Computed Tomography-Derived Three-Dimensional Printed Models versus Two-Dimensional Transesophageal Echocardiography for Left Atrial Appendage Occlusion Device Planning: A Systematic Review and Meta-Analysis
Garly Saint Croix1, Syed Imran Zaidi1, Viky S. Loescher3, Christos G. Mihos12
1Columbia University Division of Cardiology, Mount Sinai Heart Institute, Miami Beach, FL.2Echocardiography Laboratory, Columbia University Division of Cardiology, Mount Sinai Heart Institute, Miami Beach, FL.3Department of Radiology, Cardiothoracic & Vascular Imaging, Mount Sinai Medical Center, Miami Beach, FL.
This systematic review and meta-analysis compared computed tomography (CT)-derived three-dimensional (3D) modeling versus two-dimensional transesophageal echocardiography (TEE) for left atrial appendage occluder (LAAO) device planning.
LAAO device planning is commonly performed with TEE. However, procedures often require multiple devices and deployments due to inaccurate sizing from TEE. The use of CT three-dimensional (3D) models for LAAO device planning may improve accuracy.
Four clinical studies that reported procedural and clinical outcomes for CT-derived 3D modeling versus TEE for LAAO device planning were identified. End points were accurate device sizing, procedure failure, number of devices used per procedure, fluoroscopy time, and post-procedure leak. Risk ratio (RR) and mean difference (MD) with a 95% confidence interval (CI) were calculated by the Mantel-Haenszel and inverse variance methods.
A total of 166 participants were included. When compared with conventional imaging, the use of 3D printed models was associated with less fluoroscopy time (MD -6.98 minutes, 95% CI -12.68 to -1.28, p=0.02) and lower risk of occluder device peri-prosthetic leak (RR 0.23, 95% CI 0.07-0.73, p=0.01) for LAAO. There were signals towards lower number of devices per procedure (MD -0.56 devices, 95% CI -1.16-0.05, p=0.07) and less total procedure time (MD -13.50 minutes, 95% CI -28.14-1.14, p=0.07) with printed modeling for LAAO. There was no difference between modalities in rates of procedure failure.
CT-derived 3D printed models for LAAO device planning may offer the advantages of lower LAAO device peri-prosthetic leak and less fluoroscopy time when compared with conventional TEE guidance.
Key Words : , , ,.
Christos G. Mihos, D.O.
Director, Echocardiography Laboratory
Columbia University Division of Cardiology
Mount Sinai Heart Institute
4300 Alton Road, DHMT Suite 2070
Miami Beach, FL 33140
Percutaneous left atrial appendage (LAA) device closure is a novel treatment for embolic stroke prevention in patients with non-valvular atrial fibrillation (AF). Studies have demonstrated that LAA closure with the Watchman device (WD) (Boston Scientific, Natick, MA) is non-inferior to long-term anticoagulation with warfarin for stroke prevention 1-7. While anticoagulation remains the standard of care, device closure has now become an option for high-risk patients with contraindications to long-term anticoagulation 8.
LAA occlusion (LAAO) requires precise imaging to determine device sizing due to variability in size and shape of the LAA leading to differences in the orifice size and landing zone 9. Transesophageal echocardiography (TEE) is often used to determine appropriate sizing and target site for device deployment 10. However, LAAO procedures often require multiple devices and deployments for adequate occlusion predisposing to increased complications and cost. Recent studies have evaluated the use of three-dimensional computed tomography (3D-CT) and 3D-printing for pre-procedure planning to increase device implantation precision. However, data comparing CT-derived 3D models versus TEE for LAAO device selection and procedural efficiency is limited. This systematic review and meta-analysis sought to compare the impact of CT-derived 3D modeling versus TEE for LAAO device planning.
The study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement extension for network meta-analysis. The PRISMA flow diagram was used to depict the four phases of the review including identification, screening, eligibility and inclusion. The literature review identified randomized and nonrandomized clinical studies that reported comparisons between CT-derived 3D modeling versus TEE for LAAO device planning. Searches were limited to peer-reviewed primary research articles published in English, French and Spanish through July 2020. This research involved human subjects and described the impact of CT-derived 3D modeling versus TEE on device planning for patients who underwent LAAO. We developed the search strategy according to available guidance from the Cochrane Collaboration.
The search strategy in MEDLINE explored Medical Subject Heading (MeSH) terms related to CT-derived 3D modeling and TEE for LAAO device planning. The following Boolean strategy was applied to search PubMed/MEDLINE: (((("printing, three-dimensional"[MeSH Terms] OR ("printing"[All Fields] AND "three dimensional"[All Fields])) OR "three-dimensional printing"[All Fields]) OR ("3d"[All Fields] AND "printing"[All Fields])) OR "3d printing"[All Fields]) AND (("atrial appendage"[MeSH Terms] OR ("atrial"[All Fields] AND "appendage"[All Fields])) OR "atrial appendage"[All Fields]) AND (((("transesophageal echocardiography"[All Fields] OR "echocardiography, transesophageal"[MeSH Terms]) OR ("echocardiography"[All Fields] AND "transesophageal"[All Fields])) OR "transesophageal echocardiography"[All Fields]) OR ("transesophageal"[All Fields] AND "echocardiography"[All Fields])). The articles found to be relevant during the search were stored in EndNote. Selected articles underwent full evaluation to assess their potential inclusion in the systematic review. Finally, the reference lists of all included studies were also cross-checked for publications not captured by the Boolean search strategy.
Articles were selected for inclusion based on the use of CT-derived 3D modeling compared to conventional TEE imaging for LAAO procedural and clinical outcomes. We excluded case reports and feasibility studies that did not compare outcomes with a control group. Three authors (S.I.Z., G.S.C., C.G.M.) independently read each full text article to assess their potential inclusion in the systematic review and meta-analysis. Risk of bias in the studies was assessed at the individual level of each study.
Data were analyzed using Review Manager software 5.4. We used a random effects model to assess the combined risk and mean difference estimates. Study heterogeneity was evaluated using the I2 statistic. Analysis to determine sensitivity and publication bias was detected by funnel plots. A P-value <0.05 was considered statistically significant.
A total of 24 abstracts were identified, and all were reviewed in detail. Four studies were ultimately included in the systematic review and meta-analysis; the studies by Li et al., and Eng et al., were prospective randomized studies, while the remaining 2 studies were retrospective 11-14. [Figure 1] describes the flow-chart of included studies.
Figure 1. Flow-chart of the included studies.
A total of 24 abstracts were identified, and all were reviewed in detail. Shown above is the step-wise progression of the screening and selection process. Four studies were ultimately analyzed, which included two randomized trials and two retrospective analyses.
Baseline Characteristics of the Studies
[Table 1] shows the baseline characteristics of the included studies. All studies were published between 2017 and 2019. The 4 studies included 166 patients with 68 (41%) patients undergoing LAAO with CT-derived 3D modeling used for procedural planning. CT-derived 3D modeling was performed with ECG-gated 256-slice protocols in two studies;13,14 no details were available in the remaining two.11,12 The median age of the participants was 71.0 years (IQR, 69.7-75.8), and 48% were female. Clinical risk factors and co-morbidities included 90% hypertension, 29.5% diabetes, 37% coronary artery disease 30% congestive heart failure, and 39% chronic kidney disease. The median CHADS2-VASC score was 4.6 (IQR, 4.2-4.9), HAS-BLED score was 3.8 (IQR, 3.4-4.3), and body mass index 27.6 kg/m2 (IQR, 25.0-28.9). Single centers were sites for all of the included studies and the United States, France, and China were the countries represented.
Table 1. Patient demographics and characteristics in the studies meeting inclusion criteria for the systematic review and meta-analysis.
| Study Author, Year |
Location |
N |
3D Modeling Group |
Patients |
Age, Years |
Female |
CHF |
Diabetes |
HTN |
HLD |
| Obasare et al., 2017 |
USA |
2 |
3D Modeling* |
14 |
71 ± 9 |
36% |
29% |
36% |
100% |
57% |
| 4 |
No 3D Modeling |
10 |
69 ± 13 |
50% |
30% |
30% |
80% |
70% |
| Eng et al., 2018 |
USA |
2 |
3D Modeling |
12 |
79.9 ± 6.7 |
33% |
42% |
42% |
100% |
NR |
| 4 |
No 3D Modeling |
12 |
73.5 ± 7.6 |
50% |
42% |
25% |
100% |
NR |
| Ciobotaru et al., 2018 |
France |
7 |
3D Modeling |
21 |
78 ± 7 |
38% |
29% |
NR |
NR |
NR |
| 6 |
No 3D Modeling |
55 |
|
|
|
|
|
|
| Li et al., 2017 |
China |
4 |
3D Modeling |
21 |
68.1 ± 6.9 |
52% |
NR |
29% |
62% |
NR |
| 2 |
No 3D Modeling |
21 |
70.4 ± 8.6 |
48% |
NR |
24% |
57% |
NR |
BMI=body mass index; CAD=coronary artery disease; CHF=congestive heart failure; CKD=chronic kidney disease; CVA=cerebrovascular accident; HLD=hyperlipidemia; HTN=hypertension; N=number; NR=not reported.
3D Modeling versus Conventional Imaging for LAAO
Ciobotaru et al., Eng et al., Li et al., and Obasare et al. reported various procedural outcomes in patients undergoing LAAO with CT-derived 3D modeling compared with TEE 11-14. Ciobotaru et al. found that CT-derived 3D modeling for LAAO double disc device planning decreased the number of prostheses, incidence of leaks, fluoroscopy time, and fluoroscopy dose when compared to conventional imaging 11. Eng et al. found reduced number of utilized prostheses, guide catheters, and procedure time when CT-derived 3D modeling was compared with TEE for LAAO device planning 12. Li et al. found that CT-derived 3D modeling for LAAO device planning significantly reduced radiation exposure compared with TEE 13. Finally, Obasare et al. found reduced procedure time, anesthesia time, fluoroscopy time, and incidence of peri-device leak when CT-derived 3D modeling was compared with TEE for LAAO device planning 14.
Meta-analysis of the included studies revealed less fluoroscopy time (MD -6.98 minutes, 95% CI -12.68 to -1.28, p=0.02) with CT-derived 3D modeling for LAAO device planning as shown in the forest plot of 2 studies in [Figure 2]. Additionally, data from all 4 identified studies revealed the use of CT-derived 3D modeling decreased the incidence of peri-prosthetic leak (RR 0.19, 95% CI 0.07-0.51, p<0.01) when compared with TEE for LAAO device planning as shown in the forest plot in [Figure 3]. There were signals of a lower number of devices per procedure (MD -0.56 devices, 95% CI -1.16-0.05, p=0.07) and less total procedure time (MD -13.50 minutes, 95% CI -28.14-1.14, p=0.07) when CT-derived 3D modeling was used for LAAO device planning as shown in [Figure 4] and [Figure 5], respectively. There was no difference between modalities in rates of procedure failure as shown in [Figure 6]. None or low heterogeneity was detected for the outcomes of total fluoroscopy (I2=0%), peri-prosthetic device leak (I2=0%), and procedure failure (I2=0%). Moderate heterogeneity was detected for the outcomes of number of devices per procedure (I2=75%) and total procedure time (I2=55%).
Figure 2. Forest plot of fluoroscopy time for LAAO with and without 3D modeling.
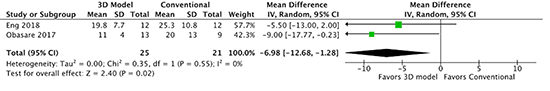
Comparison of left atrial appendage occluder device planning using 3D computed tomography printed modeling versus conventional 2D transesophageal echocardiography on total procedural fluoroscopy time.
Figure 3. Forest plot of post-procedure peri-prosthetic leak for LAAO with and without 3D modeling.
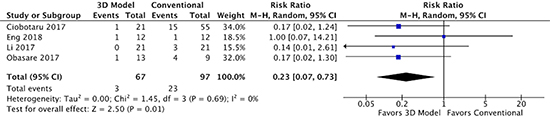
Comparison of left atrial appendage occluder device planning using 3D computed tomography printed modeling versus conventional 2D transesophageal echocardiography on the presence of post-procedure peri-prosthetic leak.
Figure 4. Forest plot of number of devices per procedure for LAAO with and without 3D modeling.
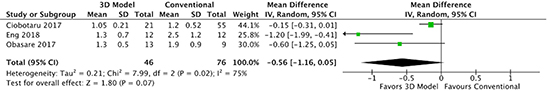
Comparison of left atrial appendage occluder device planning using 3D computed tomography printed modeling versus conventional 2D transesophageal echocardiography on the number of devices utilized per procedure.
Figure 5. Forest plot of procedure time for LAAO with and without 3D modeling.

Comparison of left atrial appendage occluder device planning using 3D computed tomography printed modeling versus conventional 2D transesophageal echocardiography on total procedure time.
Figure 6. Forest plot of procedure failure for LAAO with and without 3D modeling.
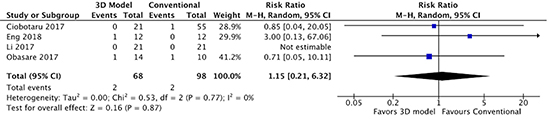
Comparison of left atrial appendage occluder device planning using 3D computed tomography printed modeling versus conventional 2D transesophageal echocardiography on procedure failure.
The present meta-analysis reported the pooled outcomes from 4 studies including 166 patients who underwent LAAO utilizing the Watchman occlude device. When compared with TEE, the use of 3D CT-derived printed modeling for device planning was found to confer the following: 1) less total required fluoroscopy time; 2) lower risk of occlude device peri-prosthetic leak; 3) numerically lower number of devices utilized and total procedural time; and, 4) a similar rate of successful device implantation.
Utilization of TEE is currently the conventional method for sizing of the LAA prior to occluder device implantation. However, the varied anatomy of the appendage, position of the ostium relative to the TEE probe, and individual cardiac positioning within the thoracic cavity often results in suboptimal LAA visualization. make it difficult to capture imaging of the LAA with TEE. Hemodynamic loading conditions and appendage contractile cycles may also result in orifice size underestimation 15-17. Indeed, registry data shows an average of 1.4 devices used per case for LAAO procedures planned with TEE 10. Finally, there are also few consistent landmarks for determining the best landing zone for the occluder device 14.
CT scanning provides 3D reconstruction of the entire LAA with detailed depiction of LAA walls, lobes, and apex [Figure 7] 9. Previous analyses have concluded that CT imaging alone improves LAAO device size prediction and procedural outcomes 18,19. The studies included in the present meta-analysis took the additional step of creating 3D-printed models to aid in procedural planning. The 3D model can help operators visually assess device fit and determine the optimal landing zone. This translates to more time-efficient procedures with less devices used for successful implantation. The process of creating 3D models is relatively straightforward and quick. The included studies report needing approximately 6 to 10 hours to obtain the 3D printed model following the CT scan, and a cost of about $10 per patient 14. This suggests that 3D modeling is likely cost effective given the reduction in procedure time and device deployments.
Figure 7. Computed tomography-derived three-dimensional modeling.
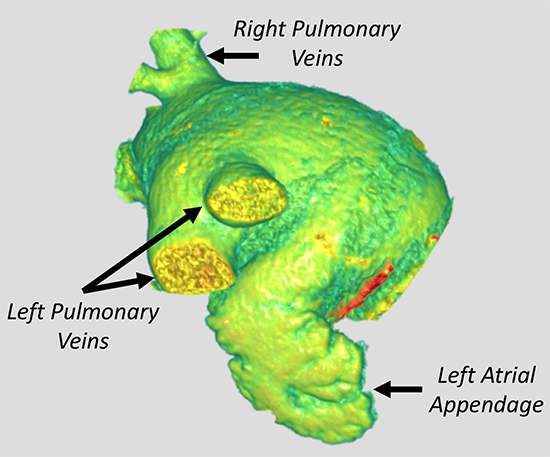
Depicted is a ‘chicken wing’ left atrial appendage and the left atrium. The left circumflex coronary artery is also shown in red in the left atrioventricular sulcus, which is an important landmark of in pre-procedural measurements and device sizing.
Several early feasibility studies evaluated the use of CT-derived 3D printing for LAAO device planning. Hell et al. found that 3D printing improved device sizing in a cohort of 22 patients undergoing LAAO. Predicted device size with the 3D model matched the final implanted device in 21/22 patients (95%) compared with 10/22 (45%) of patients with device sizing planned by TEE (20). In a cohort of 29 patients, Goiten et al., found good correlation between device sizing predicted by 3D modeling and the procedurally implanted device 21. Hachulla et al. published a case series of 15 patients that found 3D printing to improve predicted device sizing 22. The present findings support and expand upon this earlier data by analyzing studies with procedural outcome comparisons made between 3D print modeling and conventional imaging groups.
Fan et al. recently published a study using 3D-TEE data to create 3D printed models for LAAO device planning in 104 patients undergoing LAAO. Improved safety and efficacy was demonstrated for device selection with TEE 3D modeling-based sizing in patients undergoing LAAO 23. The use of TEE-derived 3D models may be preferred to CT in patients whom contrast and radiation exposure is a concern, particularly in the setting of chronic kidney disease or contrast allergy. However, issues with complete visualization of the LAA by 2D TEE translate to 3D-TEE, with image dropout artifacts as the most common impediment to widespread application 14. Nevertheless, in appropriate candidates with good imaging windows, 3D-TEE may be a viable alternative to 2D TEE or contrast-required 3D CT-derived printed modeling.
There are limitations to the present meta-analysis that should be considered when interpreting the data. Firstly, the sample size of the individual studies and pooled cohort was small, which limits power for statistical analyses. Furthermore, the outcome measures were not uniformly reported across all of the studies; this may be interpreted as a form of attrition or information bias. Secondly, between-center variability in procedural volume, assessment protocol for procedural results and outcomes, and availability of advanced imaging limits the generalizability of the results. These variables are closely related to and impact the learning curves for the structural interventionalist and imaging cardiologist, which introduces uncontrollable confounding. Thirdly, there are important knowledge gaps within the included studies that may bias the analyses. In the study by Obasare et al., there was a greater number of patients with chronic kidney disease in the non-3D (TEE) modeling group which may have led to avoidance of contrast utilization and increased procedure time 14. In the study by Eng et al., the time between CT scanning and modeling and LAAO was several weeks to months. The lack of standardization allows for the impact of possible anatomic changes, such as chamber size and function, on procedural planning and successful device implantation 12. Finally, two of the studies included in the pooled analysis were retrospective in nature, which confers an inherent selection bias. However, these study designs are viewed as complementary. Observational investigations allow for external validation of randomized controlled data, provide insight into physician practice patterns, and represent an accurate patient population sample.
In conclusion, CT-derived 3D printed models for LAAO device planning may attenuate the risk of post-implantation device peri-prosthetic leak and decrease the procedure fluoroscopy time when compared with conventional TEE guidance. The current data is hypothesis-generating for larger confirmatory prospective and multi-center registry studies on the optimal method for LAAO device planning, which should also incorporate the use and further study of 3D TEE LAA modeling.