High Power Ultra Short Duration Ablation with HD Grid Improves Freedom from Atrial Fibrillation and Redo Procedures Compared to Circular Mapping Catheter
John D. Day1, Brian Crandall2, Sriharsha Kanuri2, Michael Cutler2, Jeffrey Osborn2, Jared Miller2, Charles Mallender3, Dhanunjaya Lakkireddy4
1Electrophysiology, Intermountain Medical Center Heart Institute, Murray, Utah.2Arrhythmia Research Fellow, Kansas City Heart Rhythm Institute (KCHRI), Overland Park, KS, USA.3Internal Medicine, Intermountain Medical Center Heart Institute, Murray, Utah.4Executive Medical Director, Kansas City Heart Rthym Institute, Overland Park, KS, US.
High power ultra-short duration (HPUSD) ablation has been advocated to prevent esophageal injuries during atrial fibrillation (AF) ablation procedures. Prior research using the standard circular mapping catheter (CMC) has shown that ultra-short ablations may compromise lesion durability resulting in an increased need for redo procedures. The purpose of this study was to determine if HD mapping of concealed pulmonary vein (PV) connections could improve freedom from atrial fibrillation and redo procedures compared to CMC guided AF ablation.
A total of 472 consecutive first time AF ablation procedure patients with at least one year of follow up were included with an average follow-up of 18 months. HPUSD AF ablation consisted of 50 W for 2-3 seconds on the posterior wall and 5-15 seconds on the anterior wall of the left atrium. Acute pulmonary vein isolation (PVI) was defined as no concealed 1) PV signals, 2) activation into PVs, or 3) voltage into PVs with no intra-procedural waiting period utilizing the HD Grid catheter versus entrance/exit block with a 30-minute wait with the circular mapping catheter. Freedom from atrial fibrillation and all atrial arrhythmias following a 90-day blanking period were assessed.
Acute pulmonary vein isolation was achieved in all 472 patients. HPUSD ablation using the HD Grid was associated with shorter procedure (70.2 vs 104.3 minutes, p<0.001) and fluoroscopy times (4.2 vs 15.0 minutes, p<0.001) when compared to CMC. The recurrence of any atrial arrhythmias at 1 year was 13% with HD Grid and 25% with CMC (p<0.001) with the need for redo procedures of 6% for HD Grid and 20% for CMC (p<0.001). No esophageal ulcerations/perforations were seen. No deaths, strokes, or TIAs were observed in either group.
HPUSD AF Ablation, as guided by HD Grid mapping, may prevent esophageal injuries while at the same time improve freedom from any atrial arrhythmias and the need for redo procedures. Procedure and fluoroscopy times were also significantly decreased when compared to traditional CMC mapping.
Key Words : HD Grid Mapping, CMC mapping, AF ablation.
Correspondence to: Dr John D. Day
Director, Intermountain Heart Institute Heart Rhythm Specialists
Intermountain Medical Center
5169 Cottonwood St,
Murray, UT 84107
Two key limitations to atrial fibrillation (AF) ablation procedures are the need for redo procedures to achieve durable pulmonary vein isolation and the potentially fatal risk of collateral thermal damage to the esophagus. Indeed, the 2017 AF ablation consensus statement reported an AF catheter ablation procedural redo rate of 15 to 50% largely to correct this problem or pulmonary vein electrical reconnection1.
To help address the challenge of pulmonary vein reconnection, newer technologies such as contact force to deliver a more effective ablation lesion and more recently high-density mapping techniques have been proposed to eliminate any potential pulmonary vein isolation gaps or areas of dormant conduction1. While newer mapping techniques offer the promise of being able to see previously “concealed” pulmonary vein electrograms or residual conduction or voltage into the pulmonary veins, it is not known whether eliminating these “gaps” or areas of dormant conduction will improve procedural success or decrease the need for redo procedures.
With regards to decreasing the risk of the potentially fatal complication of collateral thermal injury to the esophagus, the high power short duration approach to radiofrequency delivery has been proposed by our group as well as others2. While there is no agreement on how high is high power or how short is short duration, our group has proposed a high power ultra-short duration (HPUSD) approach to radiofrequency catheter ablation of AF3.
The HPUSD approach offers the potential of a much safer and quicker ablation procedure; however, our reported tradeoff is a somewhat increased need for redo procedures to achieve durable pulmonary vein isolation. The purpose therefore of this study was to evaluate whether newer HD mapping techniques to eliminate any potential pulmonary vein isolation gaps at the time of the index procedure, in conjunction with the UPUSD ablation technique, can offer an AF ablation procedure that a much more effective and safe procedure.
A total of 472 consecutive first time AF ablation procedure patients with at least one year of follow up were included in this study. The average follow-up was 18 months. Of these 472 consecutive AF ablation procedure patients, 402 underwent HPUSD AF ablation with pulmonary veins isolation as determined by a 10-pole circular mapping catheter (decapolar Spiral mapping catheter, Abbott Laboratories, Chicago, IL) and 70 underwent HPUSD of AF with pulmonary vein isolation as determined by the recently released HD Grid mapping catheter (Abbott Laboratories, Chicago, IL). [Figure 1] shows a side-by-side comparison of the right upper pulmonary vein using both the HD Grid and CMC. On the left concealed pulmonary vein potentials and voltage into the vein are clearly seen whereas they are absent using the traditional CMC.
Figure 1. Image showing the difference between HD grid pulmonary vein mapping versus the circular mapping catheter.
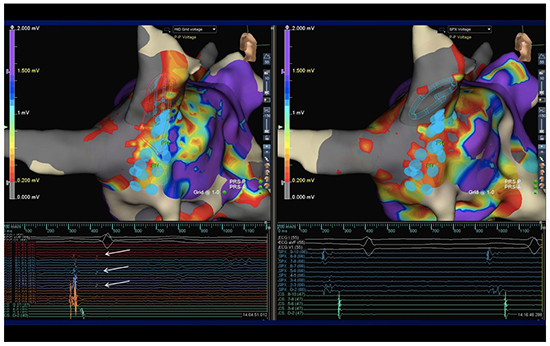
There were no significant differences in ablation catheter technology and the HPUSD ablation approach was the same in both groups. The only significant difference between the two groups was acute pulmonary vein isolation (PVI) being determined by the generally accepted circular mapping catheter (entrance/exit block with 30-minute wait time) versus the new acute PVI criteria using HD Grid mapping catheter. The two groups were then compared with regards to freedom from all atrial arrhythmias and the need for redo procedures.
The outcomes evaluated included recurrent atrial fibrillation and/or atrial flutter. The recurrence of either arrhythmia was determined through the electrocardiography database that includes electrocardiograms, ambulatory monitoring reports, and symptom and auto-triggered event monitor reports from all Intermountain Healthcare facilities as well as outside facilities. In general, follow-up ambulatory monitoring was performed at 3-month intervals post-ablation up to the first year and then with symptomatic recurrence thereafter, although the frequency of this monitoring was determined by the electrophysiologist performing post-ablation care. Discontinuing anti-arrhythmic drug therapy was advocated 3 months post-ablation. Long-term use was variable and dependent on recurrent symptoms of arrhythmia and varied by provider preferences. Other end points included stroke, heart failure hospitalization, and death. These were investigated through interrogation of the electronic medical records, and for death, the Social Security Death Index. Endoscopy was performed for all patients reporting gastrointestinal symptoms potentially indicative of an esophageal injury in the first month following ablation.
High Power Ultra-Short Ablation Technique
All ablation procedures were performed by operators with 1000 ablation experiences. All cases were done with manual catheter manipulation. High power ablation was delivered at 50 W using an irrigated tip catheter with a goal of frequent to continuous catheter movement for an ablation time of 2–3 seconds at each site on the posterior wall and 50 W for 5–15 seconds on the anterior wall in both groups. Each ablation site was then marked on a 3-dimensional map. Isolated single spot ablation procedures were delivered in both groups only if there was a delineable gap, and in this approach a point-by-point approach was not used as an upfront strategy. Normal saline was infused at a rate of 30 mL/min according to manufacturer’s recommendations (Abbott Laboratories, Chicago, IL) for the catheter tip technology used. Catheter movement on the posterior wall was guided primarily by dwell time, but on the anterior walls by attenuation of local electrograms and dwell time. All sites were retargeted in a focused manner with similar dwell times until pulmonary vein isolation was achieved in both groups.
Determination of Pulmonary Vein Isolation
Acute pulmonary vein isolation (PVI) was determined by entrance and exit block as well as a 30-minute wait time for the 402 patients included in the circular mapping catheter group. Use of adenosine or the pace/capture technique to evaluate for dormant conduction was performed at the discretion of the operator.
However, for the 70 patients included in the HD Grid mapping group (Abbott Laboratories, Chicago, IL), PVI was defined as no concealed 1) PV signals, 2) activation into pulmonary veins, or 3) voltage into pulmonary veins. There was no intra-procedural waiting period required for the HD Grid mapping group.
The Student t test and the c2 statistic were used to evaluate baseline and clinical characteristics by using the ablation approach. Continuous variables were described as mean, SD and discrete variables as frequency (percentage). Frequency of 1- and 3-year incidences of AF, AFL, heart failure hospitalization, stroke, and death were determined and compared using the c2 statistic. The Kaplan-Meier survival estimate and the log-rank test for survival were used to evaluate the initial associations of the ablation strategy with the end points. Multivariate Cox proportional hazards regression (SPSS version 22.0, IBM Corporation, Armonk, NY) was performed to determine the risk of stroke, heart failure hospitalization, and death by group. Final models retained only significant (P 0.05) and confounding (10% change in hazard ratio [HR]) co-variables. Two-tailed P values of 0.05 were designated to be nominally significant.
The baseline demographic characteristics are listed in [Table 1]. While the HD mapping group was somewhat younger than the circular mapping group (63.2 13.2 vs 67.1 10.5 years old), the HD mapping group was more likely to have diabetes, hyperlipidemia, tobacco use, renal failure, and coronary artery disease.
Table 1. Demographic and baseline variables of HD grip and CMC group.
| Variable |
CMC (n=402) |
HD Grid (n=67) |
P |
| Age |
67.1 ± 10.5 |
63.2 ± 13.2 |
NS |
| Male |
63% |
70% |
NS |
| Hypertension |
89% |
85% |
NS |
| Diabetes |
31%% |
55% |
<0.001 |
| Hyperlipidemia |
18% |
61% |
<0.001 |
| Smoking |
1% |
7% |
<0.001 |
| Heart Failure |
47% |
37% |
0.02 |
| Stroke/TIA |
12% |
8% |
0.05 |
| Renal Failure |
10% |
25% |
<0.001 |
| PCI/CABG |
14% |
27% |
<0.001 |
| CHADS-VASc |
2.2 |
2.6 |
0.02 |
| Persistent AF |
53% |
55% |
NS |
| Ejection Fraction |
55% |
54% |
NS |
| Body Mass Index |
31 |
31 |
NS |
In both groups, all pulmonary veins were acutely isolated. [Table 2] shows that three-dimensional mapping points were predictably much higher in the HD Grid mapping group when compared to traditional mapping with the circular mapping catheter (20,012 vs 2,818, p>0.001). Notably, procedure times were approximately 30 minutes shorter in the HD Grid mapping group likely due to elimination of a 30-minute wait time to confirm pulmonary vein isolation (70.2 vs 104.3 minutes, p>0.001). Likewise, fluoroscopy times were also markedly reduced with the HD Grid likely due to ease of manipulation with the catheter when compared to the traditional circular mapping catheter (4.2 vs 15.0 minutes, p<0.001). In both groups there were no deaths, esophageal perforations/fistulas, or strokes.
Table 2. Procedural variables of HD grid and CMC groups
| Procedure Variables |
| Variable |
CMC (n=402) |
HD Grid (n=67) |
P |
| All PVs Isolated |
100% |
100% |
NS |
| Fluoroscopy Time |
15.0 |
4.2 |
<0.001 |
| Procedure Time |
104.3 |
70.2 |
<0.001 |
| Mapping Points |
2,818 |
20,012 |
<0.001 |
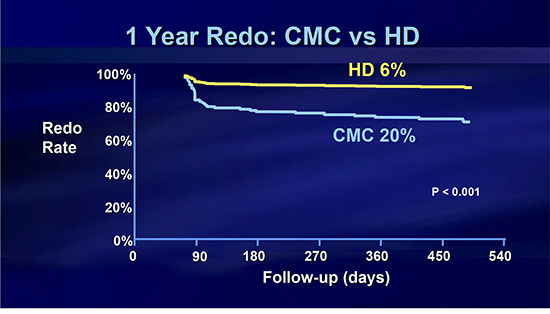
In [Figure 2], the 12-month antiarrhythmic-free freedom from any atrial arrhythmias is shown. A statistically significant 87% success rate was achieved for the HD Grid mapping group when compared to the 75% traditional mapping with the circular mapping catheter. In [Figure 3], the 12-month need for a redo atrial fibrillation procedure is displayed. The redo procedure rate for the HD Grid group was 6% versus the 20% redo rate for the traditional circular mapping catheter group.
Figure 2. Kaplan Meiyer curves showing higher 1 year survival rate of HD mapping group (87%) as compared to CMC group (75%). (P value < 0.001)
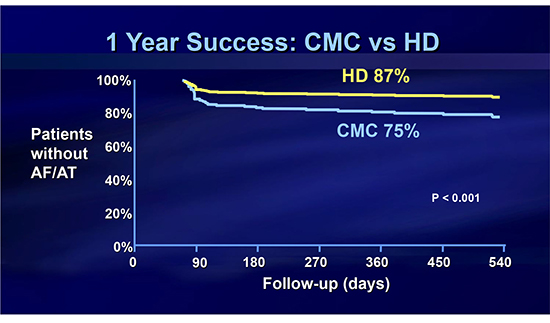
Figure 3. Kaplan Meiyer curves showing lower 1 year redo procedure rate of HD mapping group (6%) as compared to CMC group (20%). (P value < 0.001)
This study shows that HD mapping with the HD Grid mapping catheter at the time of the index atrial fibrillation ablation case may significantly increase the single procedure success rate while at the same time reducing the need for a redo procedure and avoiding any esophageal injury, including even superficial esophageal ulcerations. In addition, HD mapping may also offer more efficient pulmonary vein isolation with less reliance on fluoroscopy.
As pulmonary vein isolation is the cornerstone to atrial fibrillation ablation procedures, durable PVI has been shown to be the key factor to improving freedom from any atrial arrhythmia4. To improve the durability of PVI, many techniques have been proposed such as using adenosine or a pace/capture technique to evaluate for dormant conduction or the use of a contact force sensing ablation catheter to ensure a durable lesion that eliminates any possible residual dormant conduction. More recent studies have suggested that HD mapping may also play a role in detecting incompletely ablated future PVI gaps (areas of dormant conduction) at the time of the index ablation 5.
In this study we used an ultra-short ablation technique of just 2-3 seconds when ablating on the posterior wall of the left atrium as compared to the 5-10 second lesions typically employed with the high power short duration technique. In contrast, our lesions are 5-15 seconds, depending on contact force, when ablating on the anterior wall of the left atrium. This ultra-short ablation protocol when ablating on the posterior wall of the left atrium is an approach that has evolved over the 16 plus years we have used this technique. Initially, we ablated for 2-5 seconds at each posterior wall location, however, that was later decreased to 2-3 seconds after we experienced a single esophageal mucosal ulcer in 2010.
While ultra-short ablations on the left atrial posterior wall have allowed us to completely avoid any esophageal injury, including superficial mucosal ulcerations, in thousands of ablation cases and without the need for esophageal temperature monitoring, the clear drawback of limiting lesions to just 2-3 seconds (50 W with goal contact force of 5-10 g) on posterior wall is that incomplete lesions with residual dormant conduction may occur. The advantage of using the HD Grid in this study is that HD mapping allowed us to see these areas of dormant conduction thereby improving success rates and decreasing redo procedure rates while not having to sacrifice the esophageal safety benefit our ultra-short ablation approach on the posterior wall.
While it has long been known that the traditional circular mapping catheter cannot adequately assess for residual dormant conduction at the time of the index ablation procedure, with the advent of HD mapping catheters there is much excitement that these new technologies may overcome the shortcomings of traditional mapping for PVI gaps. Indeed, our current study suggests that through elimination of 1) all pulmonary vein electrograms, 2) all activation into the pulmonary veins, and 3) all antral voltage less than 0.5 mV as per mapping in sinus rhythm with the HD Grid catheter that dormant conduction can be extinguished during the index procedure thereby significantly improving 12-month success rates as well as reducing the need for redo procedures.
HPUSD AF Ablation, as guided by HD Grid mapping, may significantly improve freedom from any atrial arrhythmias and the need for redo procedures as well provide operators utilizing this technique reassurance that any potential esophageal injury is likely extremely low. In addition, HPUSD AF ablation, as guided by HD Grid mapping, may allow for a marked shortening of procedure and fluoroscopy times when compared to the traditional circular mapping catheter.