Electrophysiological Changes of the Atrium in
Patients with Lone Paroxysmal Atrial Fibrillation
Osmar Antonio Centurion, MD, PhD, FACC1, Shojiro Isomoto, MD, PhD2, Akihiko Shimizu, MD, PhD, FACC3
1Division of Electrophysiology and Arrhythmias, Cardiovascular Institute, Sanatorio Migone-Battilana, Asuncion, Paraguay. Departamento de Cardiologia. Primera Catedra de Clinica Medica. Universidad Nacional de Asuncion.2Health Screening Center, Japanese Red Cross Nagasaki Atomic Bomb Hospital, Nagasaki.3Faculty of Health Scinces, Yamaguchi Gradeate School of Medicine.
Paroxysmal atrial fibrillation (PAF) is a common arrhythmia, and it is associated with various cardiac conditions. On the other hand, lone PAF has no identifiable underlying cause, and can occur any time for no apparent reason. The underlying causes may modify the electrophysiological properties of the atrium in different ways and extent. However this setting may be different in patients with lone PAF. We sought to investigate the atrial electrophysiological properties in lone PAF.
This study included 62 control subjects (Control group) and 58 patients with lone PAF (LAF group). The following atrial vulnerability parameters induced by programmed atrial stimulation were assessed and quantitatively measured: 1) the atrial effective refractory period (ERP), 2) the atrial conduction delay (CD) zone, and 3) the maximum CD.
The mean atrial ERP of the Control group was 215±29 ms, and that of LAF group was 208±28 ms, p<0.05. The mean atrial CD zone of the LAF group was (50±28 ms) significantly greater than that of controls (34±22 ms) (p<0.01). The mean maximum CD of the LAF group (62±29 ms) was also significantly greater than that of controls (43±20 ms) (p<0.01).
There is a greater conduction delay of the atrium and shorter refractoriness in patients with lone PAF. Patients without underlying causes for the development of PAF exhibit abnormalities in the electrophysiological properties of the atrium.
Key Words : Atrial Vulnerability, Atrial Refractory Period, Atrial Conduction Time, Atrial Fibrillation, Atrial Conduction Delay.
Correspondence to: Zhinong Wang, Room 204, NO.24 Building, Changhai Hospital, 168# Changhai Road, Shanghai
200433.
The mechanism of atrial fibrillation (AF) is considered to be either a spiral wave with a continuously changing activation wavefront pattern, random multiple independent reentrant wavelets wandering in the atria around arcs of refractory tissue, or accentuation of focal activity originating mainly from the pulmonary veins or superior vena cava.1-5 Paroxysmal atrial fibrillation (PAF) is a common arrhythmia, and it is associated with various cardiac conditions, including ischemic heart disease, cardiomyopathy, pericardial disease, valvular heart disease, hypertension, congestive heart failure, preexcitation syndrome, and sick sinus syndrome. Non-cardiac conditions, such as aging, autonomic tones, thyroid function, acute alcohol intoxication, metabolic or electrolyte disturbances, and drugs, also affect the initiation, maintenance, and termination of AF.6 On the other hand, lone PAF has no identifiable underlying cause, and can occur any time for no apparent reason. The underlying causes may modify the electrophysiological properties of the atrium in different ways and extent. However, this setting may be different in patients with lone PAF. Ectopic foci from the pulmonary veins may act as drivers for maintaining AF, which has been proven by the results of the surgical or catheter isolation of the pulmonary vein orifices.7 Since not all patients with atrial arrhythmias initiate AF, a substrate for atrial propensity to AF is required for AF initiation and maintenance.8-10 Abnormal responses of the atrium can be elicited by programmed atrial stimulation, such as repetitive atrial firing, fragmented atrial activity, and intraatrial conduction delay, and have been more frequently observed in patients with nonidiopathic PAF. Shorter atrial effective refractory period has been also shown to be of electrophysiological significance in the genesis of AF.11-13 We sought to investigate the atrial electrophysiological properties in patients with lone PAF.
This study included 58 patients with lone PAF (32 men and 26 women; age 17-84 years, mean 55±13) and 62 patients without a history of AF as control subjects (34 men and 28 women; age 16-83 years, mean 53±22), who underwent electrophysiological study. All patients had normal electrocardiogram, echocardiography and chest radiography. No patients with congestive heart failure, ischemic heart disease, cardiomyopathy, Wolff-Parkinson-White syndrome, cerebrovascular accidents, or serum electrolyte disturbances were admitted to the study. No patient in whom echocardiography showed atrial enlargement was included in this investigation. In the control group, electrophysiological study was performed because of atrioventricular nodal reentrant tachycardia in 46 patients, sinus nodal reentrant tachycardia in 4 patients, and idiopathic ventricular tachycardia in 12 patients.
Electrophysiological Study
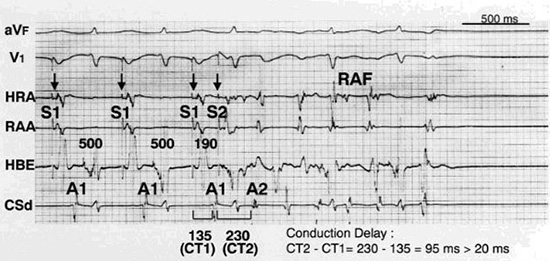
The study protocol was approved by the regional ethics committee. Written informed consent to the study was obtained from all patients. All cardioactive medications were withdrawn for a washout period equal to >5 elimination half-lives before the electrophysiological study. Amiodarone was not used in any of the patients. Five catheters were placed at the right atrial appendage, high lateral right atrium, coronary sinus, His bundle area, and right ventricular apex. Using a programmable electric stimulator with rectangular current pulses of 2 ms duration at twice the diastolic threshold, programmed single premature atrial stimulus was delivered after a train of eight atrial paced beats at a cycle length of 500 ms from the right atrial appendage. S1 and A1 refer to the driving stimulus and the atrial electrogram, respectively, of the basic drive beat. S2 and A2 refer to the stimulus artifact and the atrial electrogram, respectively, of the induced premature beat Figure 1. The coupling interval between S1 and S2 was decreased in 10 msec steps until S2 was no longer captured.
Figure 1. Programmed atrial extrastimulus testing in a patient with lone paroxysmal AF showing atrial CD. S1 and A1 refer to the driving stimulus and the atrial electrogram, respectively, of the basic drive beat. S2 and A2 refer to the stimulus artifact and the atrial electrogram, respectively, of the induced premature beat. The atrial extrastimulus was programmed at a coupling interval of 190 ms with a driving cycle length of 500 ms. The S1-A1 interval (CT1) in the distal coronary sinus (CSd) was 135 ms. At the premature beat, S2-A2 interval (CT2) prolonged to 230 msec. The maximum CD in this patient was 95 msec. HLRA indicates high lateral right atrium; RAA, right atrial appendage; and HBE, His bundle area
Measurements and Definitions
The ERP of the right atrial appendage was defined as the longest S1-S2 interval that did not elicit an atrial depolarization. The conduction time from the stimulus artifact to the distal electrode pair placed at the coronary sinus was measured as the interatrial conduction time during a single premature stimulation performed from the right atrial appendage.
The atrial CD was defined as an increase of 20 ms or more in the conduction time as a result of premature stimulation. The atrial CD zone was defined as the range of S1-S2 intervals that resulted in the atrial CD. When the atrial CD was not induced, the CD zone was expressed as zero. The maximum CD was defined as the maximum S2-A2 interval (CT2) minus the S1-A1 interval (CT1) achieved at the distal coronary sinus Figure 1.
Results are expressed as the mean values ± standard deviation. Statistical significance was determined using the Student’s t test for unmatched pairs. A p value of less than 0.05 was considered significant.
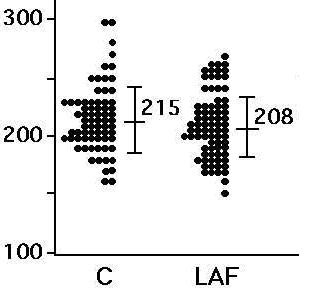
The mean atrial ERP of the control group (215±29 ms) was significantly longer than that of the LAF group (208±28 ms, p<0.05) Figure 2.
Figure 2. The Mean Atrial ERP in controls, and in patients with lone PAF. The mean atrial ERP of the control group (215±29 ms) was significantly longer than that of the LAF group (208±28 ms, p<0.05) C indicates control subjects; LAF, patients with lone PAF
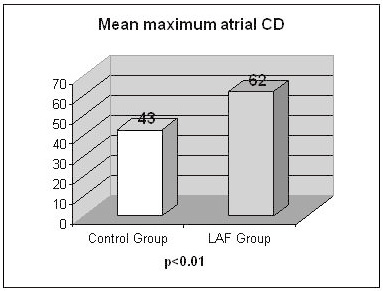
The mean atrial CD zone of LAF group (50±28 msec) was significantly greater than that of controls (34±22 ms) (p<0.01) Figure 3. The mean maximum atrial CD of LAF group (62±29 ms) was also significantly greater than that of controls (43±20 msec) (p<0.01) Figure 4.
Figure 3. The Mean Atrial CD Zone in controls, and in patients with lone PAF. The mean atrial CD zone of LAF group (50±28 ms) was significantly greater than that of controls (34±22 ms) (p<0.01)
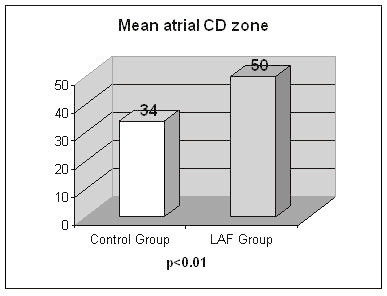
Figure 4. The Mean Maximum Atrial CD in controls, and in patients with lone PAF. The mean maximum atrial CD of LAF group (62±29 ms) was also significantly greater than that of controls (43±20 ms) (p<0.01)
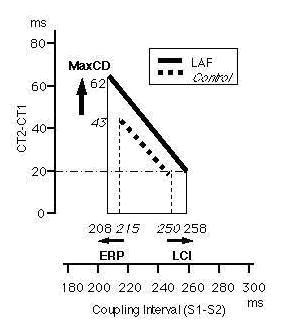
The average relation curve between the coupling interval (S1-S2) and the atrial CD (CT2-CT1) in both groups is shown in Figure 5. The longest coupling interval giving rise to atrial CD (LCI), which was the outer limit of the atrial CD zone, could be calculated as the atrial ERP plus the CD zone. The mean LCI was 258 ms in LAF group, and 250 ms in controls.
Figure 5. The average relation curve between the coupling interval and the atrial CD. The average relation curves between the coupling interval (S1-S2) and the atrial CD (CT2-CT1) of the Control Group, and of the LAF group (A) are compared. The longest coupling interval giving rise to atrial CD (LCI), which was the outer limit of the CD zone, could be calculated as the atrial ERP plus the CD zone
In general, arrhythmias are generated by the presence of substrates, triggers, or modifying factors for arrhythmias. Since the beginning of this century, the debate on ectopic foci versus reentry as the mechanism underlying AF in humans has continued. Recently, the accentuation of focal activity originating mainly from the pulmonary veins, or the superior vena cava, or the ligament of Marshall, is considered to play an important role in the development of AF.14-20 Although, a reentrant activation sequence of multiple wavelets has been demonstrated in human AF by epicardial mapping just before the surgical treatment of this arrhythmia,21 this method is not suitable, nor feasible for diagnostic or investigational purposes in the clinical electrophysiologic laboratory. In addition, once AF is induced in the laboratory, electrical stimulation by a catheter electrode cannot convert it to sinus rhythm. Pharmacologic or electrical cardioversion is required to restore sinus rhythm.
However, these interventions make it impossible to repeat the baseline study for the mechanism of AF. Therefore, clinical electrophysiologic studies have focused mainly on the electrophysiologic properties of the atrium during sinus rhythm and on the atrial electrical responses elicited by the premature stimulation method. Using this technique, the major findings of the present study were as follows. Compared with controls, the LAF group showed a greater atrial CD, and a shortened atrial ERP. The conduction velocity and ERP are the determinants in the genesis of reentrant arrhythmias according to the wavelength theory.22,23 The wavelength is calculated by multiplying the refractory period by the conduction velocity, and is defined as the distance traveled by the depolarization wave during the duration of the refractory period. If the atrial wavelength is long, reentry may not be maintained and AF may terminate spontaneously. On the other hand, if the atrial wavelength is relatively short because of either a short refractory period, or depressed conduction, or both, then a greater number of wavefronts can circulate through the atrium and AF may be sustained. Thus, prolonged atrial CD and/or shortened ERP could be expected to increase the propensity to develop AF. In the LAF group, the greater atrial CD and shorter atrial ERP probably resulted in shortening of the atrial wavelength, which predisposed the development of AF.
The slowing of intra-atrial conduction is considered to be one of the most important requirements for the initiation of reentry and, thus, for AF to develop.24-26 An intra-atrial conduction delay, measured from the stimulus artifact to the atrial electrogram at the distal coronary sinus level, reflects an actual intra-atrial conduction delay that is not influenced by local latency at the site of stimulation.27-29 The conduction velocity of a premature atrial depolarization decreases when it encounters a period of incomplete recovery of excitability. This slowing in conduction may set the background for reentry to occur. Although an increase of 20 ms or more in the atrial conduction time in response to early extrastimulus appears to be a physiological response of the normal atrium, patients with non-idiopathic PAF show longer atrial CD zones and maximum atrial CD than control subjects without atrial arrhythmias.27-29 Thus, the atrial CD zone and maximum CD are believed to be good indices of a tendency to develop AF. We showed in this study that these indices were also significantly greater in patients with lone PAF than in control subjects.
Ramanna et al.30 demonstrated that patients with idiopathic AF have increased spatial dispersion of atrial refractoriness unrelated to electrical remodeling. Of interest, all their 18 study patients with PAF had accessory pathways. Due to the well known association between AF and the Wolff-Parkinson-White syndrome, we excluded patients with accessory pathways from our study. Although we did not investigate dispersion of atrial refractoriness, we demonstrated that the mean atrial ERP in the LAF group was significantly shorter than that in the control group, suggesting that a shortening of the atrial ERP is an important feature in patients with lone PAF. In clinical studies, the duration of the monophasic action potenpotential has been suggested to predict recurrence of AF after electrical cardioversion. Patients with persistently short monophasic action potential duration during sinus rhythm had a higher incidence of recurrent AF than patients with normal action potential duration.31 Therefore, any condition which shortens atrial ERP and action potential duration can be arrhythmogenic. Thus, a shortening of the atrial ERP is expected to increase the propensity for AF in patients with lone PAF, who may have no organic substrate in the atrium. Because the atrial ERP was shorter in the LAF group than in controls, earlier atrial premature beats with coupling intervals close to their shortened atrial ERP could facilitate atrial CD predisposing the development of intraatrial reentry and, thus, AF.
There are certain limitations that should be considered. First, only the right atrial ERP was measured, and therefore the responses of other areas of the atrium to pacing-induced CD are unknown. Second, aging should be considered as a contributing factor to the propensity for AF.32-34 However, there was no significant difference in the mean age between the 2 groups. Third, the control subjects in the present study were not really “normal” since they all suffered from some form of arrhythmia. However, this is a common limitation in electrphysiological studies, since nobody will perform an invasive EP study to a patient that has no arrhythmia. Fourth, it is well established that asymptomatic episodes are common in patients with AF,35 and clinical evaluation based on the presence or absence of documented AF may be of limited value. However, these are common limitations in most clinical electrophysiological studies. Although limited for these reasons, we demonstrated that patients with idiopathic PAF exhibit electrophysiological changes of the atrium that predispose them to develop AF. In conclusion, in this study, we demonstrated that there is a greater conduction delay of the atrium and shorter atrial refractoriness in patients with lone PAF. Patients without underlying causes for the development of PAF exhibit abnormalities in the electrophysiological properties of the atrium. An understanding of these electrophysiological abnormalities in patients with lone PAF is important when choosing effective antiarrhythmic drugs. Selecting an adequate antiarrhythmic drug should be based on the electrophysiological properties of the arrhythmia in question. We hope that our results will help in determining effective therapies for lone paroxysmal AF.