Post Ablation Left Atrial Tachycardia: Understanding Mechanism, Prevention and Treatment
Carlo Pappone, MD, PhD, FACC, Vincenzo Santinelli MD.
Department of Arrhythmology, Maria Cecilia Hospital, GVM Care and Research, Cotignola, Ravenna, Italy.
Currently, post-ablation Atrial Tachycardias (ATs) represent a growing clinical problem particularly in patients with persistent AF undergoing a more extensive substrate ablation. Understanding mechanisms and location of potentially widely located arrhythmogenic substrates in the left atrium is crucial for successful ablation. Mapping and ablation are challenging since complex and multiple ATs may frequently develop during the index procedure and before conversion to sinus rhythm. Use of irrigated ablation guided by detailed 3-D electroanatomic activation maps combined with entrainment pacing is effective with excellent acute and long-term success rates, rarely requiring multiple procedures.
Correspondence to: Carlo Pappone, MD, Department of Arrhythmology, Maria Cecilia Hospital, Via Corriera 148010 Cotignola, Ravenna, ITALY.
Within the next twenty to thirty years, the number of patients suffering from Atrial Fibrillation (AF) is expected to double or triple due to an increased AF incidence and aging of the populations in developed western countries. Radiofrequency catheter ablation (RFA) for AF has become one of the most common ablation procedures performed in many centers worldwide. At present, this new treatment option has gained prominence as an alternative to chronic antiarrhythmic drug administration and has been incorporated into the current guidelines for management of AF as class 1 with level of evidence A.1 With the exponential increase in catheter ablation for AF more patients with persistent AF are undergoing extensive substrate modification in an attempt to restore a stable sinus rhythm.2–7 Patients with persistent AF, however, frequently have associated comorbidities and larger atria requiring a greater amount of left atrial ablation than patients with the paroxysmal form, thus fac ilitating the occurrence of post-ablation left Atrial Tachycardias (ATs). Unlike paroxysmal AF8–21 where rapid firing from PVs is the main cause, the mechanisms of persistent long-standing AF are complex involving a significant amount of arrhythmogenic substrate widely distributed in both atria in addition to PVs. As a result, a greater amount of substrate ablation creates an electrophysiologic milieu for both small circuit and macroreentry facilitating the development of ATs.5–7,22–28 This has led to a significant increase in the incidence of ATs among patients with AF undergoing catheter ablation and a need to re-evaluate our understanding of their mechanisms and management. The development of post-ablation left ATs after PV isolation alone (PVI) is considered to be an uncommon complication requiring a redo procedure in a minority of cases.8–21 Typically, after PVI patients complain of worsening symptoms due to a faster ventricular rate than during pre-ablation. However, s ymptoms at rest are easily controlled medically although in some cases a good rate control with activity may be challenging. Catheter mapping and entrainment pacing are able to identify the arrhythmia location and mechanism frequently originating from reconnected PVs, resulting in successful catheter ablation. Left ATs occurring in patients with persistent AF undergoing extensive catheter ablation of the arrhythmogenic substrate are more challenging to both map and ablate, often requiring repeat procedures because of unpredictable location and extent of the substrate .5,6 Most of these ATs before AF termination convert to intermediate ATs with varying cycle length or location supporting the presence of multiple drivers simultaneously operating to maintain the arrhythmia perpetuation. Currently, post-ablation ATs poses specific challenges to all electrophysiologists and this article reports new information about incidence, electrophysiological mechanisms and management of such ATs in patients with AF.
The classification of post-ablation left ATs is based on mechanism and anatomy. Left ATs are classified as tachycardias with a regular atrial rate arising from the left atrium and are further categorized as either focal or macro-reentrant. The differentiation has important implications for ablation therapy, because macro-reentrant ATs require mapping of large segments of the circuits and linear lesions by delivery of a single or multiple point-by-points RF applications for arrhythmia termination. Focal ATs are due to automatic, triggered, or micro-reentrant mechanisms, usually from reconnected PVs or LA. Focal AT is defined as follows: atrial activation radiating centrifugally in all directions from a single site of earliest activation and range of activation duration less than the AT cycle length. Macroreentrant AT is defined as follows: 1 continuous sequence of atrial activation, with earliest activation adjacent to latest activation; 2 range of activation times > 90% of the AT cycle length ; an d3 demonstration of entrainment with post-pacing interval approximated the AT cycle length in at least 3 disparate atrial sites. Typically, ATs are considered to be left atrial in origin if the distal coronary sinus atrial electrogram preceded the proximal coronary sinus atrial electrogram or in any case if left atrial electrograms are recorded throughout diastole with the ablation catheter.
Incidence of post-ablation left ATs
The incidence of post-ablation left ATs occurring during and after catheter ablation of AF varies significantly ranging from 4 up to 50% and is critically dependent on the type and extent of the index ablation strategy, LA diameter, and completeness of lesion lines.2–28 Catheter ablation of paroxysmal AF is associated with a low incidence of left ATs (about 5%) predominantly as a result of PV reconnection. The incidence of ATs increases further if PV isolation is not confirmed as the electrophysiologic endpoint. In our experience, about 4% of patients with paroxysmal AF undergoing CPVA (Circumferential Pulmonary Vein Ablation) which is PVI plus additional lines, may experience post-ablation left ATs as compared with 10% of patients who undergo PVI (Pulmonary Vein Isolation) alone suggesting that validation of bidirectional mitral isthmus block is critical to reduce the risk of macroreentrant ATs due to mitral isthmus reentry.8 A more extensive ablation procedure, as perform ed by the stepwise approach in chronic AF, is associated with a further significant increase in the incidence of post-ablation ATs with studies reporting ATs > 50%.14
Mechanisms of post-ablation left ATs
The electrophysiological mechanisms underlying post-ablation ATs include abnormal automaticity, micro or macroreentry due to conduction gap on previous ablation lines. Distinguishing focal from macroreentrant ATs is crucial for a successful catheter ablation. Iatrogenic ATs have been reported after either surgical or catheter ablation of AF as a proarrhythmic complication, if linear lesions are incomplete. Focal and/or localized microreentry from reconnected PV ostia are commonly seen in patients undergoing PVI alone (Figure 1–2) while macro-reentry is more likely to be seen following linear lesions in addition to PVI, as in CPVA (Figure 3–4). In our experience, single or multiple gaps and foci may develop more frequently at the anterior aspect of the left PVs and at the septal aspect of the right PVs or at LA roof.8,12 It is not surprising that the mechanisms underlying post-ablation left ATs in patients in whom multiple extensive ablation strategies are simultaneously used (PVI pl us multiple sequential linear lesions and/or sites of complex fractionated electrograms) are complex, often multiple and not yet well defined.2–28 Ablation of persistent AF typically involves a more extensive arrhythmogenic substrate which increases the risk of iatrogenic ablation-related ATs due to multiple gaps and micro or macro-reentrant circuits. Using an extensive complex fractionated electrogram ablation strategy, Nadamanee et al29 reported that 36% of patients had ATs, with half having macro-AT and half having focal mechanisms. The potentially extensive nature of the CFAE (Complex Fractionated Atrial Electrogram) ablation strategy based on "learning by burning" is potentially proarrhythmic and post-ablation ATs may be more symptomatic than the index arrhythmia. In patients undergoing a step wise ablation approach for persistent long-lasting AF, multiple often-changing ATs are frequently encountered before restoration of normal sinus rhythm probably excluding the proarrhythmic mec hanism in their genesis. Characteristically, such ATs may be localized to virtually all aspects of the atria and thoracic veins with circuits incorporating areas of slow conduction particularly in regions of fractionated atrial electrograms, but when occurring after ablation, they are rarely dependent on or related to reconnected PVs.5 Occasionally, they can be localized around the fossa ovalis, the coronary sinus, the septum or the posterior wall. During the index procedure a progressive organization of AF is usually associated with an increase of AF cycle length until sinus rhythm conversion or to intermediate ATs which may become slower or may accelerate to a faster AT with or without CL or activation sequence changes. These observations suggest that an extensive sequential ablation in patients with persistent long-standing AF may result in a necessary arrhythmia defragmentation to achieve a stable sinus rhythm, but the surrounding mechanisms are unknown. In our experience, direct transition to a new AT is frequently seen before sinus rhythm conversion and then it may be considered as an "organization" of the arrhythmia from AF to AT requiring an accurate mapping and more ablation. However, mapping and successful ablation of such ATs are challenging requiring availability of multiple simultaneous recording sites and several entrainment attempts at the time of AT transition. In our experience, some of these macro-ATs have a very short cycle length (150-200 ms), mimicking AF. Characteristically, before sinus rhythm is achieved, the cycle length of ATs gradually increases as the number of eliminated intermediate subsequent ATs increases. The reentrant circuits may be localized to multiple and unusual atrial areas and may not rotate around predefined anatomical obstacles. Single or multiple concealed AF driving circuits may become manifest by entrainment (reentrant circuits) or by overdrive (automatic focal circuits). These observations are intriguing and suggest that although the proarrhythm ic theory seems to be the widely favored hypothesis to explain the development of new post-ablation ATs, a contributing role of intermediate ATs to the AF process by multiple simultaneous mechanisms should be taken into account.
Figure 1. A color-coded three-dimensional electroanatomic activation map as performed by CARTO system shows a focal left AT from a reconnected left superior PV in the posterior-anterior (PA) view. Activation spreads away from this focal red region in all directions centrifugally
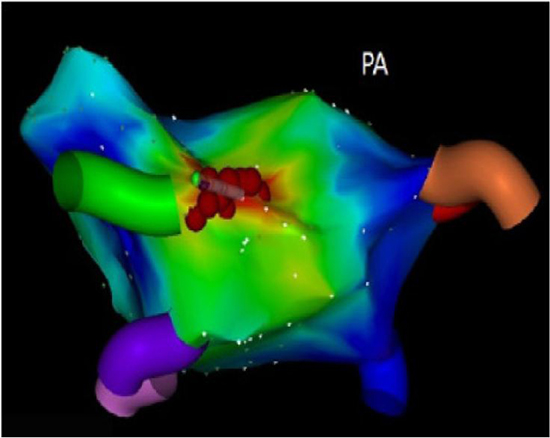
Figure 2. For the same patient in Figure 1, the activation map shows the activation sequence in the antero-posterior (AP) view. The AT cycle length was 348 ms; the activation time during mapping covered 100 ms before to 40 ms after the reference point accounting for a total of 140 ms, which is less than 40% of the AT cycle length of 348 ms, consistent with a focal origin of AT. The deep red dots around LSPV ostium indicate the sites where radiofrequency current was applied
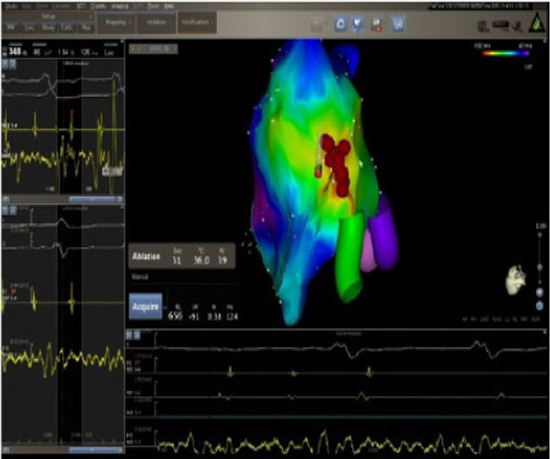
Figure 3. An activation map of a septal left macro-reentrant AT by NavX system in the antero-posterior (AP) view is shown. The map was created with a temporal reference located in the coronary sinus. Orientation of the left atrium is indicated by the orientation of the body at the top of the panel. Activation propagates from the middle septum (area of earliest activation, white color) to the anterior septum (area of latest activation, violet color). AT cycle length was 218 ms; the activation time during mapping covered 110 ms before to 85 ms after accounting for a total of 195 ms, which is more than 90% of the AT cycle length, consistent with a macro-reentrant circuit. Also shown is the reference catheter positioned in the coronary sinus
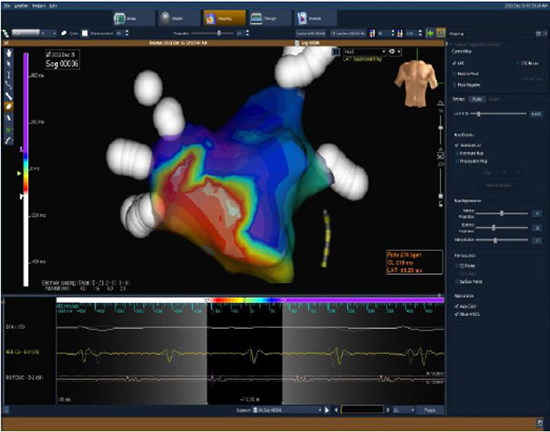
Figure 4. For the same patient in Figure 3, the activation map shows the late activation progression in scale color of the reentrant circuit in the postero-anterior (PA) view
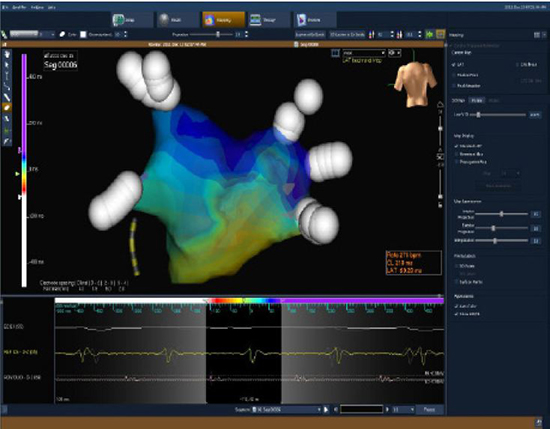
ECG characteristics in post-ablation AT
ECG localization of post-ablation macro-reentrant ATs is much more complex than localization of focal post-ablation ATs mainly because macro-circuits are influenced by altered anatomy, previous ablation lines, and conduction of the atrial wavefront. Analysis of P wave morphology, duration, and AT cycle length may be highly useful in defining the likely mechanism and site of focal ATs.31 The 12-lead ECG may be also helpful in suggesting a focal source for the LA tachycardia from a reconnected PV or macroreentry around the mitral annulus.27 In our experience, ATs after extensive LA ablation may create atypical surface ECG patterns during tachycardia making 12-ECG lead analysis limited. P-wave cycle length stability/variability with clear isoelectric line between the P-wave in all 12 ECG leads may differentiate focal from reentrant AT. Stable AT cycle length with long P wave duration without any or short isoelectric intervals and concordant positive P-waves from V1-V6 suggest the presence of a left atrial macro-reentrant AT. ATs arising from the PVs are characterized by entirely positive P waves in lead V1 in 100% of cases, isoelectric or negative in lead aVL in 86%, and negative in lead aVR in 96%. Lead aVL can be biphasic or positive in right-sided PV ATs. Left PV ATs have several characteristics (as compared with right PV ATs): positive notching in the P waves in two or more surface leads, an isoelectric or negative P wave in lead I, P wave amplitude in lead III/II ratio > 0.8, and broad P waves in lead V1. ATs arising from the superior PVs have larger amplitude P waves in the inferior leads than those in ATs arising from the inferior PVs. However, P wave morphology generally is of greater accuracy in distinguishing right-sided from left-sided PVs in contrast to superior from inferior PVs. Despite prior posterior LA ablation, the surface ECG morphology of ATs originating from the PV ostia in patients with prior AF ablation procedures are similar to those in patients without prior ablation. However, ATs originating from the bottom of the right or left PVs after prior PV isolation can have a significant negative component or can be completely negative in the inferior leads. This may be related to prior ablation in the supero-posterior LA or to a more inferior origin of the tachycardia after prior ablation outside the PV ostium. ATs arising from the right superior PV are associated with P waves that are narrow, positive in the inferior leads, of equal amplitude in leads II and III, biphasic or slightly positive in lead V1, and isoelectric in lead I . Recently, it has been reported that achievement of mitral isthmus block is associated with change of P wave morphology in lateral leads which further emphasizes the usefulness of ECG analysis in the management of patients with ATs.32
Mapping and Ablation of AT
To determine both mechanisms and location of post-ablation ATs conventional manual or 3-D electroanatomic mapping systems are currently used. Conventional mapping system requires several intracardiac catheters (usually up to 3 catheters) to pace and map the direction of activation with a fixed atrial reference catheter. By combining activation mapping and entrainment mapping at several sites under extensive use of fluoroscopy the mechanism of the arrhythmia may be identified. The advent of computer mapping techniques with a variety of 3-D mapping tools has allowed a more precise and rapid identification of arrhythmia mechanisms for better defining sites guiding mapping and successful catheter ablation, even in cases where multiple circuits are involved.25 Many systems also offer algorithms for the automatic analysis of complex fractionated atrial electrograms (CFAEs). Image integration with cardiac CT or MRI may further improve the accuracy of left atrial anatomy, particularly in pati ents with left atrial enlargement. These mapping systems help determine the three-dimensional position of the ablation catheter more accurately than fluoroscopy alone and are useful in evaluating the completeness of linear lesions minimizing unnecessary ablation. Before the advent of 3-D systems, a mapping strategy with at least 3 manual catheters and entrainment pacing at several sites under extensive use of fluoroscopy has been used and continues to be used. Since the use of manual catheters to map and ablate post-ablation left ATs is time consuming and frequently requires an extensive use of fluoroscopy to demonstrate the arrhythmia site and mechanism, a practical approach combining both activation and entrainment mapping has been recently suggested.24,33 Since 1999 we use a combined strategy of 3-D mapping systems with entrainment pacing8,9 which is successful in almost all cases of post-ablation ATs without use of fluoroscopy so that manual mapping catheters are rarely used, if nec essary. In the last 3 years we have performed about 800 successful procedures in patients with post-ablation left ATs in patients with AF. Three-dimensional electrical information are collected by a specific mapping catheter all of which are projected into a detailed electroanatomic activation map, but a correct interpretation of the colors of the map requires an appropriate choice of both the reference electrogram and window of interest as well as an appropriate number of points (Figure 1–4). An accurate reconstruction of focal or reentrant circuits before ablation is made by a meticulous analysis of data points which are continuously acquired during catheter movements (Figure 1–7). Different colors on the map indicate early or late sites of activation relative to an arbitrarily chosen reference point. Depending on each 3-D mapping system, red or white colors indicate an early activation while yellow-green and finally the blue or violet colors indicate progressively late activation (Figure 1–9). In the p resence of a focal mechanism, the earliest activation generally represents the optimal and successful site of RF application (Figure 1,2,6) and usually precedes the P wave by at least 30 ms. Interpretation of 3-D color-coded activation maps of macroreentrant AT may be challenging since in reentrant ATs there is no true earliest or latest activation. Therefore, the entire reentry circuit should be mapped with a total activation time equal to the AT cycle length with adjacent earliest and latest activation (head meets tail; Figure 3,4,8,9). In our experience, the substrate for left macro-ATs usually is represented by a large heterogeneous atrial fibrosis which on 3-D electroanatomic voltage map performed in sinus rhythm is manifested by a large area of low bipolar voltage (<0.1mV). The selection of a stable reference point during ATs mapping with an appropriate window of interest is indeed critical for a correct activation mapping sequence and successful ablation. We usually set the activation timing window to a value that encompasses the cycle length of AT mostly depending on the location of presumed reentrant circuit and on the distance from the chosen reference point (Figure 3,4,8,9). For example, in the presence of a macroreentrant AT with cycle length of 218 ms as in Figure 3, we set the timing window of interest for mapping to at least -120 ms (before the reference coronary sinus potential) to +80 ms (Figure 3) to be sure to include earliest and latest activation of the reentrant circuit. If an incorrect temporal reference is used for the same tachycardia an uncorrected color-coded map is constructed (Figure 5). Unlike reentrant ATs, focal ATs do not require a precise window of reference since the site of activation is rarely earlier than 50 ms from the P wave depending on its distance from the reference point. We use the coronary sinus catheter as stable reference point also in consideration that concordant respiratory movement of the coronary sinus and left atrium is of benefit in facilitating respi ratory gating. The local activation time, recorded from the catheter tip at each mapping site, is measured relative to the timing of the reference atrial potential. We routinely pace many sites within the LA for entrainment mapping as guided by electroanatomic activation maps and sites with the shortest return cycle (generally < 20 ms longer than the tachycardia cycle length) are considered as critical for successful ablation. However, in our experience entrainment pacing has 2 major limitations which may preclude identification of critical isthmuses for ablation. First, it may produce the same atrial activation sequence and P wave (concealed fusion) with post-pacing interval equal to AT cycle length at many sites and/or it may occasionally terminate AT or may convert reentrant AT to another reentrant AT. In these cases, visualization of multiple reentrant or multiple focal circuits on electrical activation maps may be challenging and time consuming as individual maps are required for each AT. The use of multielectrode catheters with high-density 3-D electroanatomic mapping may be useful to rapidly create sequential activation maps every time the circuit location of ATs is changing to another AT which requires rapid remapping of the new AT thus facilitating the identification of critical isthmuses for successful ablation by a single RF application.25 Focal or microreentrant ATs are also represented on color coded activation maps with the earliest activation at the center of a spread of colors in an omnidirectional fashion. Unlike macroreentrant ATs, focal ATs have a point source with centrifugal activation from the center and usually require discrete ablation at the critical spot as defined on 3-D electroanatomic activation maps until AT is eliminated (Figure 1,2,6). In localized reentrant ATs, the target site is the key isthmus as defined by an electroanatomic map, usually the latest activation. In our experience, macro-reentrant ATs may be rarely ablated by a single RF application because iden tification and ablation of critical isthmuses is challenging. We routinely use long linear lesions with irrigated tip catheters guided by detailed electroanatomic mapping systems for successful ablation of macro-reentrant ATs (Figure 1,2,6,7,9). For mitral isthmus ablation we perform a point-by-point 3-D electroanatomic-guided line which is drawn cranially from the mitral annulus to the left inferior PV (Figure 9). Irrigated ablation is always used with a power of up to 35 W and continuous titration of flow from 2 to 30 mL/min to achieve a target temperature of up to 40 degree. Epicardial ablation in the coronary sinus with atrial voltage abatement on the endocardial aspect is required in about 30% of cases to achieve completeness of block. During ablation within the coronary sinus we limit the power to 25 W while increasing the flow up to 30 mL/min without serious complications. Roof dependent ATs also require a long linear line (roof line). Usually, focal ATs are ablated at earliest site with power settin g up to 35 W in the anterior LA and up to 30 W in the posterior LA while localized reentry is ablated at slow conduction areas with shorter post-pacing interval. The endpoint of the ablation procedure is AT elimination and noninducibility. After sinus rhythm restoration we reassess completeness of linear block by differential pacing at the cavotricuspid isthmus, roof and mitral isthmus.
Figure 5. Same map as in Figure 4. The activation map shows changes in the color-coded activation sequence caused by changing window of interest settings. Frames of the window of interest extend from 130 ms before to 65 ms after the reference point. The reference point is the same as well the AT cycle length and the duration of window of interest, but the early area is markedly shifted from middle septum to the posterior wall with changes in activation sequence
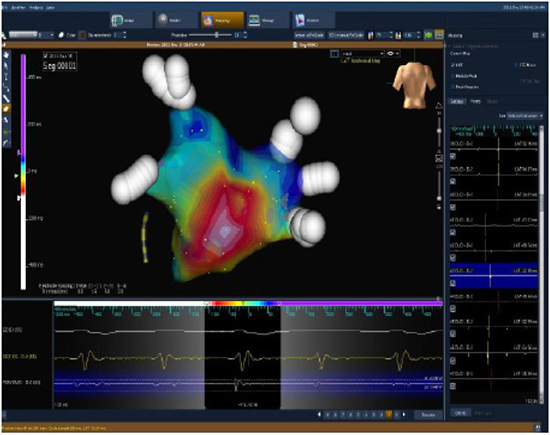
Figure 6. AT ablation guided by an activation map of a left focal microreentrant AT from reconnected PVs by CARTO system in the antero-posterior (AP) view. A point-by-point long-linear lesion around LSPV ostium resulted in termination of AT
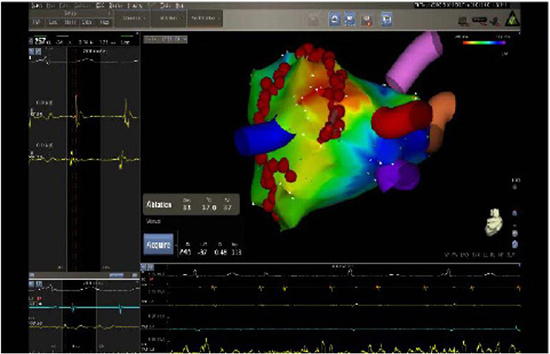
Figure 7. For the same patient in Figure 3, a point-by-point long-linear lesion line on the septum resulted in termination of a macroreentrant septal AT. The yellow circle marks the successful site of AT termination
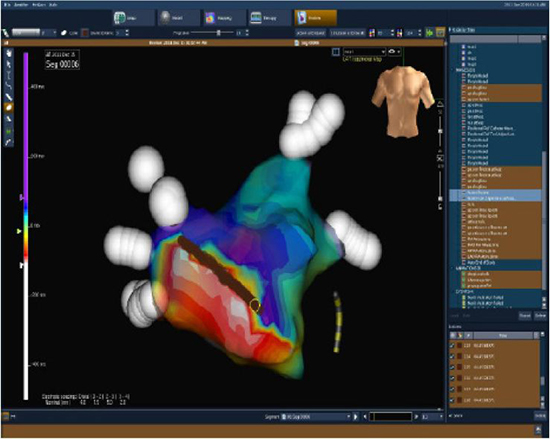
Figure 8. A color-coded activation map sequence by NavX system of a mitral macro-reentrant AT rotating around the mitral annulus in different views is shown. RAO indicates right anterior oblique; AP indicates antero-posterior and PA postero-anterior
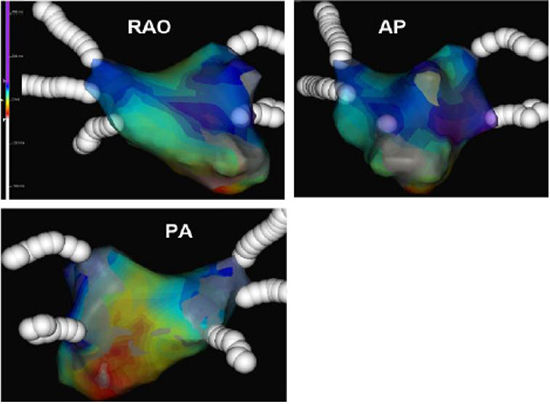
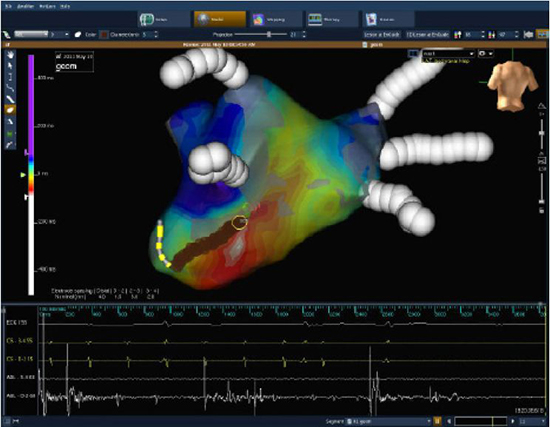
Figure 9. For the same patient shown in Figure 8, perimitral AT ablation guided by three-dimensional electroanatomic activation map is shown in the postero-anterior (PA) view. Orientation of the left atrium is indicated by the orientation of the face at the top right corner of the panel. A point-by-point long-linear lesion (brown dots) connecting mitral annulus to the inferior left PV ostium resulted in termination of AT with restoration of normal sinus rhythm (bottom panel). The yellow circle marks the site of successful ablation. On the bottom: ECG, intracardiac recordings from coronary sinus (CS), and ablation catheter electrograms (ABL) during successful termination
Unlike paroxysmal AF, mechanisms, prognosis and management of post-ablation left ATs in persistent AF are complex and not well defined. In this patient population a greater amount of bi-atrial ablation by multiple linear lesions or ablation of complex fractionated electrograms results in higher incidence of new iatrogenic or organized ATs either during or after the index procedure The mechanisms of such ATs are complex and may be simultaneously active in maintaining perpetuation of the arrhythmia. Mapping and ablation of such complex ATs are technically challenging as they are unpredictable and can be widely distributed throughout the atria. In our experience, use of irrigated ablation guided by detailed 3-D electroanatomic activation maps combined with entrainment pacing is effective with excellent acute and long-term success rates, rarely requiring multiple procedures.
Dr. Carlo Pappone received > 10,000 US Dollars from St.Jude Medical and > 10,000 USA Dollars from Biotronik Spa as direct personal payment (Speaker fee, honoraria, consultancy or investigator).
Dr. Vincenzo Santinelli has no financial disclosures to declare.