A Rare Case of Bronchopericardial Fistula Following Atrial Fibrillation Ablation
Ghulam Murtaza2, Daisy Young 1, Krishna Akella 2, Rakesh Gopinathannair2, Dhanunjaya Lakkireddy2, Alap Shah2
1Internal Medicine Resident at Stony Brook Southampton Hospital, New York.2Kansas City Heart Rhythm Institute, Overland Park, KS.
Radiofrequency ablation (RFA) for atrial fibrillation (AF) has emerged as an effective and reliable treatment modality. Since its introduction in the 1990s, major and minor complications have been identified. Major complications include periprocedural death, atrioesophageal (AE) fistula, stroke, cardiac perforation and tamponade, pulmonary venous stenosis, phrenic nerve injury, retroperitoneal hematoma, and arrhythmias. Minor complications include pseudoaneurysms and arteriovenous fistulas . We report an extremely rare and life-threatening complication of bronchopericardial fistula following AF ablation resulting in respiratory complications.
Key Words : Radiofrequency ablation (RFA), Atrial fibrillation (AF), Atrioesophageal (AE).
Correspondence to: Ghulam Murtaza
Radiofrequency ablation (RFA) for atrial fibrillation (AF) has emerged as an effective and reliable treatment modality. Since its introduction in the 1990s, major and minor complications have been identified. Major complications include periprocedural death, atrioesophageal (AE) fistula, stroke, cardiac perforation and tamponade, pulmonary venous stenosis, phrenic nerve injury, retroperitoneal hematoma, and arrhythmias. Minor complications include pseudoaneurysms and arteriovenous fistulas [1-3]. We report an extremely rare and life-threatening complication of bronchopericardial fistula following AF ablation resulting in respiratory complications.
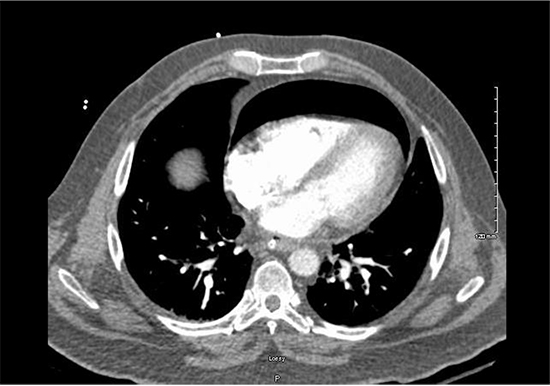
A 51-year-old male with a past medical history of symptomatic paroxysmal atrial fibrillation (PAF), hypertension, hyperlipidemia, obesity, depression, and obstructive sleep apnea presented with complaints of fatigue and palpitations. Holter monitor documented a 50% AF burden. Transthoracic echocardiogram revealed normal left ventricular function and LA diameter of 4.5cm. Patient was started on flecainide and referred to an electrophysiologist for continued fatigue and PAF. Ablation was recommended but patient preferred medical therapy. He was admitted for dofetilide. After the 4th dose, QT became prolonged. He was started on amiodarone and underwent RFA. 2 weeks after the ablation, patient endorsed non-productive cough without fever and chills. Chest x-ray was obtained and showed pneumopericardium [Figure 1]. Suspicion for AE fistula was high and patient was started on broad spectrum antibiotics. Patient required intubation for chest CT due to hypoxia upon lying supine. CT chest showed pneumopericardium [Figure 2]. CT surgery was consulted and pericardial window was performed to evacuate the pneumopericardium. EGD revealed no evidence of esophageal injury or fistulous connection. Subsequent bronchoscopy revealed distal left mainstem bronchus ulceration which presumably led to a bronchopericardial fistula. Patient had a prolonged course in the ICU with supportive care and serial bronchoscopies. Tracheostomy was performed and patient was eventually extubated after 10 days. Patient returned to baseline with rehab over the next 3 months and amiodarone was eventually stopped.
Figure 1. Chest x-ray revealing pneumopericardium most evident surrounding the left heart structures.
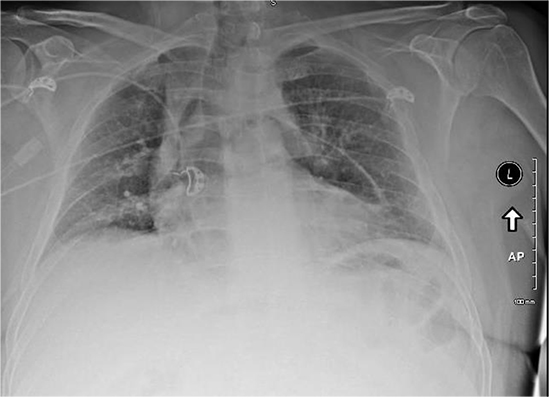
Figure 2. CT chest with large pneumopericardium noted in the anterior chest.
RFA procedures involve a 3-dimensional mapping system to reconstruct the left atrium and pulmonary veins. A catheter delivers radiofrequency energy in a circumferential fashion 1 to 2 cm from the left- and right-sided pulmonary veins [4]. Though regarded as a safe procedure, complications may arise. Our patient suffered a very rare complication: bronchopericardial fistula and pneumopericardium.
Bronchopericardial fistulas involve a communication between the tracheobronchial tree and the pericardial sac. Air enters the pericardium, resulting in a pneumopericardium and this may lead to cardiovascular collapse secondary to cardiac air tamponade [5]. A small pneumopericardium may present as cough, whereas a larger pneumopericardium may present with shortness of breath and chest pain. These fistulas have been identified as complications from bronchogenic carcinoma, necrotizing pulmonary infections such as tuberculosis and invasive pulmonary aspergillosis, penetrating chest trauma, and iatrogenic causes such as coronary artery bypass grafts and transbronchial biopsies [6]. It is rarely seen as a complication from RFA and has been reported in fewer than 5 cases. Given its low incidence, the pathophysiology is not fully understood.
However, the pathophysiology can be derived from an understanding of AE fistulas, which are well-documented as a major complication of RFA with high mortality. The pathophysiology of AE fistulas arise from direct thermal injury to the esophagus, excessive catheter tip contact force, and extended RFA energy duration in the setting of a thin left atrial wall. Oftentimes, thermal injury occurs where ablation lines overlap. The injury creates a fistulous communication between the esophagus and left atrium, which are directly adjacent anatomically [7].
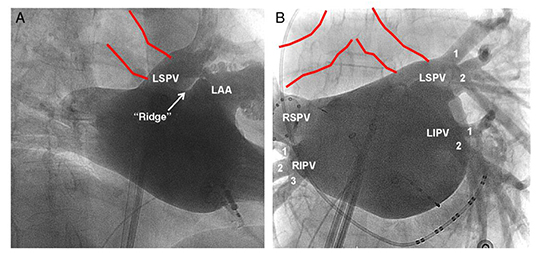
Similarly, the left atrium and pulmonary veins lie in close proximity to neighboring anatomic structures such as the aorta, left main bronchus, and phrenic nerves [Figure 3] and [Figure 4]. In our patient, RFA likely produced indirect thermal injury and ischemic necrosis to the left main bronchus and left atrium. The added risk factors of likely thin and dilated left atrial wall secondary to high AF burden and dilated pulmonary vasculature secondary to obstructive sleep apnea and obesity may have also contributed.
Figure 3. Anatomic depiction of the close proximity between the bronchial tree and pulmonary venous structures.
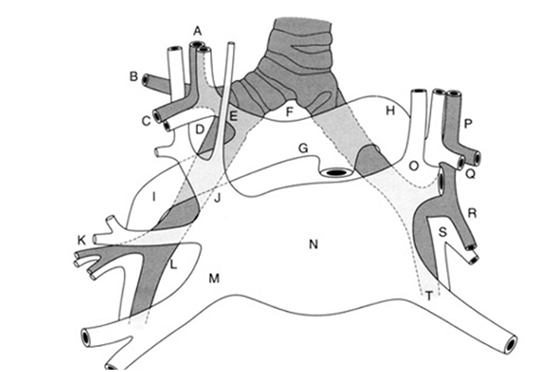
Figure 4. A) Angiographic depiction of the proximity between the left main bronchus (red lines) and the left superior pulmonary vein, which is the focus of radiofrequency ablation. B) Angiographic depiction of the right and left main stem bronchi in relation to the pulmonary veins.
Though respiratory symptoms can be encountered after cryoballoon ablation (CBA) for AF, these symptoms are not commonly seen following RFA [8] . A study by Verma et al in which 10 patients underwent CBA with real-time bronchoscopy showed that ice formation in the left mainstem bronchus was seen in 70% of patients and in 59% of lesions. No ice formation was seen on the right side [9]. This can explain the cough and hemoptysis in some patients after CBA.
Perhaps early intervention via bronchoscopy needs to be considered in patients who present with respiratory symptoms after AF ablation. In the event of pneumopericardium, early intervention and evacuation of the pericardial space is paramount. Whether obstructive sleep apnea and/or pulmonary hypertension are risk factors for RFA fistulous complications is unknown.
The left mainstem bronchus is in close proximity to the left superior pulmonary vein and evaluation of bronchial injury or pneumopericardium should be considered in patients presenting with cough and/or hemoptysis post AF RFA.