Atrial fibrillation (AF) is a common, distressing cardiac arrhythmia, occurring in 1–2% of the general population. Radiofrequency (RF) catheter ablation of AF is an established treatment for the management of symptomatic drug refractory patients. Pulmonary vein (PV) isolation is the cornerstone of treatment for any type of AF.1,2 In order to facilitate ablation and reduce the risk of PV stenosis, it is widely accepted that ablation should be undertaken on the atrial side, forming a circumferential lesion around one or both ipsilateral PVs.3,4 For persistent AF, the ablation strategy is still controversial among leading centers. However, ablation targeting fractionated signals and linear ablation in addition to PV isolation are popular strategies.5 These procedures are generally effective and safe, but devastating complications may occasionally occur, some of them ultimately leading to death of the patient.6 Concerns include collateral damage to extracardiac structures, such as esophageal trauma, phrenic nerve (PN), and vagal nerve injury. This review focuses on the collateral damage from catheter ablation of AF.
Esophageal injury and esophageal fistula are considered life-threatening complications that may occur after RF is applied in the posterior wall of the left atrium (LA).7-9 In patients undergoing RF catheter ablation of AF, the reported incidence of this complication is around 0.01%.10,11 Pappone et al. initially reported 2 cases of esophageal fistula.7 One of the patients died, and the other survived after surgery. Scanavacca et al. described a case of esophageal fistula after ablation with an 8-mm catheter using a maximum power of 60 W and a maximum temperature of 55°C.8 Nowadays this complication is widely recognized as one of the important complications due to AF ablation and several methods to avoid this complication have been proposed.
The Location of Esophagus
Understanding the anatomical relationship between the esophagus and LA is important. The relationship is variable in each patient. Pre-procedural esophageal location can be identified by computed tomography (CT) after a barium swallow, magnetic resonance imaging (MRI) using swallowed barium sulfate paste and gadolinium diglutamate,12,13 although they give a static picture of the esophagus and cannot evaluate the possibility of esophageal motion during the procedure. The anatomic location of the esophagus may change during ablation, and therefore registration of a preacquired static picture of the esophagus might not be adequate. In addition, fluoroscopic guidance may be misleading to avoid damage to the esophagus because it does not provide the border of the esophageal tube. Intracardiac echocardiography allows us to know the real-time monitoring of esophageal location.
The mechanism of esophageal injury is not completely known. Thermal injury seems to be the most likely cause (with area of necrosis surrounded by inflammatory cells), although an ischemic mechanism has been considered as well. Martinek et al. demonstrated that LA-to-esophagus distance is the only significant risk factor for occurrence of esophageal ulceration following AF ablation.14 Grubina et al. reported a case with esophageal pericardial fistula following AF ablation in which natural history of esophageal injury could be observed.15 This case suggested that esophageal thermal injury may be the preceding event with subsequent esophageal inflammation and perforation. That is, acid enzymes and possibly food material may then violate the integrity of the adjacent pericardium, creating an esophageal fistula. According to the data on monitoring esophageal temperature during the procedure, lesions placed within the LA near the esophageal course significantly increased the luminal temperature of the esophagus from baseline.16
Clinical Symptoms and Outcome
Nonspecific signs and symptoms, such as dysphagia, anorexia, chest pain or hematemesis, began 3–35 days after the catheter ablation procedure. Subsequently, endocarditis/pericarditis and finally embolism, including that associated with a myocardial infarction and cerebral infarctionoccurred.7,8,9Thoracic CT enhanced with the use of oral and intravenous contrast medium was the most useful examination method, and very specific findings of an air pocket or fistula tract were observed in the mediastinum. Mortality ranges from 75% to 83%, with rapid progression from symptom onset to death. Immediate recognition and emergent surgical intervention are therefore vital. The treatment for esophageal fistula is surgical, with only one documented attempt at esophageal stenting.17 Esophageal resection with end-to-end anastomoses and primary pericardial patching are usually required.
Several strategies have been described to help prevent or reduce gastroesophageal injury during AF ablation. The primary means of limiting esophageal injury during RF ablation relies on avoiding ablation sites adjacent to the esophagus. This is accomplished by detailed mapping of the cardiac-esophageal interface by pre- and/or intraprocedural imaging.12,13 Temperature probes were used to monitor energy delivery and temperature increase in the esophagus in other studies.18 As stated in these studies, luminal esophageal temperature monitoring has major limitations, such as malalignment of the probe to the RF catheter and posterior LA wall, the underestimation of intramural temperature by an intraluminal probe, and the too-slow esophageal temperature increase to prevent esophageal ulceration. Additionally, because parameters followed during energy delivery such as power or impedance are not predictive of temperature rises, it is difficult to predict the effects on the esophagus.
Reducing the RF power and/or contact force when ablating the LA posterior wall, mechanical esophageal deviation19 and esophageal cooling with a cooled water-irrigated intraesophageal balloon 20 might reduce the risk of esophageal injury.
Anesthesia protocol might influence the occurrence of the complication. Biase et al. reported that the use of general anesthesia seems to increase the risk of esophageal tissue damage.21 They speculated that the reduced motility and the reduced deglutination of the esophagus during general anesthesia result in ablation at the same esophageal location throughout the duration of the lesion. On the other hand, with conscious sedation, paincaused by RF delivery could trigger active peristalsis and swallowing, resulting in cooling and inconsistent heat transfer to the esophageal wall. Finally, when thermal injury to the esophagus is thought to have occurred, further damage by gastric contents can be minimized with judicious use of acid-lowering medications such as proton pump inhibitors or H2 receptor blockers.
Phrenic nerve (PN) injury can occur during a variety of catheter ablation procedures, such as for AF, Wolff-Parkinson-White syndrome, and ventricular tachyarrhythmias.22-24 Regarding AF ablation, right PN injury could occur during ablation targeting the right superior (RS) PV and the superior vena cava (SVC), and left PN injury occurs during ablation targeting the LA appendage. The prevalence of PN injury as a complication of endocardial catheter ablation has been reported around 0.1% to 0.48%.10,23,24
The course of the right PN is lateral and slightly posterior to the SVC and right atrium (RA), and descends anteriorly to the RSPV near the RSPV-LA orifice along side of the pericardium, anterior to the lung hilum before reaching the right diaphragm.25 A prior study showed that the minimum distance between the right PN and the RSPV or SVC was shorter than that between the right PN and RSPV-LA or SVC-RA junction.26
Inside the chest, left PN runs dorsal to the left brachiocephalic vein, anterolateral to the aortic arch and pulmonary trunk where it runs on the surface of the fibrous pericardium to course between the mediastinal pleura of the left lung and the lateral surface of the left ventricle. In the lung hilum, the nerve passes anterior to the left superior and inferior PVs. The relationships of the nerve to left heart structures depended on which of the following three courses the nerve took as it descended toward the cardiac apex: (1) along a course related to the anterior surface of the heart (anterior course); (2) passed leftward along a course related to the apex of the LA appendage and obtuse margin of the left ventricle (lateral course); or (3) passed posteriorly to the inferior (“posterior”) surface of the left ventricle (inferior course).27
Generally, RF, ultrasound, and laser energy create tissue lesions mainly by heating while cryoablation achieves lesions by hypothermic injury. Although some advantages of ultrasound, laser, or cryoablation have been addressed in relation to RF ablation, PN injury occurred regardless of the energy source used for ablation.28,29Moreover, this complication has been observed with a variety of catheters (4-mm, 8-mm, irrigated tip). Several mechanisms have been proposed to explain PNI after catheter ablation: 1) direct heat transfer from the ablation site to the nerve; 2) electromagnetic field generated at the catheter tip; and 3) generation of a resonance current around the heart. Bunch et al. investigated the mechanism of PN injury using dogs.30 The study showed that the PN tended to retain heat after ablation that may result in higher local temperatures with subsequent energy deliveries and that with direct nerve heating using radiofrequency energy, direct current effects and transient injury occurred early and at low temperatures. The study data demonstrate that transient nerve injury occurs early and uniformly before permanent injury. In areas at high risk of PN injury, which require ablation, PN pacing during the energy delivery could be used to detect early injury and allow termination of the ablation prior to the onset of permanent injury.
Clinical Symptoms and Outcome
Symptoms of PN injury are variable depending on patient’s preexisting lung condition. Typical symptoms are dyspnea especially while lying down, chest pain, atelectasis, pleural effusion and pleural infection. PN injury is diagnosed on a chest X-ray. Although PN function recovery occurs in the majority of patients (66% with full recovery, 18% with partial recovery) without need of special therapy,23 more serious complications in patients with compromised lung function can occur. Indeed, surgical series have suggested that once persistent PN injury occurred, it takes at least 2-3 months for the nerve regeneration.31 It is possible that clinical recovery does not equate to the restoration of normal cell and tissue characteristics. Whether this could have implications on the long-term function of the diaphragm is unknown. Since the clinical manifestations vary broadly from asymptomatic to severe respiratory dysfunction requiring prolonged mechanical ventilation and mortality following open heart surgery, the prevalence of PN injury may be greater than reported. In patients who develop dyspnea after atrial ablation procedures, particularly in the setting of preexisting lung disease, PN injury should be considered early in the differential diagnosis.
Although no technique has been previously described for preoperative location of the PN in clinical settings, Matsumoto et al. demonstrated that 64-slice multidetector CT allowed us to detect right and left PN and their relation to cardiac anatomic structures in more than half of the patients.32 Holton et al. also demonstrated that imaging the right pericardiophrenic artery using a 64-slice multidetector CT scanner can reliably locate the right PN before the ablation procedure.26 Imaging before a procedure may also prove useful for the planning of LA, left ventricle, and epicardial abla
tion procedures in which the course of the left PN may be avoided.
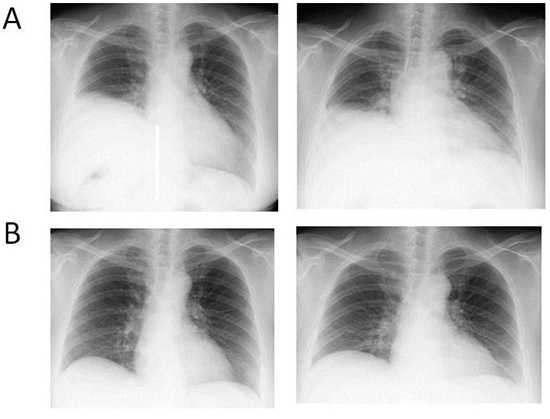
Figure 1. Typical example of right phrenic nerve injury after the AF ablation. Left panels show chest X-rays during maximal inspiration. Right panels show chest X-rays during maximal expiration. A. Right phrenic nerve paralysis was observed just after the AF ablation. B. The phrenic nerve palsy recovered little by little and finally completely recovered at 20 months after the procedure.
Pace mapping of the sites where the nerve can be stimulated from the endocardium has been the most commonly accepted real-time technique. The absence of PN capture within the pacing virtual electrode defines a space where RF delivery can be safely made. It is possible that ablating while monitoring PN function through high-output pacing might help in preventing inadvertent PN damage. The thickness of the atrium averages 4 mm, but decreases from 3.7 to 2.5 mm at the venoatrial junction. The PV tissue further decreases in thickness by 1–2 mm outside of the PV orifice.25 With less tissue available to receive and diffuse heat in the PV versus the atrium, energy delivery at or outside the orifice of the PV may have untoward collateral effects on PN. Recently, epicardial protection of PN has been reported in several reports. Lee et al. successfully eliminated AT arising from the crista terminalis region, which is close to right PN, by inserting a peripheral angioplasty balloon or steerable ablation catheter and sheath into the epicardial space between the PN and atrium.33 Biase et al.reported the feasibility of several methods to protect left PN in patients who underwent catheter ablation for VT or inappropriate sinus tachycardia, such as inflation of large balloon, introduction of air, introduction of saline, and introduction of both air and saline.34 These studies include a small series of patients; however they seem to be feasible approaches to prevent PN injury.
More recently, Franceschi et al. showed the compound motor action potential (CMAP) guided catheter ablation.35,36 They demonstrated that reliable diaphragmatic electromyographic signals could be recorded during cryoballoon ablation and that a reduction in the amplitude of the CMAP during pacing a right PN predictably presages hemidiaphragmatic paralysis.
Periesophageal Nerve Injury
Periesophageal nerve injury probably results from left atrially delivered RF energy affecting the periesophageal vagal plexus.37 Nerve fibers innervating the pyloric sphincter and the gastric antrum are thought to travel within the left vagal trunk [usually located on the left/anterior portion of the esophagus] but may also course within intercommunicating fibers. However, each cadaver was shown to have a distinctive anatomy, making any predictions impossible.
Clinical Symptoms and Outcome
Shah et al. reported the occurrence of acute delayed gastric emptying as a complication of percutaneous catheter ablation of AF in 4 cases.37 The clinical presentation of all four patients was very similar and was characterized by abdominal bloating and discomfort occurring within a few hours to two days after the procedure. Improvement occurred within two weeks in two patients who eventually recovered completely, whereas corrective interventions were required in the remaining two cases.
Because extracardiac penetration of endocardially delivered RF energy is almost certainly the basis of this complication, a strategy of reduced RF power and shorter RF delivery duration at vulnerable sites is advisable. Although knowledge of the position of the underlying esophagus may be of help, it is not clear to what extent this correlates with the location of relevant nerve fibers.
Esophageal temperature monitoring and avoiding LA endocardium overlying the esophagus may be useful clinical strategies for avoiding injury to the vagal plexus or its components and are under evaluation. Imaging or identification of periesophageal vagi (which may not correspond to the esophageal lumen) and/or titrating RF energy with respect to the local myocardial thickness and surrounding structures may ultimately be necessary to avoid extracardiac complications. Kuwahara et al. demonstrated that titration of the duration of the ablation energy delivery while monitoring the esophageal temperature could prevent periesophageal nerve injury due to the AF ablation, without decreasing the success rate of maintaining sinus rhythm.18