Chronic Kidney Disease and Atrial Fibrillation: A Contemporary Overview
Nitin Kulkarni MD, Nilusha Gukathasan MBBS, Samantha Sartori, PhD, Usman Baber MD MS.
Mount Sinai School of Medicine, New York, NY 10029.
Chronic kidney disease (CKD) is associated with substantial cardiovascular morbidity, including myocardial infarction, heart failure and stroke. Similar to CKD, atrial fibrillation (AF) is a prevalent arrhythmia that increases risk for both stroke and overall mortality. Recent studies demonstrate that both prevalence and incidence of AF is higher in patient with versus without renal impairment and risk for developing AF increases as renal function worsens. Potential mechanisms for the higher burden of AF in CKD patients include but are not limited to augmented sympathetic tone, activation of the renin-angiotensin-aldosterone system and myocardial remodeling. Similar to the general population, AF confers an increased risk for both stroke and overall mortality in the CKD population. The safety and efficacy of antithrombotic therapy across the spectrum of CKD remains unknown, however, as patients with advanced renal failure are frequently excluded from randomized trials. While treatment with vitamin K antagonists appears to reduce ischemic complications without significant bleeding harm in patients with mild to moderate CKD and AF, the risk benefit ratio of anticoagulation among thosewith advanced renal failure on dialysis requires further investigation. Prospective, randomized trials are war ranted to define the impact of antithrombotic therapy on reducing stroke risk in patients with both AF and CKD.
Correspondence to: Usman Baber MD MS, One Gustave L. Levy Place, Box 1030, New York City, NY, 10029.
Chronic kidney disease (CKD), defined as an estimated glomerular filtration rate (eGFR) below 60 mL/min per 1.73 m2 or kidney damage for 3 or more months, is a common pathologic condition that is strongly associated with increased risk for adverse cardiovascular events.1 Prevalence estimates from the National Health and Nutrition Examination Surveys (NHANES) suggest that approximately 13% of the US adult population has CKD with over 500,000 adults classified as end-stage renal disease.2,3 The public health implications of CKD are significant as these patients sustain higher rates of cardiovascular complications in multiple vascular territories, including lower extremity peripheral arterial disease, myocardial infarction and ischemic stroke.4-6 Despite the well-established epidemiologic link between CKD and cardiovascular risk, causal mechanisms have not been fully elucidated. Atrial fibrillation (AF) is an extremely common cardiac arrhythmia and confers an approximate 4-fold increased risk for ischemic stroke in the general population.7 Both AF and CKD share common risk factors, including hypertension, diabetes mellitus and coronary artery disease.8,9 Not surprisingly, emerging data indicate that AF is more common in patients with versus without CKD and that the presence of renal disease augments thrombotic risk in the setting of AF. In addition, risk benefit calculations of anticoagulation for strokeprevention in AF were largely derived in non-renal cohorts and might not apply to CKD populations as bleeding complications occur more frequently in patients with renal impairment. In the present report, we will review contemporary studies supporting an association between CKD and increased risk for AF and the potential implications these findings have for AF treatment in this high-risk population.
Several population-based studies evaluating the association between AF and CKD have consistently shown a higher prevalence of AF in those with versus without renal disease (Table 1). Moreover, AF prevalence increases in a dose-dependent fashion as renal function worsens.10-12 In a community based Japanese study of 41,417 individuals, the prevalence of AF was 0.9%, 1.2%, and 2.8% among those in the high, mid and low eGFR tertiles, respectively.10 Among US adults enrolled in the prospective Chronic Renal Insufficieny Cohort (CRIC) study (n=3267), the overall prevalence of AF was 18%. While an eGFR < 45 ml/min/1.73m2 vs. ≥ 45 was associated with a higher prevalence of AF (20.4% vs. 16.0%, p < 0.001), this relationship was attenuated after multivariable adjustment (OR 1.12, 95% CI: 0.92 – 1.35; p=0.27).11 The lack of significance between AF and declining renal function in the CRIC study might have resulted from misclassification bias as most cases of AF were self-reported. In contrast, a recent study from the large and nationally representative reasons for Geographic and Racial Differences Study (n=26,917) evaluated the cross-sectional association between electrocardiographic (ECG)-documented AF and kidney dysfunction.12
The prevalence of AF was 1.0% among adults without CKD, and 2.8%, 2.7% and 4.2% among adults with stage 1 to 2, stage 3 and stage 4 to 5 CKD, respectively. The age-, race- and sex-adjusted odds ratios for AF associated with stage 1 to 2, stage 3 and stage 4 to 5 CKD were 2.67 (95% CI, 2.04 – 3.48), 1.68 (95% CI 1.26 – 2.24) and 3.52 (95% CI 1.73 – 7.15), respectively, compared to those without CKD.12
Other measures of kidney dysfunction, such as albuminuria, are also associated with an increased risk for AF.13,14 In a cross-sectional study of 956 adults with coronary artery disease, an albumin to creatinine ratio (ACR) > 15 mg/g was associated with a 4.6-fold increased odds for prevalent AF compared to an ACR < 7 mg/g.13 In a community based study of 7,546 adults in The Netherlands, microalbuminuria was similarly associated with greater prevalence of AF (OR 1.93, 95% CI: 1.10 - 3.37, p<0.05).14
Although most studies evaluating CKD and AF are cross-sectional, similar results have also been observed in several longitudinal reports. In the Japanese Niigata preventive medicine study, for example, 223,877 adults with a mean age of 60.9 years were followed for an average of 5.9 ± 2.4 years.15 AF developed in 2,947 adults (1.3%), with an age-adjusted incidence rate of 2.2 per 1000-person years in adults with an eGFR ≥ 60 compared to 5.2 per 1000-person years in those with an eGFR < 60 ml/min per 1.73m2 (HR 1.38, 95% CI: 1.14-1.66, p < 0.001). Risk for subsequent AF increased with lower baseline eGFR and results were unchanged after excluding patients with hypertension or diabetes at baseline.
Consistent with these findings, the Atherosclerosis Risk in Communities (ARIC) study also found that reduced kidney function and albumuinuria increase risk for developing AF.16 In this community-based study in the US, 10,328 adults without AF at baseline were followed for a median of 10 years, during which 788 patients developed AF. After controlling for traditional risk factors, including age, diabetes mellitus, systolic blood pressure and body mass index, the hazard ratios for AF among those with stage 2, stage 3 and stage 4 CKD were 1.29 (95% CI, 1.05-1.58), 1.70 (95% CI 1.31-2.20) and 3.41 (95% CI 2.18-5.32), respectively, compared to those without CKD. Moreover, the incidence of AF (per 1000 person-years) was 5.8, 14.6 and 26.3 among those with an ACR <30, 30-299 and > 300, respectively (Ptrend < 0.0001).
The presence of both albuminuria and reduced eGFR appear to exert a synergistic impact on AF risk. In the REGARDS study, for example, albuminuria was associated with a 2.5- and 1.5-fold increased odds for AF among those with an eGFR 30-59 and > 60, respectively. Similarly, incident AF increased with lower eGFR in each stratum of ACR in the ARIC study. Among those with an ACR of 30-299 mg/g, for example, the incidence of AF increased from 8% to 30% as eGFR declined from > 90 ml/min/1.73m2 to 15-29 ml/min/1.73m2.
In contrast to the previous reports that mainly included those with non-dialysis dependent CKD, prevalence estimates for AF are generally higher in patients with advanced renal failure requiring dialysis (Table 1). The prevalence of AF ranges from 5% to 27% depending on the duration of dialysis therapy, associated risk factors and patterns of AF (paroxysmal, persistent or permanent).17-21 Concerning trends in the overall prevalence and morbidity associated with AF among United States hemodialysis patients were recently reported by Winkelmayer et al. Using data from the United States Renal Data System (USRDS), they found that overall prevalence of AF has increased from 3.5% in 1992 to 10.7% in 2006. This might be partially attributable to an aging population as the prevalence of HD patients greater than 85 years of age increased from 10% to 22.5% in the same time frame. Moreover, one-year mortality is markedly higher among HD patients with versus without AF (39% vs. 19%).21
Table 1. Studies Reporting Prevalence or Incidence of Atrial Fibrillation (AF) in Chronic Kidney Disease
| Study |
Study type |
Sample size |
Kidney dysfunction |
Prevalence/Incidence |
| REGARDS |
Cross-sectional |
26,917 |
eGFR > 60 ml/min/1.73m2 + albuminuria eGFR 30-59 ml/min/1.73m2 eGFR < 30 ml/min/1.73m2 |
2.8% 2.7% 4.2% |
| KAMS |
Cross-sectional |
41,417 |
eGFR > 75.5 ml/min/1.73m2 eGFR 62.6 – 75.5 ml/min/1.73m2 eGFR < 62.6 ml/min/1.73m2 |
0.9% 1.2% 2.8% |
| CRIC |
Cross-sectional |
3267 |
eGFR < 60 ml/min/1.73m2 |
18% |
| Niigata |
Prospective |
235,818 |
eGFR 30-59 ml/min/1.73m2 eGFR < 30 ml/min/1.73m2 |
5.1 6.6 |
| ARIC |
Prospective |
10,328 |
eGFR 30-59 ml/min/1.73m2 eGFR 15-29 ml/min/1.73m2 albumin/creatinine ratio 30-299 mg/g albumin/creatinine ratio ≥ 300 mg/g |
9 36 15 26 |
| DOPPS |
Cross-sectional |
17,513 |
Hemodialysis |
12.5% |
| Vazquez et al. |
Cross-sectional |
190 |
Hemodialysis |
13.6% |
| USRDS** |
Cross-sectional |
223,477 |
Hemodialysis |
10.7% |
| Genovesi et al. |
Cross-sectional |
488 |
Hemodialysis |
27% |
| USRDS** |
Cross-sectional |
25,825 |
Peritoneal dialysis |
7% |
*Cross-sectional studies reported prevalence estimates as percentage while prospective studies reported incidence expressed per 1000 person-years. **Data from 2006. **Data from 2005. REGARDS – Reasons for Geographic and Racial Differences in Stroke; CRIC – Chronic Renal Insufficiency Cohort; ARIC – Atherosclerosis Risk in Communities; KAMS – Kurashiki City Annual Medical Survey ; DOPPS – Dialysis Outcomes and Practice Patterns Study; USRDS – United States Renal Data System; eGFR – estimated glomerular filtration rate
Mechanisms for Increased AF in CKD
Shared risk factors between CKD and AF suggest a common pathophysiologic substrate between these two conditions. In fact, several studies have found similarities in clinical correlates of AF among those with and without CKD. In the REGARDS study, for example, advanced age, left ventricular hypertrophy and heart failure were positively associated with prevalent AF across all levels of renal function.12 While the association between CKD and AF remains independent after adjusting for traditional risk factors, novel or unaccounted for mechanisms may also contribute to the greater burden of AF in those with renal dysfunction. These include but are not limited to alterations in the renin-angiotensin-aldosterone system (RAAS), increased inflammation and sympathetic tone, myocardial fibrosis and abnormalities of myocardial structure and function (Table 2).
Activation of the RAAS in the setting of CKD leads to progressive renal deterioration.22 The renoprotective effects of angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARB) further demonstrate the central role of RAAS dysregulation in the pathogenesis of CKD. Angiotensin II (AngII) is a profibrotic molecule that is associated with cardiomyocyte apoptosis and reactive interstitial fibrosis.23 Through direct effects and downstream activation of transforming growth factor-β1, AngII might contribute to structural remodeling such as atrial fibrosis that enhances risk for AF.24,25 Inflammatory pathways in CKD patients might also lead to greater AF in these patients. Multiple studies, for example, have shown higher levels of several inflammatory biomarkers in those with versus without CKD and inflammation predisposes to AF. An important role for inflammation in AF prevalence among those with renal dysfunction is supported by results of the REGARDS study as the association between elevated C-reactive protein (CRP) and prevalent AF was significant only among those with stage 3-5 CKD (OR 1.93, 95% CI: 1.20 – 3.09, p <0.001). Similar findings were reported from the Prevention of Renal and Vascular Endstage Disease (PREVEND) study. While AF was twice as common in those with versus without microalbuminuria, the combination of both elevated CRP and microalbuminuria resulted in an approximate 4-fold increased odds for prevalent AF (OR 3.80, 95% CI: 1.89-7.63, p<0.001). Modulation of sympathetic tone in CKD26,27 might also contribute to increased risk for AF. Sympathetic hyperactivity in CKD was initially suggested by clinical studies demonstrating increased concentrations of plasma catecholamines and enhanced sensitivity to norepinephrine.28,29 The marked reduction in blood pressure following renal denervation using a catheter-based approach observed in a recent randomized trial highlights the critical role of renal sympathetic activity in hypertension, a common risk factor for both renal dysfunction and AF.30 Whether or not these benefits might result in lower risk for both CKD and AF warrants further investigation. Myocardial functional and structural abnormalities common in CKD might also act as mediators for AF in these patients. Diastolic dysfunction and left atrial dilatation, for example, are more common in patients with versus without CKD and are associated with increased risk for AF.31-33 Several studies have also found significant associations between left atrial enlargement and AF in those with both mild to moderate and dialysis-dependent CKD.17,34 In addition, myocardial fibrosis has been demonstrated to occur early in CKD patients35 and might increase AF risk by enhancing atrial re-entry.36 It is important to note that although the association between CKD and AF is consistent with certain tenets of causality, including a dose-response effect, biologic plausibility and independence, causality cannot be inferred based on existing data. The potential for residual confounding on the association between CKD and AF is always possible as renal dysfunction might reflect prolonged exposure to or poor control of traditional risk factors. Pending further data, existing hypotheses regarding AF burden in CKD remain putative and require confirmation.
Table 2. Potential mechanisms of Atrial Fibrillation in Chronic Kidney Disease
| Advanced age |
| Heart failure |
| Left ventricular hypertrophy |
| Left atrial enlargement |
| Inflammation |
| Sympathetic activation |
| Myocardial fibrosis |
| Activation of the renin-angiotensin-aldosterone system |
*Selected abnormalities
Thromboembolic Risk in CKD and AF
While the most feared complication of AF is disabling ischemic stroke,7 risk is not uniform in all patients. As a result, validated and widely used clinical algorithms, such as the CHADS2 score, facilitate decisions regarding use of antithrombotic therapy in AF patients. Although CKD is not included in the CHADS2 algorithm, results of several studies have consistently shown that renal dysfunction confers additional and considerable risk for thromboembolic complications in AF patients.
Pooling data from the 3 trials of the Stroke Prevention in Atrial Fibrillation (SPAF), Hart et al. evaluated the association between kidney dysfunction and embolic events in participants not receiving adjusted-dose warfarin. At 2 years the primary outcome, a composite of ischemic stroke and systemic embolism, occurred more frequently among those with versus without CKD (9.2% vs. 4.1%, p=0.004). CKD was associated with a 40% increased risk for primary events after adjusting for the CHADS2 score (HR 1.4, 95% CI: 0.8 – 2.3; p=0.2). In a separate report of over 10,000 patients with non-valvular AF, Go et al. examined the associations between eGFR and proteinuria on thromboembolic events. The crude thromboembolism rates per 100 person-years were 1.63, 2.76 and 4.22 among those with an eGFR > 60, 45-59 and < 45, respectively (Ptrend <0.001). Compared to patients with an eGFR ≥ 60 ml/min/1.73m2, the adjusted HRs for thromboembolism were 1.16 (0.95 – 1.40) and 1.39 (1.13 – 1.71) among those with an eGFR 45-59 and < 45, respectively.37 Similarly, the presence of proteinuria was associated with a 54% increased risk for thromboembolism compared to no proteinuria. In a smaller single-center study of 387 AF patients with AF Nakagawa et al. found that CKD was associated with increased risk for ischemic stroke irrespective of baseline CHADS2 score.38 In aggregate, these data suggest that renal dysfunction augments thromboembolism in the setting of AF, which is consistent with CKD-associated changes in coagulation that increase risk for both venous and arterial thrombosis.39-42 Conversely, separate studies have shown that the presence of AF magnifies thromboembolic risk in CKD patients.
Studies in dialysis patients, for example, have generally shown that AF is associated with increased risk for both mortality and cerebrovascular events. In the DOPPS(Dialysis Outcomes and Practice Patterns Study) Wizemann et al. found that AF increased risk for subsequent stroke or cerebrovascular events by approximately 30% (HR 1.28, 95% CI: 1.01 – 1.62; p=0.048).43 Data from the USRDS and DOPPS have also shown that both all-cause and cardiac mortality is significantly higher in dialysis patients with versus without AF.44 Similar findings were reported by Vazquez et al. in a separate single-center study of 190 HD patients followed for approximately 3 years. Mortality was significantly higher among those with versus without AF (80.8% vs. 28.7%, p=<0.001) and the association remained significant after multivariable adjustment (OR 2.1, 95% CI: 1.2-3.9; p=0.009).45 AF was also associated with an increased risk of thromboembolic events, including stroke (OR 4.6, 95% CI 2.4-8.6). Another longitudinal study of 477 patients on HD found a higher one-year mortality rate among patients with versus without AF (31.3% vs. 10.4%).17
Therapeutic Considerations
The most widely used and extensively studied treatments for primary and secondary stroke prevention in AF remain vitamin K antagonists (VKA). Compared to control, adjusted dose warfarin results in a large and significant 60% reduction in ischemic stroke based on a recent meta-analysis of 29 trials involving over 28,000 patients with AF.46 Extrapolating these findings to renal cohorts is problematic, however, as patients with advanced renal failure were frequently excluded from randomized trials evaluating antithrombotic therapy in AF and bleeding propensity increases with worsening renal function. Uremic platelet dysfunction, for example, is a well-described phenomenon of dialysis-dependent renal failure47 and CKD is a consistent correlate of higher bleeding risk in multiple bleeding algorithms.48 Although limited, existing data suggest that the net clinical benefit of lower ischemic complications without greater bleeding harm following anticoagulation in CKD patients with AF may be greater in those with mild to moderate versus advanced dialysis-dependent renal failure (Figure 1). In a post-hoc analysis of the 3 trials of SPAF, Hart et al. compared ischemic and bleeding outcomes among patients with stage 3 CKD (n=537) randomized to adjusted-dose warfarin versus fixed, low-dose warfarin and aspirin. Renal impairment was modest in the CKD group, with a mean eGFR of 50 ml/min/1.73 m2. Adjusted-dose warfarin significantly reduced the relative risk of ischemic stroke or systemic embolization by 76% (p<0.001) without a concomitant increase in major bleeding (3.6% vs. 2.5%, p=0.65).49 These benefits were consistent to those observed in patients with an eGFR ≥ 60 ml/min/1.73m2.Analogous results were demonstrated in a single-center retrospective analysis comparing outcomes in CKD patients (n=566) with AF treated with or without warfarin.50 CKD severity was greater in this study compared to the report of Hart et al., with 23% of patients on HD. Over a follow-up period of approximately 2 years, warfarin use was associated with a significant reduction in new thromboembolic stroke (9% vs. 26%, p <0.001). The incidence of major bleeding was 11.8%, substantially higher than the 2.1% rate observed in the less severe CKD patients enrolled in the 3 trials of SPAF.50 Warfarin use was associated with a higher, albeit not statistically significant, incidence of major bleeding compared to no warfarin (14% vs. 9%, p=NS).
Figure 1. Relative Risk for Stroke or Systemic Embolism and Major Bleeding Associated with Anticoagulation Use in Chronic Kidney Disease (CKD)
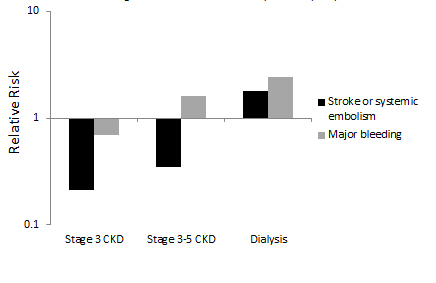
Results for stage 3 CKD abstracted from Hart et al.49; results for stage 3-5 CKD abstracted from Lai et al.50; results for dialysis abstracted from Chan et al.52 for stroke or systemic embolism and Vazquez at al.53 for major bleeding
Quantifying the overall impact of anticoagulation in dialysis-dependent CKD is complicated by confounding influences of dialysis mode, membranes and timing on multiple thrombotic parameters. Although several studies have shown an expected pattern of greater bleeding risk with anticoagulation in dialysis patients,51 reductions in ischemic complications or stroke are inconsistent. In fact, several studies suggest greater harm following anticoagulation in dialysis patients with AF. In a retrospective cohort study of HD patients with prior AF (n=1671) from the USRDS, for example, Chan et al. evaluated the impact of warfarin, clopidogrel and aspirin on ischemic and hemorrhagic complications over 1.6 years. Compared to no antithrombotic or antiplatelet therapy, warfarin use was associated with a significantly increased risk of both hemorrhagic (HR 2.22; 95% CI 1.01 5 to 4.91) and ischemic stroke (HR 1.81; 95% CI 1.12 to 2.92).52 Stratified analyses by INR monitoring suggested that the excess risk of warfarin was greatest in those patients without any INR surveillance. Similar results were reported by Wizemann et al. in an analysis using data from the international Dialysis Outcomes and Practice Patterns Study (DOPPS). In this study warfarin use was associated with an increased risk for stroke, which was most apparent in those over 75 years of age.43 A potential explanation for these findings is the well-documented increased risk of bleeding with use of anticoagulants in the dialysis population.53 In addition, anticoagulation likely magnifies the inherent risk for both ischemic and hemorrhagic stroke associated with CKD.6 In support of this hypothesis Chan et al. found that 29% of dialysis patients with AF stopped using anticoagulants after a stroke, suggesting that these events were likely hemorrhagic. Data demonstrating increased harm with anticoagulation use in HD patients with AF must be interpreted with caution, however, as these studies were limited by their observational design, potential for confounding by indication and misclassification bias in stroke ascertainment.
In contrast to these findings, separate studies suggest no significant differences in stroke rates in HD patients treated with or without warfarin,while another report indicates a survival advantage among hospitalized dialysis patients with AF who were on warfarin.54,55 Not surprisingly, the uncertainty surrounding both the efficacy and safety of anticoagulation in dialysis-dependent CKD patients has led to substantial variability in clinical practice patterns for AF in these patients. Results of the DOPPS study, for example, demonstrate that warfarin use in dialysis patients with AF ranges from a low of 2% in Germany to 37% in Canada.43 The conflicting data regarding the impact of anticoagulation in dialysis patients combined with the substantial morbidity associated with AF in these populations warrants definitive, prospective trials to further clarify the risk benefit ratio of anticoagulation use in dialysis-dependent renal failure.
Emerging Treatments: Catheter Ablation in Atrial Fibrillation and CKD
Catheter ablation therapy of AF is an alternative treatment modality for the management of AF, and is currently indicated for patients with symptomatic AF who cannot tolerate or are refractory to class I or III antiarrhythmic therapy.56 Success rate of ablative therapy has improved over the past few years, with a recent multicenter study showing an overall 70% success rate resulting in patients not requiring anti-arrhythmic drugs, with the greatest success rates in patients with paroxysmal AF.57 However, patients with large left atrial size, obesity, sleep apnea, heart failure, or a longer duration of time spent in AF tend to have higher recurrence rates after ablation.56
The role of catheter ablation for AF in CKD patients has recently gained more attention as a potential treatment modality in this population. While certain studies suggest that this therapy might be efficacious in the CKD population, kidney dysfunction also appears to be a correlate of recurrence after AF ablation. A study published by Takahashi et al., for example, demonstrated that after AF ablation in 386 patients with AF and CKD, 72% of patients were free from arrhythmia after 1 year of follow-up.58 Conversely, in a study of 232 patients with paroxysmal AF the recurrence rates following ablation after a mean follow-up of 25 months was 6.9%, 14.5% and 38.9% in patients with an eGFR ≥ 90 ml/min/1.72m2, stage II CKD, and stage III CKD or worse, respectively.59 Other studies have reported similar results, suggesting that CKD is an independent predictor of recurrence after AF ablation.60,61 These studies allude to the complex interplay between cardiac and renal disease, and given that CKD patients have higher rates of complications after ablative therapy62, further prospective studies are needed to fully evaluate the risks and benefits of AF ablation in this patient population.
CKD and AF are common pathologic conditions that share several predisposing risk factors. The prevalence of AF is higher in patients with versus without CKD and prevalent AF increases as renal function worsens. Similar to the general population, the presence of AF in CKD is associated with increased mortality and thromboembolic risk. Although limited, existing data suggest that the net clinical benefit of anticoagulation in CKD patients with AF is attenuated in those with the most severe levels of renal impairment. Prospective, randomized trials of antithrombotic therapy in CKD patients with AF are required to guide clinical decisions and optimize treatments in this high-risk population.