Left Atrial Volume Index Predicts Arrhythmia-Free Survival in Patients with Persistent Atrial Fibrillation Undergoing Cryoballoon Ablation
Alfred J Albano1, Jared Bush 1, Jessica L Parker 1, Kristin Corner1, Hae W Lim 2, Michael P Brunner1, Musa I Dahu1, Sanjay Dandamudi 1, Darryl Elmouchi 1, Andre Gauri 1, Alan Woelfel 1, Nagib T Chalfoun 1
1Department of Cardiology, Spectrum Health Heart and Vascular, Grand Rapids, Michigan .2Medtronic, Inc., Minneapolis, Minnesota .
Pulmonary vein isolation (PVI) using cryoballoon ablation (PVI-C) is increasingly performed as a first-line strategy for the treatment of patients with persistent atrial fibrillation (PersAF); however, follow-up data and predictors of procedural success are lacking.
To study the efficacy of PVI-C in patients with PersAF, focusing on predictors of procedural success.
By retrospective review, 148 consecutive patients with PersAF who underwent PVI-C were analyzed. The impact of several variables on outcome was evaluated in univariate and multivariate analyses and Cox proportional hazards regression models.
After a mean follow-up of 19.2±10.9 months, 75 (50.7%) patients remained arrhythmia-free without the need for antiarrhythmic drug therapy. Patients with a normal left atrial volume index (LAVI) achieved a 71.0% arrhythmia-free survival. LAVI was the most powerful predictor of procedural success.
Arrhythmia-free survival after PVI-C in select patients with PersAF are promising. Moreover, LAVI is a valuable measurement to help guide ablation strategy and predict outcome when using cryoballoon ablation.
Key Words : Catheter ablation, Cryoablation, Cryoballoon, Left atrial volume index, Persistent atrial fibrillation, Pulmonary vein isolation.
Correspondence to: Alfred J Albano, M.D.
Department of Cardiology, Spectrum Health Heart and Vascular
100 Michigan St NE
Grand Rapids, MI 49503
Pulmonary vein isolation (PVI) by catheter ablation is a well-established technique for the treatment of patients with persistent atrial fibrillation (PersAF).[1] Although the ideal treatment strategy for patients with PersAF remains incompletely defined, a PVI-only approach by cryoballoon ablation has been demonstrated to be an effective and reasonable therapy as a stand-alone treatment for patients with PersAF during the index procedure.[2-7]
Also, cryoballoon ablation is non-inferior to radiofrequency current catheter ablation, and a PVI-only strategy has been found to be non-inferior to PVI plus the addition of linear and complex fractionated atrial electrogram ablation in patients with atrial fibrillation (AF).[8-10]
Most available data on PVI using cryoballoon ablation (PVI-C) in patients with PersAF involve small studies, short follow-up duration, and limited post-procedure monitoring. The focus of this retrospective analysis is to study the efficacy of PVI-C in a larger patient population with PersAF over a longer follow-up period. Specifically, we sought to identify predictors of procedural success that might guide treatment approach and ablation strategy.
Consecutive patients with symptomatic PersAF (refractory to medical therapy) who underwent PVI-C (Arctic Front Advance; Medtronic, Inc.) between March 2013 and January 2015 at the Meijer Heart Center (Spectrum Health Heart and Vascular, Grand Rapids, MI) were retrospectively analyzed by two independent reviewers. All data was obtained by review of the electronic medical record, and any incongruent data was appropriately adjudicated by the two independent reviewers. Approval for the study was granted by Spectrum Health’s institutional review board. Patients who had undergone prior catheter ablation and/or had less than 3 months of follow-up post-procedure were excluded from the analysis.
PersAF was defined as AF lasting continuously for greater than 7 days but less than one year.[1] Patients with longstanding persistent AF lasting continuously for greater than one year were excluded. All patients were refractory and/or intolerant to at least one antiarrhythmic drug (AAD) or in normal sinus rhythm on amiodarone prior to catheter ablation. Amiodarone failure was defined as recurrence of AF requiring cardioversion or catheter ablation following a standard loading period (typically 400mg twice daily for two weeks), or adverse side effects necessitating discontinuation. Demographic data and baseline characteristics were collected on all 148 patients [Table 1].
Table 1. Baseline patient characteristics (N=148).
| Characteristic |
Value |
| Age (y) |
62.2±9.1 |
| Sex: male |
106 (71.6%) |
| Body mass index (kg/m2) |
32.8±6.2 |
| CHA2DS2-VASc score |
2.0±1.3 |
| AF duration (months) |
37.4±44.9 |
| Persistent AF duration (months)
|
8.2±14.9 |
| Number of cardioversions prior to ablation
|
2.1±1.1 |
| Amiodarone failure
|
64 (43.0%) |
| Co-morbid conditions |
| Hypertension
|
110 (74.3%) |
| Left ventricular hypertrophy
|
15 (10.2%) |
| Type II diabetes
|
31 (21.1%) |
| Coronary artery disease
|
42 (28.4%) |
| Prior myocardial infarction
|
11 (7.4%) |
| Prior percutaneous coronary intervention
|
11 (7.4%) |
| Prior coronary artery bypass surgery
|
8 (5.4%) |
| Congestive heart failure
|
31 (21.0%) |
| Chronic kidney disease
|
21 (14.3%) |
| Chronic obstructive pulmonary disease
|
16 (10.8%) |
| Obstructive sleep apnea
|
72 (48.7%) |
| CPAP* compliance (N=72)
|
52 (72.2%) |
| Echocardiographic parameters |
| Left ventricular ejection fraction
|
55.2±6.7 |
| Left atrial diameter (cm; N=141)
|
4.4±0.5 |
| Left atrial volume index (ml/m2; N=135)
|
36.5±8.1 |
* CPAP= continuous positive airway pressure
Cryoballoon Ablation Procedure
All patients underwent catheter ablation at Spectrum Health Heart and Vascular (Grand Rapids, MI) where more than 400 cryoballoon ablation procedures are performed annually. PVI was performed by a total of five board-certified cardiac electrophysiologists using standard protocols.[11] In brief description, all patients underwent a pre-operative transesophageal echocardiogram to exclude left atrial (LA) appendage thrombus, and all subjects were treated under general anesthesia. Groin access was obtained using ultrasound guidance in all patients. Immediately following groin access, a heparin bolus was delivered in advance of the transseptal puncture, and activated clotting time was monitored every twenty minutes with a goal of 350-400 sec. Initial transseptal puncture was performed using either a BRK or Baylis RF needle (Baylis Medical) depending on the operator. An electrophysiology study was completed in all patients prior to ablation.
An over-the-wire method was used to deliver the cryoballoon and sheath (FlexCath; Medtronic, Inc.) into the left atrium. All patients were treated with the 28-mm cryoballoon and 20-mm Achieve mapping catheter. The number of freeze applications per PV was operator-dependent, but most patients received two (180-second) freeze applications. It was at the operator’s discretion to perform a single-freeze application based on time-to-isolation, temperature achieved at 60 sec, and/or cryoballoon thaw times. Esophageal temperature monitoring was performed in all cases. Phrenic pacing and monitoring were performed during ablation over the right-sided PVs. Cardiac mapping with EnSite (St Jude Medical) was used in most cases to guide diagnostic catheters, and evaluate post-procedural LA electrophysiology by voltage mapping. PV entrance and exit block were proven in all patients.
All patients underwent transthoracic echocardiography prior to PVI-C to measure LA dimensions, to assess for the presence of significant valvular heart disease, and to record left ventricular ejection fraction. If these studies were performed by a referring hospital, then the images from the study were obtained for review. All echocardiography studies were reviewed and interpreted by a single board-certified echocardiographer. The LA diameter was measured in the long-axis parasternal view in the anterior-posterior dimension at end-ventricular systole.
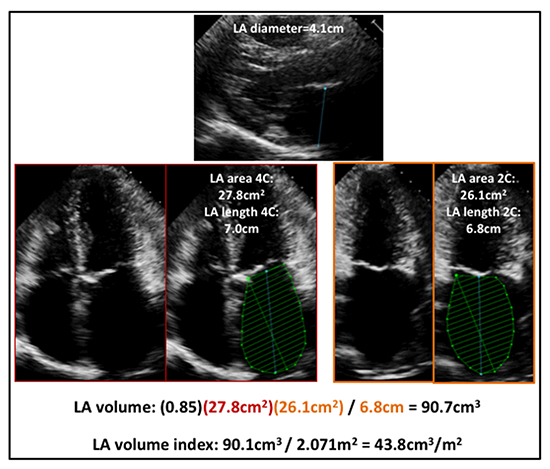
The area-length method was used to calculate LA volume index (LAVI).[12] All measurements were performed in the standard apical 4-chamber (A4C) and 2-chamber (A2C) views at end-ventricular systole. LA volume was calculated using the equation (0.85 × A1 × A2)/L, where A1 was the planimetered area measured in the A4C view, A2 was the planimetered area measured in the A2C view, and L was the shortest length measured in A4C and A2C from the back wall of the atrium to a line across the mitral valve hinge points. The LAVI was then derived by dividing LA volume by body surface area. This calculation is demonstrated in [Figure 1]. LA size was categorized using standard criteria.[12] Specifically, LA sizes were grouped into four separate cohorts, including: normal (<34 ml/m2), mildly enlarged (34-41 ml/m2), moderately enlarged (42-48 ml/m2), and severely enlarged (>48 ml/m2). Repeat echocardiography was performed in the event of symptoms such as hypotension, tachycardia, chest pain, or shortness of breath.
Figure 1. Sample left atrial volume measurement in a patient with a moderately dilated left atrium. Note that atrial size is underappreciated by left atrial diameter.
Catheter ablation procedure-related adverse events were categorized into major, moderate, and minor complications. Major complications included stroke or transient ischemic attack, myocardial infarction, pericardial tamponade, PV stenosis, emergency cardiac surgery, resuscitation, or atrioesophageal fistula. Moderate complications included hematoma requiring transfusion (major bleeding), femoral arteriovenous fistula, pseudoaneurysm, or persistent phrenic nerve paresis (PNP). Specifically, persistent PNP was defined as any PNP that remained present at the end of the ablation procedure. Minor complications including small hematoma (not requiring transfusion) and transient PNP were not quantified in this study. Transient PNP was defined as any PNP that resolved prior to the end of the ablation procedure.
Data Collection and Follow-up
Routine follow-up assessments were conducted in accordance with a standard protocol at our center. All follow-up clinic visits were scheduled at 3 months and 12 months post-procedure, then annually if the patient remained free of arrhythmias and symptoms. Each follow-up clinic visit focused on AF-related symptoms to determine arrhythmia recurrence. A 14-day patch monitor was routinely ordered at 6- and 12-months post-procedure in all patients for detection of any occult AF, and as needed thereafter to assess symptoms.
The primary efficacy endpoint for this study was defined as the freedom from atrial arrhythmia (AF, atrial flutter, and/or atrial tachycardia) outside of a landmark 90-day blanking period following the index cryoballoon ablation procedure with no usage of AADs during the 12-month follow-up period. Within the blanking period, no early recurrence of atrial arrhythmia events were counted against the long-term clinical outcome. Patients who achieved complete success had no detected atrial arrhythmia recurrence (> 30 sec) or symptoms of AF post-ablation and did not require AAD therapy beyond the blanking period. Patients who had confirmed detection of a recurrence of atrial arrhythmia (>30 sec) after the blanking period, required AADs, underwent repeat catheter ablation, and/or degenerated to permanent AF with adoption of a rate control strategy (because of symptom resolution in AF post-ablation) were deemed procedural failures.
Descriptive statistics were used to summarize patient characteristics, procedural characteristics, safety, and follow-up. Numeric variables are shown as mean ± standard deviation. Categorical variables are shown as count with frequency (%). All arrhythmia recurrence graphs were completed using a Kaplan-Meier estimate method. Univariate Cox regression models were used to determine what variables were significant by themselves in predicting arrhythmia recurrence. A significance level of P<0.10 was used for the first round of cuts. After those cuts were made the variables that had a p-value less than 0.10 were included in a multivariate Cox regression model to determine the final predictors of arrhythmia recurrence. To determine the final predictors, backward selection was used with a significance level of P<0.05. Fishers Exact test or one-way ANOVA were used to further examine demographic characteristics after multivariable analysis. All statistical analyses were generated using SAS (SAS Enterprise Guide software, Version 7.1, SAS Institute Inc, Cary, NC).
From August 2013 to November 2015, a total of 200 consecutive subjects with PersAF underwent a PVI-C. Forty-nine patients had previously undergone radiofrequency catheter ablation and were excluded from this analysis. One patient required an emergent coronary artery bypass graft within three months of PVI-C, underwent a concomitant Maze procedure, and was also excluded. Two patients did not have adequate follow-up of at least 6 months post-procedure. Finally, 148 patients were included in this analysis. Clinical outcomes were determined by retrospective chart analysis performed by two independent reviewers with initial agreement in 138/148 (93.2%) of cases. The ten cases with conflicting outcome data were appropriately adjudicated by the study authors.
Baseline patient characteristics are summarized in [Table 1]. The mean age was 62.2±9.1 years, and 28.4% of the subjects were women. Mean BMI was 32.8±6.2 kg/m2, and 43.0% of patients failed amiodarone therapy prior to ablation either because of ineffectiveness or side effects. The time from the diagnosis of paroxysmal and PersAF to catheter ablation was 37.4 months and 8.2 months, respectively. On average, patients had two prior direct-current cardioversions before the index PVI-C.
Procedural Data and Complications
Mean procedure time was 112.2±42 minutes, and the mean fluoroscopy time was 15.8±11.1 minutes. Acute PVI was achieved in 100% of the cases. Ten patients (6.8%) required adjunctive radiofrequency catheter ablation performed on a total of eleven PVs to achieve complete electrical PVI. In those aforementioned cases, the adjunctive radiofrequency ablations were required over the inferior aspect of the right inferior PV (4 patients), inferior aspect of the left inferior PV (3 patients), anterior aspect of the right superior PV (3 patients), and anterior aspect of the left inferior PV (1 patient). The procedural data are shown in [Table 2], including cryoballoon nadir temperatures during cryoballoon ablation. There were no major complications, and moderate complications occurred in five patients [Table 3]. Three patients experienced PNP (lasting beyond the end of the procedure), all of which completely resolved within 6 months. One patient developed a hematoma, and one patient had a femoral pseudoaneurysm.
Table 2. Cryoballoon ablation procedural characteristics (N=148).
| Characteristic |
Value |
| Procedural time (min)
|
112.2±42 |
| Fluoroscopy time (min)
|
15.8±11.1 |
| Cryoablation time (min)
|
27.4±7.0 |
| Acute PVI* success rate (treated veins/target veins)
|
585/585 (100%) |
| Adjunctive radiofrequency ablation for PVI (patients)
|
10 (6.8%) |
| Nadir cryoballoon temperatures (oC)
|
|
| Left superior pulmonary vein (N=140)
|
-48.3±5.9 |
| Left inferior pulmonary vein (N=140)
|
-44.6±4.7 |
| Right superior pulmonary vein (N=148)
|
-46.9±13.8 |
| Right inferior pulmonary vein (N=148)
|
-45.2±6.1 |
| Left common pulmonary vein (N=8)
|
-41.1±8.4 |
* PVI= pulmonary vein isolation
Table 3. Procedural complications (N=148).
| Characteristic
|
N (%) |
| Moderate complications (Total)
|
5 (3.4) |
| Persistent diaphragmatic paralysis
|
3 (2.0) |
| Hematoma
|
1 (0.7) |
| Femoral pseudoaneurysm
|
1 (0.7) |
| Arteriovenous fistula
|
0 (0.0) |
| Major complications (Total)
|
0 (0) |
| Stroke/transient ischemic attack
|
0 (0.0) |
| Pericardial tamponade/effusion
|
0 (0.0) |
| Pneumothorax/hemothorax
|
0 (0.0) |
| Pulmonary vein stenosis
|
0 (0.0) |
Echocardiographic Characteristics
Mean LA diameter measured 4.4±0.5cm, and the mean LAVI measured 36.5±8.1 ml/m2 [Table 1]. Raw echocardiography data for review and analysis was available on 135 (91.2%) patients, and LA dimensions could not be measured and confirmed in 13 (8.8%) patients. Using LAVI, 62 (45.9%) patients had a normal LA size (29.8±3.1 ml/m2), 42 (31.1%) patients had a mildly enlarged LA (37.5±2.3 ml/m2), 18 (13.3%) patients had a moderately enlarged LA (44.7±1.4 ml/m2), and 13 (9.6%) patients had a severely enlarged LA (53.5±3.4 ml/m2).
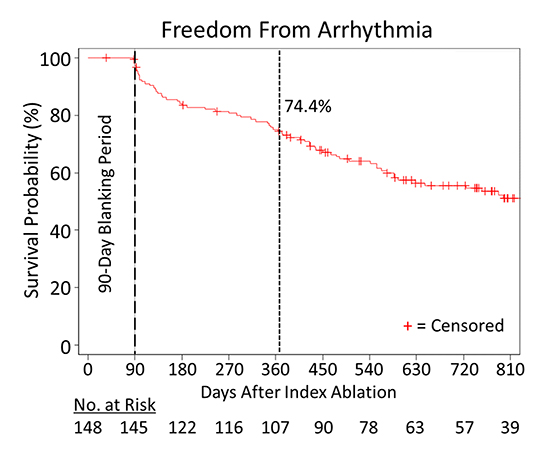
One-year complete arrhythmia-free success rate was 74.4% (95% CI, 66.4-80.7%). After a mean follow-up of 19.2±10.9 months, 75 patients (50.7%) patients remained in sinus rhythm without any evidence of AF recurrence off AAD therapy [Figure 2].
Figure 2. Arrhythmia-free survival without antiarrhythmic drug therapy following pulmonary vein isolation by cryoballoon ablation.
Post-Procedure Electrocardiographic Monitoring
Follow-up clinic visit with accompanying ECG was completed by 98.7% of patients (146/148). During follow-up, 126 (85.1%) patients were evaluated with some form of long-term monitoring, and 104 (70.3%) patients received a 14-day patch monitor. Typically, 14-day monitors were placed at 6- and 12-months post-procedure. Nine patients (6.1%) were fitted with a 30-day event monitor, six patients (4.1%) were monitored with an implantable loop recorder, and eleven patients (7.4%) had pacemakers. These data are summarized in [Table 4].
Table 4. Post-procedure follow-up and monitoring (N=148).
| Characteristic |
N (%) |
| Follow-up clinic visit with accompanying ECG
|
146 (98.7) |
| Long-term rhythm monitoring
|
126 (85.1) |
| 14-day patch monitor
|
104 (70.3) |
| 30-day event monitor
|
9 (6.1) |
| Loop recorder
|
6 (4.1) |
| Pacemaker implant
|
11 (7.4) |
Predictors of Arrhythmia Recurrence
LAVI was identified as a primary predictor of arrhythmia recurrence (P=0.003;) [Table 5]. LA diameter and cryoballoon nadir temperature were also predictors of arrhythmia recurrence (P=0.021 and P=0.034; respectively). Multiple covariates (including age, BMI, OSA, valvular heart disease, and prior coronary artery bypass graft) did not predict procedural success [Table 1].
Table 5. Arrhythmia-free survival model
|
Univariate Analysis |
Multivariate Analysis |
| Covariate |
Coefficient (bi) |
HR [exp(bi)] |
95% CI |
P-value |
Coefficient (bi) |
HR [exp(bi)] |
95% CI |
P-value |
| Number of pre-procedure electrical cardioversions |
0.17 |
1.18 |
(0.98-1.44) |
0.086 |
|
|
|
0.114 |
| Nadir LSPV* balloon temperature |
0.04 |
1.04 |
(1.00-1.08) |
0.056 |
0.04 |
1.05 |
(1.00-1.09) |
0.034 |
| Left atrial AP diameter |
0.05 |
1.05 |
(1.01-1.10) |
0.026 |
-0.07 |
0.93 |
(0.88-0.99) |
0.021 |
| Hypertension |
0.56 |
1.75 |
(0.94-3.26) |
0.076 |
|
|
|
0.860 |
| Left atrial volume index |
|
|
|
<0.001 |
|
|
|
0.003 |
| 1-Normal |
(0.00) |
(1.00) |
|
|
(0.00) |
(1.00) |
|
|
| 2-Mild |
1.10 |
3.00 |
(1.65-5.45) |
|
1.13 |
3.11 |
(1.62-5.95) |
|
| 3-Moderate |
0.76 |
2.14 |
(1.00-4.57) |
|
0.88 |
2.41 |
(0.89-6.51) |
|
| 4-Severe |
1.96 |
7.07 |
(3.36-14.89) |
|
1.58 |
4.86 |
(1.69-13.98) |
|
*LSPV= left superior pulmonary vein
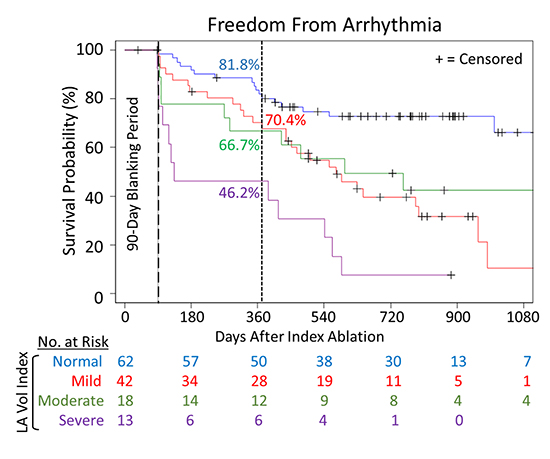
Patients with a normal LAVI achieved 71.0% arrhythmia-free survival during the long-term follow-up period. Patients with mildly and moderately enlarged atria achieved 33.3% and 38.9% arrhythmia-free survival, respectively. Patients with a severely dilated LA (by LAVI) only achieved 7.6% arrhythmia-free survival. These results (including one-year success rates) are displayed in [Figure 3]. Further evaluation of all patient baseline characteristics by LAVI sub-groups uncovered five unique interactions [Table 6]. Patients with severe LA dilation (by LAVI) were generally older in age compared to the normal, mild, and moderate sub-groups of patients (P=0.054). Also, patients with normal LAVI had a lower baseline incidence rate of hypertension, prior coronary artery bypass graft, and/or valvular heart disease (P=0.030, 0.010, and 0.013, respectively). Finally, LA diameters (A/P) were progressively larger from normal to severe LAVI, as expected (P<0.001); however, LAVI remained the most predictive baseline characteristic regarding long-term arrhythmia-free survival.
Table 6. Demographic analysis by left atrial volume index subcategories
|
Left atrial volume index |
|
| Variables |
Normal (N=62) |
Mild (N=42) |
Moderate (N=18)
|
Severe (N=13)
|
P-value |
| Age |
60.6±9.6 |
62.2±8.8 |
61.9±7.1 |
68.3±10.4 |
0.054 |
| Hypertension |
38 (61.3%) |
35 (83.3%) |
16 (88.9%) |
10 (76.9%) |
0.030 |
| Left atrial A/P diameter |
40.7±3.9 |
44.2±4.5 |
48.6±3.8 |
49.8±5.3 |
<0.001 |
| Prior CABG |
0 (0%) |
4 (9.5%) |
2 (11.1%) |
2 (15.4%) |
0.010 |
| Valvular heart disease |
3 (4.8%) |
5 (11.9%) |
6 (33.3%) |
1 (7.7%) |
0.013 |
* CABG= coronary artery bypass
Figure 3. Examination of sub-groups (by left atrial volume index) from normal to severe left atrial enlargement. Arrhythmia-free survival without antiarrhythmic drug therapy following pulmonary vein isolation by cryoballoon ablation.
Our study has two important clinical findings. First, PVI-C used in the treatment of patients with PersAF is safe and effective, which is in agreement with other large multicenter real-world observations.[4-8] Second, the data demonstrated that LAVI is a strong predictor of PVI-C procedural success in patients with PersAF. Previous associations have only been described in paroxysmal AF populations.[13-14]
LAVI as a Predictor of Procedural Outcome
It is established that LA volume predicts AF-free survival during radiofrequency catheter ablation,[15-16] and similarly, predicts response in patients with paroxysmal AF treated by PVI-C.[13-14] Yet, LAVI remains unstudied in a large cohort of PersAF patients treated by PVI-C. In fact, many studies of cryoballoon ablation in the treatment of AF fail to measure LAVI, but instead only report linear LA dimension.[2,4-8] Similar to our findings, LA diameter (A/P) has not been shown to be a reliable or strong predictor of procedural success in most studies. [2,4-8] However, the pitfalls of using LA diameter as a surrogate for LA size have been appreciated for several years.[17] It is known that atrial enlargement can occur predominately in the superior-inferior dimension, with relative preservation of the anterior-posterior dimension. This is illustrated in [Figure 1], where a LA diameter measurement underestimates the true LA size. LA volume has consistently outperformed both LA diameter and area in the prediction of cardiovascular outcomes including AF, stroke, TIA, myocardial infarction, heart failure, and death.[17]
LA enlargement is often a consequence of increased atrial pressure and hemodynamic overload, triggering stretch receptors with release of angiotensin and TGF-β, and activation of fibroblasts leading to collagen deposition.[18] Atrial fibrotic burden has been correlated with arrhythmia recurrence following ablation.[19] Although the dominant paradigm has been that all patients with PersAF have more advanced atrial remodeling and should undergo substrate modification in addition to PVI, our data suggests that LA size as determined by LAVI may be more important than AF type.
In our analysis, patients with PersAF and normal LAVI were observed to have success rates similar to patients with paroxysmal AF undergoing PVI with the second-generation cryoballoon. Chierchia [20] and Furnkranz [21] reported one-year arrhythmia free survival in 83% and 84% of patients, respectively, undergoing cryoballoon ablation for paroxysmal AF. This compares to our one-year success rate of 82% in patients with a normal LAVI. Similarly, the PLAAF score predicts that PersAF is only one of five risk factors for AF recurrence and that a long-term freedom from AF can be predicted in male patients with non-enlarged LA, normal 4-PV anatomy, and who have a shorter history of AF.[13] Based on these findings, it is reasonable to approach initial ablation using a PVI-only strategy in patients with PersAF and a normal LAVI.
For patients with significant atrial enlargement, especially severely dilated LA by volume index, a PVI-only strategy is likely to fail. Although there is not consensus on how to approach non-PV trigger elimination and substrate modification after effective PVI, there are several strategies that have been proposed including posterior wall ablation, superior vena cava and appendage isolation, ligament of Marshall ablation, trigger mapping, and hybrid ablation techniques.[22,23] Our data show that patients with mild-to-moderately dilated LA can still have a favorable outcome with a PVI-only strategy, and in patients with multiple co-morbidities, this strategy may be preferable to a higher-risk ablation procedure with extensive substrate modification.
Moderate-to-severe LAVI was the primary predictor of failure to achieve long-term arrhythmia-free survival. Importantly, in this study, patients with moderate and severe LAVI were older, hypertensive, had prior coronary artery bypass graft, and/or had a history of at least moderate valvular heart disease. Consequently, other baseline characteristics may be of importance when considering an ablation strategy, and usage of scoring systems (e.g., PLAAF) may be an imperative part of the final decision.[13]
Moderate complications occurred in 3.4% of patients and were limited to either persistent phrenic injury or groin complications. We observed PNP in 2% of our patient population (although rates of PNP after PVI with the second-generation cryoballoon can range up to 5.5%).21 Access-site complications occurred in 1.4% of our patients, which is comparable to previously reported rates.[1] There were no major complications, and minor complications were not assessed.
Our study was a single-center, retrospective, observational analysis without a control group. In addition, a large percentage (45.9%) of our patients were found to have a normal LAVI, which may (in part) explain our favorable outcome data. All consecutive patients with PersAF undergoing an index PVI-C were included (reducing but not eliminating selection bias since not all patients with PersAF were offered ablation). We also did not assess the outcome on AAD therapy without ablation.
Our follow-up period is moderate in duration and not necessarily predictive of longer-term outcomes. We also recognize that intermittent rhythm monitoring as used in this study is inferior to continuous monitoring with an implantable loop recorder. However, 85% of our patient population underwent some form of long-term monitoring, mostly using a 14-day patch monitor. This is an improvement over most historical trials which use clinic follow-up and 24-hour Holter monitors for post-ablation arrhythmia surveillance.[1] Also, we did have a 98.7% clinic follow-up rate, and chart analysis performed by two independent reviewers with initial agreement in 138/148 (93.2%) of cases.
LAVI was meticulously calculated by a board-certified echocardiographer. However, LAVI by echocardiography is imperfect, and other techniques such as three-dimensional echocardiography or cardiac CT/MRI may provide a more accurate assessment.
Although one physician operator did perform intermittent single-shot ablation, because of the date range of the collection (2013-2015), only 8 pulmonary veins were treated using this technique. This precluded us from making any meaningful analysis of single vs two-shot ablation. It is now acknowledged that single-shot protocols are widely adopted, and that cryoballoon usage parameters are important towards outcomes. Unfortunately, use parameters were not evaluated in the current study.
Arrhythmia-free survival after PVI-C in selected patients with PersAF are promising. LAVI is a valuable measurement to help guide ablation strategy and predict outcome when utilizing cryoballoon ablation.