index (48%; p<0.01) 21.
Propafenone, a class IC AAD, was not recommended in VM-AF because of its co-existing beta-blocking effect that may facilitate VM-AF during bradycardia 16. The Na+ channel blocking effect of Class IC AADs is not specific for VM-AF in comparison to disopyramide, which has a Na+ channel blocking and a vagolytic effect.
Class III antiarrhythmic drugs
The main antiarrhythmic mechanism of Class III AADs is to prolong action potential duration by blocking the rapid component of the delayed rectifier current (IKr) which is not specific for VM-AF 20. The first class III AAD evaluated in an animal model was bretylium tosylate. In 1986, Goldberger et al 23 successfully developed a VM-AF canine model; 100% of AF was terminated by bretylium tosylate. Bretylium has an autonomic effect as it prevents sympathetic neurotransmitter release from nerve terminals 35,36.
In 1998, Hayashi et al 21 reported MS-551, a class III AAD structurally similar to pyrimidinedione, as an effective AAD for VM-AF in 6/8 canine models. MS-551 inhibited IK and decreased the transient outward current (Ito) and the IK1 current 37. However, MS-551 has not been developed as a clinical AAD.
Fedorov et al 20 reported that nibentan is highly effective in reducing the number of reentrant wavelets in VM-AF in a canine model. This study showed significant dose-dependent increases in AERP (55±9%, 82±12%, and 90±6% at the dose of 0.063, 0.125, and 0.250 mg/kg; p < 0.01) and wavelength (47±7%, 68±12%, and 72±4%; at the dose of 0.063, 0.125, and 0.250 mg/kg; p< 0.01). Nibentan has not been developed as an AAD for clinical use.
Tedisamil was investigated to treat VM-AF 26. Tedisamil was effective in terminating 11/11 episodes of VM-AF in a canine model. It also prevented re-induction of AF within 30 minutes of drug administration. The fibrillation cycle length increased before conversion (112±25 ms to 232±15 ms; p<0.01) suggesting lengthening of the AERP 26. Tedisamil has not been developed as an AAD for clinical use.
Sotalol has class III antiarrhythmic and beta-blocker effects. Yesil et al 30 compared sotalol in patients with presumed VM-AF to those with purported adrenergic AF. Only 36% of the patients with VM-AF returned to sinus rhythm; however, 71% of adrenergic AF patients were successfully terminated with sotalol. The study concluded that sotalol is more effective in treating adrenergic AF due to its combined class III and beta blocking effect 30,33,38.
IKAch inhibition is a specific therapeutic target and strategy for VM-AF 22,27,28. Tertiapin was the first IKAch selective inhibitor tested in a canine model. Hashimoto et al 22 reported that tertiapin prolongs the AERP (134±9 to 162±7 ms; p<0.05) without affecting ventricular repolarization (171±8 to 172±3 ms) during vagal nerve stimulation and terminates AF with 100% efficacy. Moreover, tertiapin did not affect PR, QRS and corrected QT intervals (raw data was not presented by the authors) 22.
Drugs being investigated in vitro
NIP-151 is an IKAch selective inhibitor in an in vitro model (single whole cell voltage clamp experiment) with minimal effect on IKr 28. In a canine model, NIP-151 dose-dependently prolonged AERP (28±5, 32±7, and 33±7 ms at the dose of 15,30, and 75 μg/kg/min, respectively; p<0.05) but did not affect the ventricular effective refractory period (raw data were not shown). Given its minimal effect on IKr, there were no significant electrocardiographic changes as opposed to dofetilide (IKr blocker), which causes QT prolongation (23±4 and 32±2 ms at the dose of 0.3 and 3μg/kg/min, respectively) 28.
NTC-801, another IKAch selective inhibitor, was studied in guinea pig atrial cells and a VM-AF rapid pacing canine model 27. NTC-801 selectively and dose dependently prolonged AERP (21.3±3.3, and 30.0±2.6 ms at the dose of 1 and 3 μg/kg/min; p=0.0204 and p=0.0006, respectively) but was frequency-independent (38.7±7.9, 38.0±7.4, and 35.7±7.1 ms at basic cycle length of 300, 250, and 200 ms; p=0.0038, p=0.0038, and p=0.0037, respectively) without affecting ventricular effective refractory period (raw data was not presented by the authors) 27.
VM-AF is seen in a patient population with unique clinical characteristics and is not likely due to isolated vagal activation. Disopyramide can be useful in patients with VM-AF because of its vagolytic and Class IA antiarrhythmic drug effects. Flecainide could be considered although there are limited data. Selective IKach channel blockers and novel genetic targets for the treatment of VM-AF are undergoing investigation with promising results.
Clinical Characteristics of Vagally-Mediated and Adrenergic Atrial Fibrillation
According to the 2014 AHA/ACC/HRS guidelines, clinical characteristics of AF are classified into categories by duration of episodes including paroxysmal, persistent, long-standing persistent, and permanent AF. These definitions for AF pattern have been useful for the management of AF but are limited by the lack of correlation to their underlying pathogenic mechanisms 33.
The original observational study of 18 VM-AF patients by Coumel et al 1 showed a predominantly male population with a ratio of men to women of 4:1. These patients were younger; the ages of first symptom onset were 25-60 years. The frequency of AF episodes was variable from patient to patient, ranging from sporadic events to recurrent daily events, and lasting from a few minutes to several hours. The AF episodes usually occurred during the night, after the digestive period of a large meal when vagal tone surged, and often ended in the morning. Interestingly, many patients in this study reported that AF episodes were preventable by exercising, but the resting or relaxation period following emotional distress was often followed by onset of AF episodes and symptoms 1Table 2.
Table 2. Summary of clinical characteristics of patients with vagal-mediated and adrenergic-mediated atrial fibrillation in the included studies
| Vagal-mediated AF |
Adrenergic-mediated AF |
| More common in middle-age men and athletes |
More common in older population |
| Patients with structurally normal heart or without any identifiable heart disease |
Patients with structurally abnormal heart or with identified heart disease |
| Occurs with vagal stimulus such as sleep, alcohol consumption, postprandial or post-exercise |
Provoked by physical or emotional stress |
| Presence mainly during the night |
Presence mainly during daytime |
| Preceded by bradycardia |
Preceded by tachycardia |
| Lower ventricular response rate |
Higher ventricular response rate |
| Worsen by beta-blocker |
Improved/suppressed with beta-blocker |
| Less likely to progress to permanent AF |
More likely to progress to permanent AF |
Makrides et al 40 reported a case with transient ST elevation in the inferolateral leads in VM-AF that resolved after cardioversion. The mechanism was hypothesized to be from high parasympathetic tone. Increased vagal stimulation may augment early repolarization in the inferolateral leads causing transient ST elevation 39,40. Other plausible explanation such as coronary vasospasm cannot be excluded. VM-AF was also reported in ischemic stroke due to the enhanced central vagal activity in basal ganglion infarction 41. Cardioinhibitory neurocardiogenic syncope, which was aggravated by nausea, was also found to co-present with VM-AF 42.
In contrast to VM-AF, adrenergic AF is caused by a high adrenergic state (high sympathetic tone), such as, exercise, is associated with emotion, daytime hours, and higher heart rate. It can also occur in older patients with structurally abnormal hearts, and improves with beta-blocker use. The differences in clinical characteristics between VM-AF and adrenergic AF are summarized in Table 21,8,14,33,43-46.
The identification of triggers for paroxysmal AF is based on clinical autonomic characteristics, is qualitative with expected overlaps, and is not likely due to isolated vagal activation. Nevertheless, the characteristics clinically distinguish a select group of young patients with a clear association between episodes of AF and vagal activation. This distinction warrants consideration of novel therapeutic options for these young patients who are otherwise healthy.
Prevalence of Vagally-Mediated AF
In two small studies of AF patients who were referred for pulmonary vein isolation, VM-AF and adrenergic AF prevalence was approximately 12-27% and 7-16%, respectively 8,47. In the larger Euro Heart Survey of 1,517 patients with paroxysmal AF, the prevalence of VM-AF was 6%, and the prevalence of adrenergic AF was 15%. 38. This VM-AF prevalence estimated from selected populations are likely an over-estimation from the general population because patients with AF from the general population are more likely to be elderly with comorbidities and a persistent pattern of AF and are less likely to be referred for ablation. The true prevalence of VM-AF is still unknown.
The first large descriptive observational study of VM-AF was reported by the Euro Heart Survey Registry. In contrast to Coumel’s definition, VM-AF was defined as AF that occurred after meals and/or was present during the night only without evidence of any adrenergic triggers regardless of structural heart disease. In the Euro Heart Survey Registry, the prevalence of VM-AF in paroxysmal AF, age, body mass index, and other characteristics were similar between men and women. Of the total, 19% of VM-AF patients experienced progression from paroxysmal AF to persistent AF 38.
VM-AF is also believed to play a role in endurance athletes presenting with AF. The incidence of AF in male athletes was 1.8-8.8 fold higher than non-active men. Endurance athletes are known to have elevated vagal tone as well as dilated atria causing stretch to the atrial wall. Stretching of the atrial wall activates the stretch receptors, shortening the action potential and AERP, thus precipitating AF (48). However, the prevalence of VM-AF has not been well established in the athletic population.
Sympatho-Vagal Balance in the Genesis of AF and Pathophysiology of Vagally-Mediated AF
The intrinsic ganglia and nerves (ganglionated plexi) are mostly found in the atria especially at the pulmonary vein to left atrial junction, superior vena cava, right atrial junction, and atrioventricular node Figure 1A. These ganglionated plexi are innervated by nerve fibers derived from the parasympathetic nervous system and sympathetic nervous system Figure 1A. The complex sympathetic and vagal interactions at the cardiac ganglion, cellular and subcellular levels are shown schematically Figure 1B and Figure 1C.
Figure 1. A) Extrinsic and intrinsic cardiac autonomic innervation; 2B) Autonomic innervation at the cardiac myocyte; C) The cellular/subcellular channel and signal transduction pathways of substrate components of vagally-mediated AF (VM-AF) and mechanism of drug therapy for VM-AF. (IK1: Inward rectifier potassium channel; Iks : Slow delayed rectifier channel; LTCC: L-type calcium channel; FCC: Funny current channel; IKAch: Acetylcholine-activated potassium channel; RyR2: Ryanodine receptor 2 receptor; M2R : Type-2 muscarinic cholinergic receptor; SR: Sarcroplasmic reticulum).
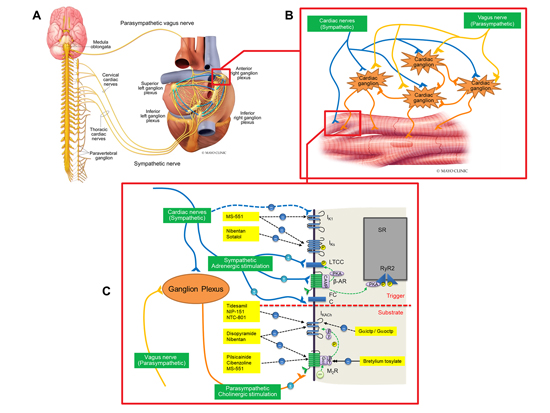
Sympathetic activation leads to focal ectopic firing through enhanced automaticity, early after-depolarization (EAD), or delayed after-depolarization (DAD)10. Enhanced automaticity is mediated by β-adrenergic activation of the funny current (If) 49 and an increased slope of phase-4 depolarization 50. Phase-3 EAD is associated with lengthening of the action potential duration, which is augmented by β-adrenergic activation through the enhanced L-type calcium channel current (ICaL) 51. Beta-adrenergic activation also increases the opening of intracellular calcium and ryanodine receptor 2 (RyR2) through protein kinase A/calcium calmodulin-dependent protein kinase II (PKA/CaMKII) activation, followed by diastolic RyR2 calcium leakage mediating DAD 10. Parasympathetic stimulation activates the acetylcholine-dependent potassium current (IKAch) causing shortening of action potential duration thereby increasing the propensity for inhomogeneity of refractoriness and reentry mediated AF substrate 52.
In summary, vagal nerve stimulation enhances the spatially heterogeneous action potential duration and refractory period, which creates a reentry substrate for AF. Together with adrenergic activity, increased automaticity, EAD and DAD provide a plausible triggering mechanism for AF. The dynamics of sympatho-vagal interaction are possible underlying mechanisms of initiation and maintenance of VM-AF 53.
Mechanism of Drug Therapy in Vagally-Mediated AF
Key pharmacological targets for VM-AF are shown in Figure 1C. Disopyramide may be useful for VM-AF although clinical data are limited. It is the only drug clinically available in the U.S. with a combined Class IA antiarrhythmic and anticholinergic effect. Cibenzoline is another Class IA AAD with anticholinergic effect but not available in the U.S. The mechanisms of anticholinergic activities between cibenzoline and disopyramide are different. Both cibenzoline and disopyramide have an antimuscarinic effect on atrial M2 muscarinic receptors; however, cibenzoline also has a direct blocking effect on IKAch channel 54. Theoretically, cibenzoline could diminish vagal tone more than disopyramide with less side effects given weaker anticholinergic actions on type-1 muscarinic cholinergic receptors in the brain and type-3 muscarinic cholinergic receptors in skeletal muscle and glands than type-2 muscarinic cholinergic receptors in the heart 17.
The use of Class IC and Class III AADs for VM-AF is based on the same principles of antiarrhythmic effects in patients with AF as in the general population. Flecainide, a Class IC AAD, can be considered for use in patients with VM-AF. Sotalol, a Class III AAD with beta-blocking effect, could be considered in selected patients with VM-AF without baseline bradycardia. Observations from three IKAch selective inhibitor studies suggest that IKAch selective inhibitors could be future therapeutic options for VM-AF without the associated risk of ventricular arrhythmia 22,27,28.
Other novel therapeutic approaches
Activation of the Gαi/oβγ protein signal transduction pathway plays a key role in the vagally-mediated shortening of AERP. Acetylcholine, released by vagal stimulation, binds to M2 receptors, which in turn initiate Gαi/oβγ protein uncoupling into Gαi/o and Gβγ subunits Figure 1C. Gβγ subunits activate IKACh , causing shortening of the action potential duration and AERP 55. A non-viral gene-based strategy was recently developed to inhibit vagal-mediated signaling selectively in the left atrium to prevent VM-AF24,25. These studies presented a novel genetic target and treatment of VM-AF at the molecular level. However, further investigation in clinical studies is needed to assess long-term efficacy and potential adverse effects.
Interventional approach for neuromodulation
Low- level vagal nerve stimulation could be used for therapeutic benefit without pro-arrhythmic effect. Although this appears to be paradoxical especially in the context of VM-AF, low level vagal nerve stimulation below the bradycardia threshold (not less than 40% reduction of baseline heart rate in a canine model) appears to be attributed to its anti-adrenergic effects56-58. In a canine model, there was no increase in AF inducibility until vagal nerve stimulation significantly slowed the heart rate 56. In the randomized clinical trial “Transcutaneous Electrical Vagus Nerve Stimulation to Suppress Atrial Fibrillation (TREAT AF)”, investigators demonstrated that sympatho-vagal modification by low-level transcutaneous electrical stimulation of the tragus nerve, an auricular branch of the vagus nerve, in patients with paroxysmal AF, significantly reduced AF burden at 6 months without complications 59 similar to previous low-level vagus nerve stimulation studies 60-62. Ganglionated plexus ablation has been evaluated in animal models and human subjects. Ganglionated plexus ablation eliminated vagal response and subsequently abolished AF 63-65.