Left Atrial Size May Predict Blood Pressure Improvement After Ganglionated Plexi Ablation in Addition to Pulmonary Vein Isolation
Daniel Sohinki1, Isma Javed2, Benjamin Scherlag23, Wesley Thomas2, Sunny Po23, Tarun Dasari2
1Department of Cardiology, Medical College of Georgia at Augusta University, Augusta, GA.2Department of Cardiology.3Heart Rhythm Institute, University of Oklahoma Health Sciences Center, Oklahoma City, OK.
Hypertension (HTN) and atrial fibrillation (AF) commonly co-exist. An improvement in control of HTN in a subset of patients undergoing AF ablation was previously demonstrated by our group. In the present study, we aimed to assess whether left atrial (LA) size based on transthoracic echocardiography may predict the patients who demonstratebetter HTN improvement after ganglionated plexus ablation (GPA) in addition to pulmonary vein isolation (PVI).
This was a retrospective chart review of patients with AF and HTN who underwent GPA+PVI. Patients were divided into 2 groupsbased on LA size: Patients with normal LA size and patients with LA enlargement. Systolic blood pressure (SBP) levelswere compared at baseline, and 3, 6, and 12 months post-ablation. The primary endpoints of the study weremean systolic blood pressure change compared between groups from baseline to 12-months, as well as the absolute difference in systolic blood pressure at 12 months follow-up.Medical therapy for HTN was also assessed before the procedure, and at 12 months post-procedure.
53 patients (37 with LA enlargement, 16 with normal LA size) met inclusion criteria. At 12 months follow-up, SBP was 136.46 ± 22.38 mmHg in patients with LA enlargementand 118.25 ± 9.81 mmHg in patients with normal LA size (estimated difference of 19.04 ± 6.98 mmHg, p = 0.01). Patients with normal LA size were on significantly fewer anti-hypertensive agents at 12 months (2.33 ± 1.49 vs. 1.44 ± 1.21, p < 0.05).
In patients undergoing PVI+GP ablation, normal LA size may predict HTN improvement at 12 months post-procedure. Normal LA size may identify hypertensive AF patients for whom autonomic modulation could be an effective therapy.
Key Words : Hypertension, Atrial Fibrillation, Left Atrial.
Daniel Sohinki, MD, MSc, FACC, FHRS
Medical College of Georgia at Augusta University
1120 15th Street
Augusta, GA 30912
Hypertension (HTN) remains the greatest single contributor to cardiovascular death worldwide, and is second only to tobacco abuse as a preventable cause of all mortality 1. Importantly, as many as 13-17% of patients have HTN that is resistant to multi-drug therapy 1, suggesting that the mechanisms targeted by most anti-hypertensive medications [e.g. renin-angiotensin-aldosterone system (RAAS) hyperactivity, hypervolemia and elevated vascular tone] may be less active in these patients.
Hypertension is the most common co-morbid condition in patients with atrial fibrillation (AF). Atrial fibrillation occurs in 3-4% of the population over the age of 65, with 80% of such patients having concomitant HTN 3. Hypertension contributes to the pathogenesis of AF via multiple mechanisms, including the development of diastolic dysfunction and elevated left atrial (LA) pressure, LA dilatation and fibrosis, and remodeling of the cardiac autonomic nervous system (CANS) which supports development of the electrophysiologic substrate that sustains AF 1, 4-6. Because of the effects of chronically elevated vascular tone and RAAS hyperactivity on cardiac afterload and ventricular and atrial wall stress, chronic HTN tends to cause predictable morphologic and functional changes in the heart that are easily assessed by transthoracic echocardiography (TTE) [e.g. left-ventricular hypertrophy (LVH), diastolic dysfunction, and LA enlargement] 9,10. It seems likely that cardiac morphologic changes vary based on the mechanism underlying HTN in a given patient. Thus, echocardiographic features could potentially be used to distinguish between different forms of HTN. In patients with concomitant AF and HTN, appropriate blood pressure (BP) management is critical in reducing the structural and electrophysiologic remodeling that allow for AF initiation and maintenance 7,8.
We have previously noted an improvement in control of HTN in a subset of patients undergoing AF ablation via pulmonary vein isolation (PVI) with additional ablation of the cardiac autonomic ganglionated plexi (GPs). Specifically, LA size was shown to predict BP improvement after PVI+GP ablation, though that study was small, and did not systematically exclude other causes of LA enlargement. Such patients may have alternative mechanisms underlying their HTN, and early identification of these patients would have important diagnostic and therapeutic implications. Thus, the purpose of this study was to assess the predictive ability of LA enlargement on TTE with respect to BP improvement after PVI+GP ablation.
The Institutional Review Board at the University of Oklahoma reviewed and approved the study protocol, and the study complies with the principles of the revised Declaration of Helsinki. Patients were eligible for the study if they had undergone catheter ablation for AF at our institution from 10/2010 through 6/2015 and remained in sinus rhythm at 12 months follow-up. Eligible patients were required to carry a previously-established diagnosis of HTN (defined as systolic blood pressure > 140 mmHg or diastolic blood pressure > 90 mmHg on two separate in-office readings), have GP ablation performed as part of the ablation lesion set and have follow-up data at 12 months post-ablation. Exclusion criteria included age less than 18, left ventricular ejection fraction (LVEF) < 40%, stroke or myocardial infarction within the past 6 months, greater than moderate valvular stenosis or regurgitation as assessed by pre-procedure TTE, prior ablation for AF, or presence of a prosthetic heart valve. Basic demographic and medical history information were collected on all participants, including age, gender, race, ethnicity, co-morbid medical conditions, medications, duration of AF prior to ablation, and basic TTE measures of cardiac structure and function.
This was a retrospective chart review study of patients with HTN presenting for catheter ablation of AF. Records of patients who had presented to our EP lab for AF ablation during the study period were individually screened for inclusion and exclusion criteria. Patients were divided into two groups according to LA size (enlarged vs. normal size). Left atrial enlargement was defined as either a left atrial volume index (LAVi) ≥ 29 ml/m2body surface area (BSA)and/or an anteroposteriordiameter ≥ 4.0 cm on the pre-procedure TTE assessed in the parasternal long-axis view. Baseline blood pressure was recorded from the pre-procedure in-office assessment. Follow-up blood pressure readings were recorded during routinely-scheduled 3, 6, and 12 month post-procedure office visits. All blood pressure measurements were taken according the published guidelines, with brachial blood pressure readings taken after the patient was seated for 5-minutes with legs uncrossed.
Regarding procedural care, our practice is to discontinue anti-arrhythmic drugs for 5 half-lives prior to the procedure, with the exception of amiodarone which is discontinued for 6 weeks. Patients taking Coumadin for stroke prophylaxis continue uninterrupted, while patients taking direct oral anticoagulants (DOACs, e.g. apixaban, rivaroxaban, or dabigatran) had this medication discontinued 24 hours prior to the procedure.
Our practice is to perform AF ablation under general anesthesia utilizing a volatile anesthetic (typically sevoflurane or desflurane), a paralytic (typically rocuronium or vecuronium) as well as an intravenous propofol infusion. Vascular access was obtained in the bilateral common femoral veins, and diagnostic catheters were placed in the right atrial (RA) appendage, coronary sinus (CS), and His-position. Invasive arterial pressure monitor via radial or femoral arterial line was performed in all patients. The CARTOSOUND® module was used to create a basic shell of the left atrium for the purpose of mapping and merging with pre-procedure computed tomography scan of the left atrium. Double trans-septal puncture was then performed under intracardiac echocardiographic and fluoroscopic guidance, and deflectable and SL-1 sheaths were advanced into the left atrium. An electroanatomic map was createdeither point-by-point via a mapping catheter (ThermoCool® SmartTouch® ST/SF) (Biosense Webster®, Irvine CA) or via fast anatomic mapping utilizing the Pentaray®(Biosense Webster®, Irvine CA) multi-electrode mapping catheter. The endocardial locations corresponding to the epicardial LA GPs were mapped by placing the mapping catheter in the correct anatomic region and performing high-frequency stimulation [Figure 1]. High frequency stimulation was performed using a Grass Stimulator (S-88 stimulator, Grass Instruments Division, Astro Med Inc.). Locations with a positive response (defined as > 3 seconds of asystole noted on the arterial pressure waveform) were marked on the map. Each marked area was targeted for ablation (25-35 W for 30 seconds per lesion). Successful GP ablation was defined as elimination of the asystolicresponse. After ablation of all LA GPs, pulmonary vein isolation was performed utilizing wide area circumferential ablation, merging with the GP lesion clusters when they were noted to be close to the left and right PV antra.
Figure 1. Locations of the epicardial left atrial ganglionated plexi - The white and yellow lines denote the boundaries of the left and right PV antra, respectively. Panel A is a PA view, with orange circles denoting the locations of the SLGP, ILGP and IRGP. Panel B is an AP view, with the orange circle denoting the location of the ARGP. Red tags are a representative sampling of locations that demonstrated a positive response to HFS. Notably, areas corresponding to the ILGP frequently extended inferomedially along the posterior wall, and areas corresponded to the SLGP frequently extended anteriorly towards the base of the LAA
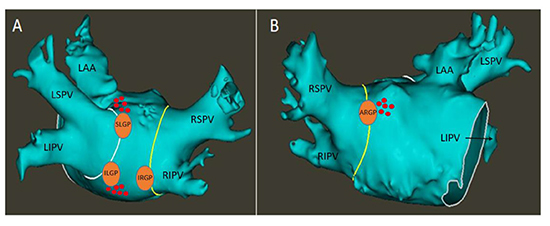
LA – Left atrium. CT – Computed tomography. PV – Pulmonary vein. PA – Posteroanterior. SLGP – Superior left ganglionated plexus. ILGP – Inferior left ganglionated plexus. IRGP – Inferior right ganglionated plexus. AP – Anteroposterior. ARGP – Anterior right ganglionated plexus. LSPV – Left superior pulmonary vein. LIPV – Left inferior pulmonary vein. RSPV – Right superior pulmonary vein. RIPV – Right inferior pulmonary vein. LAA – Left atrial appendage. HFS – High-frequency stimulation.
The primary endpoint of the study was the mean change in SBP compared between groups at 12 months post-ablation, as well as the difference in SBP at 12 months. Secondary endpoints to be analyzed included diastolic blood pressure at 12 months as well as number of prescribed anti-hypertensive medications compared between the two groups at 12 months.
Repeated measures analysis of variance (ANOVA) was used to compare themean change in SBP, DBP and change in number of medications over time between the two groups. This was a mixed model with time included as a repeated factor, and LA size and baseline demographic data included as non-repeated factors. An interaction between the group and time point was examined and dropped from the model if not statistically significant. The model included fixed effects of demographic data, time point and left atrial size, as well as random effects of subject, prescribed medications, as well and subject-by-group. A significant interaction between LA size and time point indicated a significant effect of LA size, and least squared means were then used to compare each continuous variable at each follow-up time point between groups. Residual plots were created to evaluate the appropriateness of modeling assumptions including linearity, normality of the residuals and constant variance. Continuous and categorical demographic data are presented as mean ± standard deviation [or median (interquartile range) when the Wilcoxon rank sum test was used] or percentages, as appropriate. Baseline characteristics were compared using the student’s t-test or Wilcoxon rank sum test, or chi-square test or Fisher’s exact test [if more than 20% of the expected counts from the contingency table of a demographic variable (yes/no) by group (normal LA size/LA enlargement) were less than 5 or if any of the expected counts were 0], where appropriate. The reasons for exclusion were descriptively summarized and tracked.
Because this was a retrospective study examining all patients over the specified time frame, no formal power calculation was done prior to study initiation to assess the number of patients required to detect a change in any of the continuous outcome measures. In the study by Sharma et al., SBP at 12 months in the normal LA size group was 119.2 ± 13, vs. 130.4 ± 13.4 in the LA enlargement group 11. Assuming a power of 0.8 and alpha of 0.05, we would need a total of 34 patients to detect this difference. Because our final sample size of patients who met inclusion criteria and were not excluded was 53, we felt this was an adequate number to perform the analysis.
From 10/2010 through 6/2015 a total of 314 patients presented to our lab for ablation of AF. Screening flow and reasons for exclusion are given in [Figure 2].
Figure 2. Screening and exclusion flow for the study.
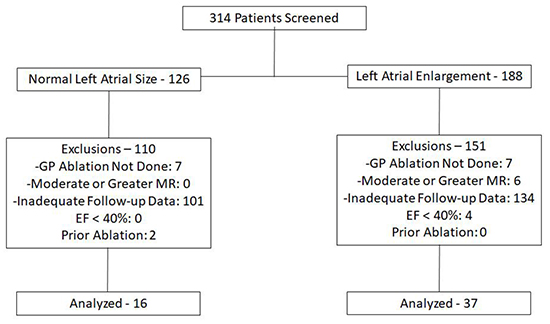
As shown, the majority of exclusions occurred because of inadequate follow-up data. The final cohort consisted of 37 patients with LA enlargement and 16 patients with normal LA size. Baseline characteristics for each group are compared in [Table 1]. As noted, the normal LA size group was a larger proportion female. A larger proportion of patients with normal LA size were diabetic. Additionally, a significantly smaller proportion of patients with normal LA size had been prescribed a calcium-channel blocker prior to ablation. With respect to cardiac structure and function, there was a significant difference in LA size (both by AP diameter and LAVi), as was expected for this study design. Interestingly, LVEFwas significantly greater in the normal LA size group as compared to the LA enlargement group. Left ventricular thickness parameters did not differ significantly between the two groups. Only a minority of studies were technically adequate to comment on diastolic function, and so this was not compared between the two groups.
Table 1. Baseline Characteristics.
|
LA Enlarged |
Normal LA Size |
p-value |
|
(n=37) |
(n=16) |
|
| Age (years) |
65.0 ± 11.65 |
60.44 ± 11.91 |
0.21 |
| Male |
26 (0.70) |
7 (0.31) |
0.01 |
| Time from diagnosis (months) |
60 (36-120) |
48 (36-180) |
0.48 |
| Diabetes |
2 (0.05) |
5 (0.31) |
0.02 |
| Coronary Disease |
9 (0.24) |
4 (0.25) |
>0.9 |
| Obstructive Sleep Apnea |
13 (0.35) |
5 (0.31) |
>0.9 |
| Body Mass Index |
33.06 ± 9.08 |
31.08 ± 7.8 |
0.57 |
| Ejection Fraction (%) |
59.46 ± 13.91 |
68.86 ± 8.21 |
0.01 |
| CHA2DS2VASc Score |
2.3 ± 1.0 |
2.63 ± 1.02 |
0.28 |
| Beta-Blocker |
28 (0.76) |
11 (0.69) |
0.74 |
| ACEi/ARB |
19 (0.51) |
5 (0.31) |
0.18 |
| Thiazide Diuretic |
6 (0.16) |
2 (0.13) |
>0.9 |
| Aldosterone Receptor Antagonist |
1 (0.02) |
1 (0.06) |
0.52 |
| Calcium Channel Blocker |
14 (0.38) |
1 (0.06) |
0.02 |
| Other (Loop diuretic, nitrate, alpha-blocker) |
6 (0.16) |
4 (0.25) |
0.47 |
| Left Atrial Size (cm) |
4.89 ± 0.63 |
3.56 ± 0.47 |
<0.0001 |
| Left Atrial Volume Index (ml/m2) |
31.4 (28.5-34) |
18.8 (16.5-21.25) |
0.005 |
| Left Ventricular Posterior Wall Thickness (End-diastole, cm) |
1.25 ± 0.44 |
1.08 + 0.24 |
0.1 |
| Left Ventricular Septal Thickness (End-diastole, cm) |
1.19 ± 0.21 |
1.14 ± 0.25 |
0.48 |
| Ganglionated Plexi Ablated (n) |
3.03 ± 0.32 |
3.08 ± 0.29 |
0.64 |
Data are presented as mean ± standard deviation, median (interquartile range) or count (proportion), as appropriate. Statistical comparisons were made using student’s t-test or Wilcoxon rank sum test as appropriate for continuous variables, or chi-square test or Fisher’s exact test as appropriate for categorical variables. ACEi – Angiotensin-Converting Enzyme inhibitor. ARB – Angiotensin Receptor Blocker.
For each of the continuous variables tested, we first used the repeated measures ANOVA model to test the group-time interaction. The interaction was significant for SBP (p = 0.03), indicating that SBP changed over time in the normal LA size group but not the LA enlargement group. Similarly, The interaction was also significant for number of anti-hypertensive medications, indicating a change in medical therapy over time in the normal LA size group, but not the LA enlargement group (p = 0.03). The interaction was not significant for DBP (p = 0.54).
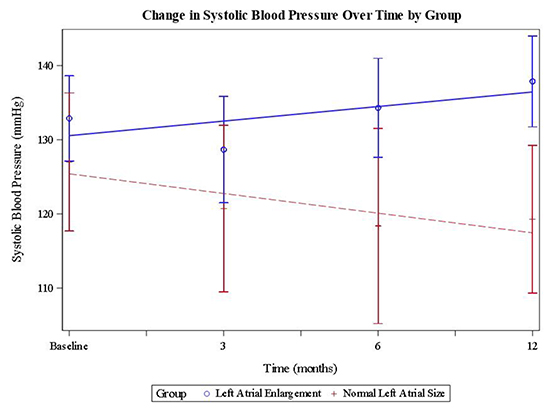
Because the interaction between LA size and time was significant, least squared means were used to estimate mean SBP change over time within each group, as well as to compare SBP at 12-months between groups.Mean SBP did not change significantly in the LA enlargement group at 12-months (increase of 3.72± 3.15 mmHg, CI -10.08 – 2.65 mmHg, p = 0.25). In the normal LA size group, mean SBP decreased by 10.33 ± 5.1 mmHg, (CI 0.06 – 20.60 mmHg, p = 0.04). At 12-months follow-up, Mean SBP was 136.46 ± 22.38mmHg in the LA enlargementgroup and 118.25 ± 9.81 mmHg in the normal LA size group. Accordingly, the ANOVA model predicted a 19.04 ± 6.98 mmHg lower SBP in the normal LA size group as compared to the LA enlargement group at 12-months months after adjusting for baseline differences in SBP (CI 4.94 – 33.14 mmHg, p = 0.01). There was no significant change in diastolic BP in the normal LA size group as compared to the LAE group at 12 months (CI -13.92 – 3.55, p = 0.24).[Table 2] gives mean BP levels (± standard deviation) at each time point for each group and p-values for the comparison at each time point.[Figure 4] shows mean SBP at baseline, 3 months, 6 months, and 12 months for each group, with fitted regression lines.
Table 2. Comparison of systolic and diastolic blood pressure at each time point between groups Data are presented as mean ± standard deviation.
| Baseline |
LAE |
Normal LA Size |
P-value |
| Systolic Blood Pressure (mmHg) |
133.65 ± 17.62 |
127.67 ± 14.23 |
0.29 |
| Diastolic Blood Pressure (mmHg) |
79.43 ± 15.2 |
72.53 ± 10.64 |
0.08 |
| 3 months |
|
|
|
| Systolic Blood Pressure (mmHg) |
127.04 ± 20.43 |
120.83 ± 11.02 |
0.24 |
| Diastolic Blood Pressure (mmHg) |
77.37 ± 11.23 |
73.50 ± 7.40 |
0.17 |
| 6 months |
|
|
|
| Systolic Blood Pressure (mmHg) |
131.60 ± 21.02 |
123.25 ± 13.31 |
0.04 |
| Diastolic Blood Pressure (mmHg) |
77.40 ± 9.81 |
70.50 ± 6.91 |
0.07 |
| 12 months |
|
|
|
| Systolic Blood Pressure (mmHg) |
136.46 ± 22.38 |
118.25 ± 9.81 |
0.01 |
| Diastolic Blood Pressure (mmHg) |
75.27 ± 10.23 |
72.81 ± 7.96 |
0.13 |
LAE – Left atrial enlargement.
Figure 4. Mean systolic blood pressure at each time point for each group with fitted regression lines. Error bars represent confidence intervals.
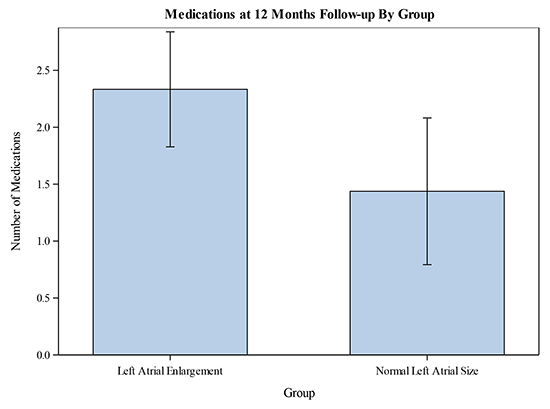
Least squared means were also used to compared number of anti-hypertensive medications between the two groups at 12-months. Patients in the LA enlargement group were prescribed 2.33 ± 1.49anti-hypertensive medications at 12 months post-ablation, while patients in the normal LA size group were prescribed 1.44 ± 1.21 anti-hypertensive medications at 12 months. Accordingly, the ANOVA model estimated reduction in the number of prescribed anti-hypertensive medications in the normal LA size group of 1.17 ± 0.37 (CI 0.42 – 1.92, p = 0.003) after adjusting for baseline differences in the number of prescribed anti-hypertensive medications. [Figure 3].
Figure 3. Mean number of prescribed medications at 12-months compared between the two groups. Error bars represent confidence intervals.
Regarding safety endpoints, there was a single pericardial effusion in each group that required pericardiocentesis. Otherwise, no significant complications were noted in any patient in either group.
The principle finding of this study is that patients with HTN and normal LA size on pre-procedure TTE experienced a significantly-greater decrease in SBP, and had significantly lower mean SBP at 12-months follow-up after PVI+GP ablation as compared to patients with LA enlargement. Notably, patients with normal LA size were also on fewer anti-hypertensive medications at 12-months follow-up, suggesting that these patients were able to have medical therapy de-escalated following PVI+GP ablation.
Relatively little research has been done into the effect of AF ablation on HTN. One retrospective report found an improvement in systolic BP in patients with a successful AF ablation (defined as lack of AF recurrence of greater than 30 seconds off of anti-arrhythmic drug therapy) as compared to patients with failed ablation. Interestingly, while that study did not further dichotomize patients with successful ablation, the reported within-group change in systolic BP in the success group was not significant (129 ± 17 mmHg baseline vs. 125 ± 14 mmHg at 1 year, p = 0.075) 12. Of note, because wide-area circumferential ablation would be expected to at least partially transect all major GPs, the relative importance of PVI vs. PVI+GP ablation remains unclear. Several prior studies have reported on the salutary endocrine 13 and clinical 14,15 effects of sinus rhythm maintenance. Our study adds to this work by more closely-examining patients who were successfully ablated, and demonstrating that improvement in blood pressure control can be predicted by echocardiographic features.
There are several possible reasons while LA size may be predictive of BP improvement after PVI+GP ablation. Most obviously, greater degrees of LA enlargement may simply be a reflection of more advanced HTN. As such, it may be that GP ablation is less effective in improving BP control once a critical degrees of atrial and vascular remodeling has occurred. As such, other measures reflecting chronicity of HTN would be expected to differ in parallel with LA size, though there was no significant difference noted in the LV thickness parameters recorded on pre-procedure TTE. Additionally, there was a non-significant trend towards lower time since diagnosis in the normal LA size group. However, this is an imperfect measure, as it does not capture the duration of subclinical HTN prior to official diagnosis.
Alternatively, the mechanism underlying HTN in patients with normal LA size may differ from patients with LA enlargement, and improvement after GP ablation may be evidence of a significant component of dysautonomia. The concept of autonomically-mediated, or “neurogenic” HTN is not new. Much of the early investigation into the mechanisms of HTN focused on neural mechanisms 16-19. Dustan et al. put forth the concept of “cardiogenic hypertension”, that was driven by elevated contractility and sympathetic tone 17. The landmark studies of Goldblatt et al. 20 and others refocused research efforts on RAAS hyperactivity, intravascular volume overload, and endocrine mechanisms to explain HTN, and much of current medical therapy is aimed at combatting these mechanisms. As noted above, a significant proportion of patients with HTN fail to respond to these therapies, and autonomic hyperactivity is a reasonable alternative mechanism. Because of the inclusion of GP ablation in the lesion set, one possibility is that HTN in the patients with normal LA size is driven by dysautonomia as opposed to RAAS hyperactivity, or chronically-elevated vascular tone. Interestingly, patients with normal LA size in our study had significantly greater LVEF as compared to patients with LA enlargement, suggesting elevated cardiac autonomic activity and increased contractility as a possible mechanism of HTN in these patients.
Relatively little clinical research has been done regarding medical therapy targeting the sympathetic nervous system as primary therapy for HTN. Neurogenic HTN would theoretically be more likely to respond to alpha and/or beta blockade, because of the direct effect of elevated sympathetic output on alpha- and beta-adrenergic receptors 21. A large meta-analysis examining the role of beta blockade in HTN management showed beta-blockers to be inferior to other anti-hypertensive agents with respect to blood pressure lowering, and even showed a higher risk of stroke in patients treated with beta blockers compared to other anti-hypertensive agents 22. However, these studies were performed in undifferentiated hypertensive patients, and it is still possible that a subset of these patients could have neurogenic HTN that responded more favorably to adrenergic blockade. Additionally, many of these studies were performed using older, “vasoconstricting” beta-blockers (e.g. Atenolol), with studies using combined alpha/beta-blocking agents (e.g. carvedilol, labetalol) being poorly-represented 23. Similarly, the lack of clear antihypertensive effects by renal sympathetic denervation may result from, at least in part, performing the procedure in undifferentiated HTN patients.
Importantly, histologic and anatomical data have shown that the epicardial GPs are mixed (i.e. consist of both sympathetic and parasympathetic neurons) 24. Thus, the mechanism underlying the salutary effect of GP ablation on AF ablation durability and HTN control may involve both sympathetic and parasympathetic denervation. Several prior studies have demonstrated evidence of sympathetic denervation after GP ablation, including shortening of the corrected QT interval (QTc) after GP ablation in patients with congenital long-QT syndrome 25, as well as both shortening of the QTc and RR interval in patients with vasovagal syncope 26. Thus, if sympathetic hyperactivity at the level of the epicardial GPs were playing a significant role in HTN in a subset of patients, GP ablation would be expected to have a beneficial effect by modulating sympathetic output from the GPs. As a result, this would reduce the likelihood of ectopic firing and structural remodeling required for AF initiation and maintenance.
A major challenge remains in identifying patients with neurogenic HTN, and our study results point to echocardiographic markers of cardiac structure and function as possible discriminating features (specifically LA size in this study). As noted above, LVEF was also significantly higher in patients who responded favorably to PVI/GP ablation, in line with previous reports showing elevated cardiac output is present in the early-stages of HTN, and this can be favorably treated with autonomic blockade [20]. Other markers of neurogenic HTN have been proposed, including HTN associated with obstructive sleep apnea(OSA) or prior stroke, labile HTN, concomitant sinus tachycardia, and a demonstrated lack of volume or RAAS system excess 23. Whether or not these markers are accurate in identifying neurogenic HTN or predicting a favorable response to autonomic modulation remains to be studied. Still, the research described above argues for a more personalized approach to the hypertensive patient, with medical therapy selected based on the presumed underlying mechanism (volume-mediated, RAAS-mediated, or neurogenic) 23. Using echocardiographic markers, including LA dimensions and LVEF, as screening tools to identify patients more likely to respond to autonomic modulation may allow for better patient selection, both in the clinical and investigational setting.
Interestingly, there was a greater proportion of diabetic patients in the normal LA size group. Diabetic patients are well-known to have significant autonomic neuropathy 27, and there is also evidence of CANS dysfunction in diabetic patients. Ayad et al. 28 demonstrated the presence of cardiac autonomic neuropathy (defined by abnormal heart rate variability and postural hypotension) in 40% of diabetic patients they screened. The presence of autonomic neuropathy independently predicted the presence of HTN, with the prevalence of HTN increasing with severity of neuropathy. Thus, the presence of other conditions known to be associated with systemic dysautonomia may increase the likelihood that concomitant HTN is neurogenic, and may predict a favorable response to autonomic modulation.
This study has several limitations. Principally, there are multiple causes of LA enlargement other than arterial HTN. While significant valvular heart disease patients were excluded, we were unable to assess the role of other factors (e.g. diastolic dysfunction) on LA size because of the retrospective nature of the study and lack of assessment of diastolic function on many of the pre-procedure TTEs. Still, patients with normal LA size are unaffected by this issue and would be expected to be less heterogeneous with respect to causes of their echocardiographic findings than patients with LA enlargement. Second, a significant proportion of screened patients were excluded because of lack of follow-up data. Given the relatively small sample size analyzed in our study, the possibility of both type I and type II statistical errors remains a concern. Third, as a general comment, this study is retrospective and non-randomized, though the possibility of a selection bias is relatively low given that patients were grouped based on an objective finding (i.e. LA size), and the entire AF ablation population for our lab over the study period was screened for inclusion. Additionally, it is unclear from our data the impact of “time since diagnosis of HTN” on blood pressure improvement after PVI+GP ablation. As noted above, elevated cardiac output is principally seen early in the clinical course of HTN, and so patients in the normal LA size group may simply have had a shorter duration of HTN than patients in the LA enlargement group, making them more likely to respond to autonomic modulation. While there was a trend towards a shorter duration of HTN in the normal LA size group, this was not statistically significant. Finally, as wide-area circumferential ablation would be expected to at least partially transect the epicardial GPs based on anatomic location, the relative importance of PVI itself vs. the addition of targeted GP ablation remains unclear from our study. Further research could include a comparison to patients who underwent PVI alone, though the practice at our institution since 2003 has been to perform PVI+GP in all patients with paroxysmal AF. Thus, no patients were available for this comparison.
In patients with AF and concomitant HTN, normal LA size predicts improvement in blood pressure control after PVI+GP ablation. This suggests an alternative underlying mechanism in this population. Because of the inclusion of GP ablation in our lesion set, and because the presence of normal LA size may indicate less LA structural remodeling, we hypothesize that these patients have a neurogenic form of HTN, and our study points towards LA size as a potential biomarker for this condition. Further evidence of autonomic dysregulation in this study population includes a greater proportion of diabetic patients, and a greater LV ejection fraction, both of which may potentially serve as markers of autonomic dysfunction that may predict response to autonomic modulating therapies.