Acute Pulmonary Vein Reconnection after Ablation using Contact-force Sensing Catheters: Incidence, Timing, and Ablation Lesion Characteristics.
Muhammad Balouch1, Dong Juang1, Bhradeev Sivasambu1, Rizma J. Bajwa1, Tarek Zghaib1, Jonathan Chrispin1, Ronald D. Berger1, Hiroshi Ashikaga1, Hugh Calkins1, Joseph E. Marine1, David D. Spragg1
1Johns Hopkins Hospital Heart and Vascular Institute, Baltimore MD.
Acute pulmonary vein (PV) reconnection predicts atrial fibrillation (AF) recurrence after ablation. Contact-force (CF) sensing catheters improve lesion delivery. We assessed the incidence, timing, location, and lesion characteristics of acute reconnection after PV isolation with CF sensing catheters.
Patients undergoing radiofrequency ablation for AF from October 2016 to February 2017 were studied. Assessment for acute reconnection at 20 and 40 minute intervals was performed in each isolated PV. Additional lesions were applied as needed. Lesion location, contact force, power, duration, impedance, and force-time integral values were compared at sites with and without reconnection.
Twenty-two patients (60.6 + 1.8 years; 36.4% female; 27.3% persistent AF; CHA2DS2VASC 1.9 + 0.3) were included. Eighty-eight veins were isolated. Eleven reconnections occurred in 10 patients; 9 occurred by 20 minutes and 2 between 20 - 40 minutes. Most reconnections (6/11) were in the left superior PV. Of 4993 ablation points analyzed, 72 were at acute reconnection sites, and no differences in average contact force (11.4 + 8.1 vs 11.3 + 7.1 gm, p=0.868), power (29.7 + 3.9 vs 29.9 + 4.6 watts, p=0.620), impedance (64.1 + 60 vs 72.5 + 60, p=0.236) and the force time integral (86.9 + 78.8 vs 99.7 + 100 gm/sec, p=0.282) were found.
Acute PV reconnection rates using CF sensing catheters are roughly 12.5%, with the majority occurring within 20 minutes. We found no significant differences in characteristics of ablation points in areas of reconnection. Optimum wait periods after isolation to check for acute reconnection may be as brief as 20 minutes.
Key Words : Contact-force Sensing Catheter, PV Reconnection, Atrial Fibrillation Ablation.
Correspondence to: Dr. David Spragg
Johns Hopkins Heart and Vascular Institute
600 N. Wolfe Street The Johns Hopkins Hospital
Baltimore, MD 21287
Catheter ablation is the cornerstone of rhythm management in patients with drug refractory symptomatic atrial fibrillation (AF) [1] Durability of pulmonary vein isolation (PVI) is important to procedural success; both acute and late pulmonary vein (PV) reconnection have been associated with atrial fibrillation recurrence. [2,3] Multiple studies have reported a high rate of PV reconnection within 60 minutes of initial isolation[4-6]. Indeed, it is because of this acute reconnection phenomenon that both the 2012 and 2017 Heart Rhythm Society Expert Consensus Statements on catheter ablation of AF recommend a 20 minute observation period and reassessment of PV sleeve conduction following initial PV isolation[1,7]. Of note, these recommendations were made based on reconnection data obtained during RF ablation with non-force sensing ablation catheters.
Catheter systems capable of providing contact force data likely facilitate improved lesion delivery[
8,
9] during AF ablation. Whether a 20 minute waiting period and PV reassessment is still needed after initial isolation in the era of force sensing catheters remains largely unanswered. Data regarding PV reconnection patterns in the current era has important clinical relevance, as it may allow for procedures to be simplified and streamlined. In the present study, we aim to assess the incidence and timing of acute PV reconnection during catheter ablation using contact force sensing catheters, and to assess any differences in lesion characteristics at sites of acute PV reconnection in comparison to sites of effective ablation.
Patients undergoing CF-guided catheter ablation for AF at Johns Hopkins Hospital between October 2016 and February 2017 were included in the study as part of a prospective database. All patients included in the current study were undergoing index procedures for either paroxysmal (n=14) or persistent (n=8) AF. All participants signed written informed consent and the study was approved by the institutional review board at Johns Hopkins hospital.
Ablation was performed under general anesthesia, with pre-procedure trans-esophageal echocardiogram done in patients deemed high-risk for LAA thrombus. Femoral site access was obtained with placement of three venous vascular sheaths followed by advancement of catheters in the coronary sinus and His bundle for measurement of intracardiac electrograms and conduction. Intravenous heparin was administered to maintain activated clotting time (ACT) >350 seconds. After performing a double trans-septal puncture, a Lasso circular mapping catheter (Biosense-Webster, Inc., Diamond Bar, CA) was positioned in the left atrium. An electroanatomic map of the left atrium was obtained using the CARTO System (Biosense-Webster, Inc., Diamond Bar, CA), and superimposed on pre-acquired CAT-Scan or MRI images. A 4mm open-tip irrigated RF Catheter with CF sensor (Thermocool SmartTouch, Biosense-Webster Inc., Diamond Bar, CA) was then positioned in the left atrium: Wide area circumferential ablation of pulmonary veins was performed, with carinal ablation performed between superior and inferior veins, using real-time automated display of RF application points (Visitag; Biosense-Webster Inc.). Visitags were applied with predefined catheter stability settings of range of motion ≤1.5 mm, duration ≥3 s, CF ≥5 g, and tag diameter at 2 mm. Starting energy delivery parameters were 25 Watts on the posterior wall and 35 Watts at other sites. Target contact force was between 10-20g at all sites. Esophageal temperature was monitored with a single sensor temperature probe, with position adjusted based on site of ablation and RF delivery paused if esophageal temperature increased by 0.5C. Electric isolation of pulmonary veins was confirmed by entrance block to individual PVs, assessed by Lasso catheter positioned at the PV antrum.
Assessment for Acute Reconnection:
Following confirmation of electrical isolation of a PV, we waited for a period of 20 minutes and assessed for acute reconnection; ablation of remaining PVs was typically performed during that waiting phase. Reconnection at 20 minutes was treated by further spot or segmental ablation until re-isolation was achieved. If no acute reconnection was identified, PVs were reassessed at 40 minutes after initial isolation. In cases where additional RF ablation was required due to reconnection, assessment was performed 20 minutes after repeat isolation. Reconnection observed after a 40 minute waiting period was treated with additional ablation, but delayed reassessment was not performed.
Continuous variables are expressed as mean+ SD and categorical variables as percentage. Univariable analysis was done using t-test and chi-squared test where appropriate. P values of <0.05 were considered statistically significant. All statistical analysis was done using the Stata Software (StataCorp, College Station, TX).
Twenty-two patients undergoing initial catheter ablation were evaluated for acute pulmonary vein reconnection. Baseline characteristics are outlined in [Table 1]. Mean age was 60.6 + 1.8 years. The cohort included 36.4% women and 27.3% with persistent AF. Mean CHA2DS2VASC score was 1.9 + 0.3 and duration of AF since diagnosis was 2.0 + 0.5 years. Average RF application time was 39.1 + 2.5 mins and procedure time was 222.8 + 6.2 mins.
Table 1. Baseline characteristics of study group.
| Variable |
Study group (n=22) |
| Age (years) |
60.6 + 1.8 |
| Female sex |
36.4 % |
| BMI (kg/m2) |
29.6 + 1.4 |
| Hypertension |
72.7 % |
| Diabetes Mellitus |
18.2 % |
| CHF |
9.1 % |
| CAD |
9.1 % |
| PVD |
4.6 % |
| OSA |
18.2 % |
| CHA2DS2VASC |
1.9 + 0.3 |
| Persistent AF |
27.3 % |
| AF duration (years)* |
2.0 + 0.5 |
| LA size (cm) |
4.3 + 0.2 |
| LVEF (%) |
55 + 1.6 |
| Presenting rhythm AF |
27.3 % |
| RF application time (min) |
39.1 + 2.5 |
| Procedure time (min) |
222.8 + 6.2 |
BMI= Body mass index; CHF= congestive heart failure; CAD= coronary artery disease; PVD= Peripheral vascular disease; OSA= Obstructive sleep apnea; CHA2DS2VASC = congestive heart failure, hypertension, age > 75 years, diabetes mellitus, stroke, vascular disease, age 65-74 years, sex category; LA= left atrium; LVEF= left ventricular ejection fraction; RF= radiofrequency. * time since diagnosis of AF.
Acute pulmonary vein reconnection:
Each patient in the study had 4 discreet PVs. Of the 88 PVs assessed,[11] (12.5%) showed reconnection during the observation window. In 10 patients, 9 PVs reconnected by 20 minutes and 2 between 20 - 40 minutes. Of these, six reconnections (5 at 20 mins, 1 at 40 mins) were in the left superior PV; two reconnections (both at 20 mins) were in the left inferior PV; there was only one reconnection (at 20 mins) in the right superior PV; and two reconnections (1 at 20 mins and 1 at 40 mins) in the right inferior PV. Sites of acute reconnection are presented in [Figure 1].
Figure 1. Schematic of the posterior LA, showing sites of PV reconnection occurring 20 minutes after PVI (blue) or between 20 and 40 minutes (orange). LSPV –left superior pulmonary vein; LIPV –left inferior pulmonary vein; RSPV –right superior pulmonary vein; RIPV –right inferior pulmonary vein.
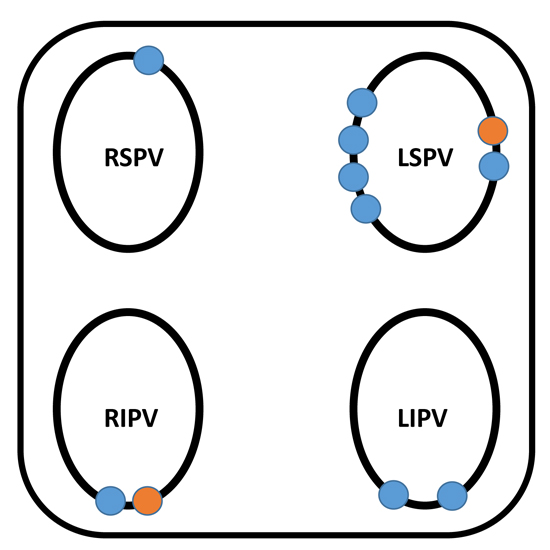
A total of 4993 ablation points were evaluated. Of these, 72 were located in regions of acute reconnection. Comparison of characteristics of the two different point groups is outlined in [Table 2]. The average contact force (11.4 + 8.1 vs 11.3 + 7.1 gm, p=0.868), power (29.7 + 3.9 vs 29.9 + 4.6 watts, p=0.620), impedance (64.1 + 60 vs 72.5 + 60, p=0.236) and the force time integral (86.9 + 78.8 vs 99.7 + 100 gm/sec, p=0.282) did not show any significant difference.
Table 2. Comparison of characteristics of ablation points in areas with and without acute reconnection.
| Variable |
Reconnected area points (n=72) |
Isolated area points (n=4921) |
P value[sig < 0.05] |
| Application Time (sec) |
8.9 + 7.0 |
9.0 + 6.8 |
0.843 |
| Contact Force (gm) |
11.4 + 8.1 |
11.3 + 7.1 |
0.868 |
| Temperature (F) |
33 + 2.7 |
33.6 + 2.9 |
0.077 |
| Power (Watts) |
29.7 + 3.9 |
29.9 + 4.6 |
0.620 |
| Impedance (Ω) |
64.1 + 60 |
72.5 + 60 |
0.236 |
| Impedance drop (Ω) |
2.9 + 4.2 |
3.3 + 4.6 |
0.385 |
| Force time integral (gm/sec) |
86.9 + 78.8 |
99.7 + 100 |
0.282 |
One procedure was complicated by a pericardial effusion that was managed conservatively.
In the current investigation we sought to determine the incidence, time course, and location of acute PV reconnection in patients undergoing PVI with force-sensing catheters. In addition, we assessed lesion characteristics including power, contact force, and impedance changes at sites of reconnection and of successful ablation. The principal findings of our study are: 1) Acute PV reconnection occurs at a rate of roughly 12.5% in patients ablated with radiofrequency, CF-sensing catheters; 2) nine of 11 reconnections occurred within a 20 minute waiting period, and 8 of 11 reconnections were at left PV sites; and 3) there were no observable differences in lesion characteristics in areas of acute reconnection when compared to the ablation points in areas without reconnection.
Investigations of PV Reconnection
Acute PV reconnection during PVI is a well-known phenomenon, and has been associated with AF recurrence in long-term outcomes studies. [2,3] Several investigations, including previous work from our institution, have characterized the incidence, time-course, and sites of PV reconnection[4-6]. These studies, largely performed in the era of non-force sensing catheters, showed recurrence rates as high as 50% of all PVs assessed, affecting 93% of patients undergoing ablation. Based on these data, the published Expert Consensus Statements addressing catheter ablation of AF recommend at least a 20 minute observation period followed by reassessment of PV isolation[1,7]. Whether this suggestion is accurate and necessary in the era of PVI with force-sensing catheters is less well established.
Two studies have provided initial data about acute PV reconnection rates using force-guided ablation. In a study by Martinek and colleagues [10] , acute PV reconnection rates were compared in patients undergoing PVI using non-force sensing versus force-sensing catheters. The investigators found that on immediate re-assessment of PV isolation (following completion of acute isolation of the last PV targeted), reconnection was reduced from a rate of 36% (in patients ablated with non-force sensing catheters) to 12% (in patients ablated with force-sensing catheters). No data was provided about the time course of reconnection, however. Our results, in terms of the incidence of reconnection, are quite similar (12.5% reconnection rate), but extend the study of Martinek by outlining the time windows during which reconnection was noted. The observation that 9/11 reconnections occurred within 20 minutes, and that 8/11 reconnection sites were located in the left PVs, provides a reasonable guide for limiting reassessment both temporally and spatially.
A second investigation by Haldar and colleagues11 randomized patients to operators blinded to or informed of contact force during PVI. Reconnection rates were assessed at a single time period (60 minutes). Knowledge of contact force during ablation reduced 1h PV reconnection rates from 21% (blinded group) to 4% (force-guided group). This study, while remarkable for the very low PV reconnection rate observed, does not provide data of the sort provided in the current investigation on rates of PV reconnection versus time. It is knowledge of this kinetics of reconnection, in part, that we think will help inform decision making on when and where to investigate for sites of reestablished conduction following initial vein isolation.
Lesion Characteristics at Reconnection Sites
The idea of the “weakest link” predicting sites of reconnection was described recently in a novel investigation by El Haddad and colleagues[12], who developed an ablation line contiguity index incorporating ablation lesion characteristics (force, time, power) and inter-lesion distance. They found that sites of reconnection were associated with poor lesion depth and lack of contiguous lesion sets. In our investigation we did not find clear differences in the force, time, power, or impedance drop seen in sites of reconnection compared to isolated segments. Visual assessment of Visitag lesions (i.e. algorithm-driven, rather than subjectively applied) on CARTO maps did not suggest discontinuity of lesions in any targeted PV. The fact that lesion characteristics at reconnection sites in our investigation were not quantitatively different from effective ablation sites suggests that other factors (e.g. tissue thickness) may play a role in lesion efficacy.
We found that rates of acute PV reconnection were relatively low, occurred typically within a 20m observation window, and were located most often at the left-sided veins. Waiting for 40 minute detected 2.3% (2/88) of additional acute reconnections of the total isolated veins. Based on those observations, a reasonable work flow would be to initially target the left PVs, to then proceed to right-sided PV isolation, and to check the left PVs (only) for durable isolation after completing isolation of the RSPV and RIPV (which presumably will take 20 minutes or more). We propose that if the left PVs are isolated at that point, further assessment may be not necessary. Clearly this approach is worthy of investigation in a prospective, randomized study.
This is a single center non-randomized study with a relatively small sample size. Although the total number of ablation points analyzed was fairly large (n=4993), those associated with acute reconnection were much fewer in number (n=72). In this study pulmonary reconnection was confirmed with entrance block. Adenosine was not used to check for dormant conduction.
Rates of acute PV reconnection during PVI performed with contact force sensing catheters is low, occurs most often within a 20 minute interval, and is typically located at the left PVs. This data suggests that a streamlined approach to PVI, with reassessment of the left PVs at 20 minutes or later after initial isolation, may be feasible. The effect of that streamlined approach on efficiency, safety, and long-term efficacy is worthy of prospective, randomized investigation.
Funding for this research was provided in part by the Edward St. John Fund for AF Research, The Roz and Marvin H Weiner and Family Foundation, The Dr. Francis P. Chiaramonte Foundation, The Marilyn and Christian Poindexter Arrhythmia Research Fund, The Norbert and Louise Grunwald Cardiac Arrhythmia Research Fund and Mr and Mrs Larry Small AF Research Fund.