Sleep Duration and Risk of Atrial Fibrillation: a Systematic Review
Negar Morovatdar 1, Negar Ebrahimi 2, Ramin Rezaee1, Hoorak Poorzand3, Mohammad Amin Bayat Tork2, Amirhossein Sahebkar4,5,6
1Clinical Research Unit, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran.2Student Research Committee, Mashhad University of Medical Sciences, Mashhad, Iran.3Atherosclerosis Prevention Research Center, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran.4Biotechnology Research Center, Pharmaceutical Technology Institute, Mashhad University of Medical Sciences, Mashhad, Iran.5Neurogenic Inflammation Research Center, Mashhad University of Medical Sciences, Mashhad, Iran.6School of Pharmacy, Mashhad University of Medical Sciences, Mashhad, Iran.
Little is known about a possible association between sleep duration and the incidence of atrial fibrillation (AF), in healthy people. In this systematic review, we conducted a literature search to examine possible association between sleep duration and the incidence of AF.
Scientific databases (PubMed, Web of Knowledge and Embase) were searched using relevant Medical Subject Headings and keywords, to retrieve studies written in English and published until November 2017. Only population-based observational studies were included. Since sleep duration categories were not consistent, it was feasible to run a meta-analysis.
Six eligible studies were included. Long sleep duration (≥ 8 hours) was found to be associated with an increased risk of AF (adjusted hazard ratio (aHR) = 1.13; 95% CI: 1.00-1.27 and aHR= 1.5, 95% CI: 1.07-2.10) in two studies. One study reported that sleep duration less than 6 hours was associated with an increased risk of AF (aHR= 1.58, 95% CI: 1.18 -2.13) compared to sleeping for 6-7 hours. In two studies, mean sleep duration was lower in AF groups compared to the non-AF group. Insomnia was associated with an increased risk of AF in another study (aHR= 1.33, 95% CI: 1.25-1.41).
Unhealthy sleep duration, defined as either less than 6 hours or more than 8 hours, may be associated with an increased risk of AF.
Key Words : Sleep, Atrial fibrillation, Systematic review.
Correspondence to: Amirhossein Sahebkar, Pharm.D, Ph.D, Fellowship
Biotechnology Research Center,
Department of Medical Biotechnology,
School of Medicine,
Mashhad University of Medical Sciences
Sleep duration and pattern show differences in developed countries due to longer work time, more availability, higher rate of shift-work, induced excessive daytime sleepiness and tiredness [1]. Several studies introduced both short and long sleep duration as risk factors for diseases like metabolic syndrome [2,3], diabetes [4], cerebrovascular accidents [5,6], obesity [7], hypertension [8], myocardial infarction [9-11], and dyslipidemia [12]. Also, effects of sleep duration on progression or suppression of different diseases were shown. Interestingly, these studies indicated that insufficient or excessive sleep is associated with mortality and morbidity caused by the aforementioned diseases.[13-15]. Atrial fibrillation (AF) is as regarded as the most common heart arrhythmia which is seen in clinical pratctice [16,17].
Atrial fibrillation can induce or exacerbate heart failure, myocardial infarction, thromboembolism, cerebral infarction [18] and chronic renal failure [19], which all negatively affect the quality of life and increase the duration of hospital stay [20,21]. More than 1% of adults develop AF, and the prevalence of AF increases with age [22]. Also, over 2 million people in the United States suffer from AF. The cost of continuous examinations and increased rates of hospitalization of AF patients amounted to 459 million Euro in 2000 [20,21,23,24]. Predictions suggest that the world will face a 2.5-fold increase in the prevalence of AF by the year 2050 [25]. Considering the importance of AF and its socioeconomic burden, it is necessary to identify the risk factors which crucially contribute to this condition. Many studies demonstrated associations between some predisposing factors like gender, age, hypertension, diabetes, smoking, heart's valve defects, myocardial infarction, heart failure and sleep-disordered breathing, and the incidence of AF [20,21,23,24]. Though there is some evidence for possible association between sleep duration and the incidence of AF, there is considerable controversy with respect to the result of the related studies.The aim of this study was to systematically search the literature to evaluate observational studies in the field and assess the possible association between sleep duration and the incidence of AF in order to obtain a quantitative estimate of the risk.
Scientific databases including PubMed, EMBASE, and Web of Sciences were searched up to November 2017 using the terms (“sleep” OR "insomnia") AND (“atrial fibrillation” OR “auricular fibrillation”). All sets included Medical Subject Heading (MeSH) and free-text terms, and papers that were written in English were evaluated. Search strategy is demonstrated in [Figure 1] No limits were applied during the search. Also, relevant publications cited in the studies were checked to increase the sensitivity of the search.
“Inclusion criteria” for the studies that are discussed in this review article were as follows: 1) all English observational studies done in human and written in English; 2) studies which included people with AF and investigated the relation between AF incidence and sleep duration; 3) study participants aging >18 years old; 4) studies in which AF was confirmed by electrocardiography (ECG); and 5) studies in which sleep duration was measured by sleep questionnaire such as, Athens insomnia scale, Pittsburgh Sleep Quality Index polysomnography (PSG) or self-reported ones.
Duplicate publications, editorials, and letters were excluded. The titles of the identified studies were assessed by two reviewers (AB and NE). If the reviewers had inconsistent ideas about an article, it was re-evaluated by a third party. We also avoided selecting duplicate or overlapping data by analyzing author names and hospitals in which patients were followed up. Quality assessment was done using Joanna Briggs Institute (JBI) critical appraisal tool that is available at http://joannabriggs.org/research/critical-appraisal-tools.html [26].
Data extraction was done by NE; then, the data were entered in Microsoft Excel 2013 and Microsoft Corp, Redmon, Washington, USA and subsequently compared with the original data by NM and AB to assure the accuracy of the extraction process. The extracted data for each study included the following information: first author name, year of publication, country, sample size, age of study population, study design and setting, AF diagnosis method, sleep assessment method, and study results. We contacted the authors who did not report data to obtain detailed information about their study, but received no response. Eligible studies are qualitatively summarized in [Table 1] It was not possible to run a meta-analysis because of the heterogeneity in sleep definition categories across the studies.
Athens Insomnia Scale (AIS) and Pittsburgh Sleep Quality Index (PSQI) are main sleep assessment questionnaires used in the studies. AIS measures the severity of insomnia using diagnostic criteria set forth by the International Classification of Diseases (ICD-10).This questionnaire evaluates sleep onset, night and early-morning waking, sleep time, sleep quality, frequency and duration of complaints, distress caused by the experience of insomnia, and interference with daily functioning. Severity of sleep difficulties which are affected them during last month, is shown by likert-type scales. The scores range varying from 0 (item without any problem) to 3 (indicating more acute sleep difficulties). Developers Soldatos and colleagues suggest a cutoff score of 6, which correctly distinguished between insomnia patients and controls in 90% of cases. [27,28]. PSQI is a self-reporting questionnaire used to measure the patterns and quality of sleep in the older adult. Different aspects of sleep was assessed by this instrument, as subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, and sleep disturbances, use of sleep medication, and daytime dysfunction over the last month. High validity and reliability of the PSQI proved by many studies. [29,30].
After finding 3003 articles in PubMed, Web of Sciences, EMBASE, we excluded duplicated and irrelevant records and finally reviewed six papers to assess eligibility for selection ([Figure 1]).In these six eligible studies ([Table 1]), a total of 186,323 individuals were recruited
Figure 1. Flow diagram of the study.
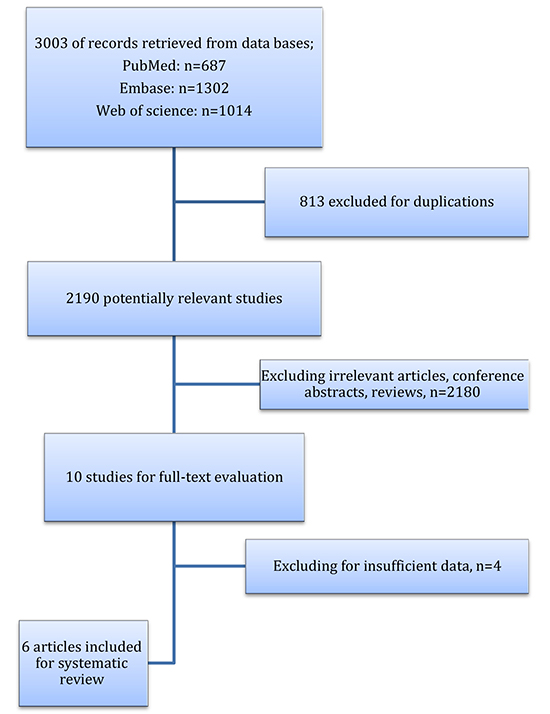
Table 1. Qualitative summary of studies.
| Study details:
First author, publication date, country
|
Sample size, sample size in AF and non-AF (n)
|
Study Age
|
Study type, setting |
AF diagnosis |
Sleep assessment |
Study result |
| Han, 2017, China |
8371
AF = 50
Non-AF= 8321
|
42.4±13.1 |
cross-sectional study
population-based
|
self-reported history or the standard 12-lead ECG one of the following criteria was present: (1) irregular RR intervals, (2) absence of repeating P waves, and (3) irregular atrial activity. |
The Athens Insomnia Scale (AIS-8) |
OR: 1.92 for insomnia
( 95% CI:1.00-3.70, P = 0.05)
OR: 1.92 for insomnia
in those age <40 years (OR: 6.52, 95% CI: 1.64-25.83, P = 0.01),
those age ≥60 years (OR: 2.29, 95% CI: 0.95-5.55, P = 0.07).
|
| Kayrak, 2013, Turkey |
1053
AF: 153
Non AF:150
|
303 patients (mean age: 63 ± 12 years) who
had nonvalvular AF
and 150 age- and gender-matched control subjects
(mean age: 61 ± 14 years)
|
cohort study
hospital-based
|
Electrocardiography
(ECG)
|
using the Pittsburgh Sleep
Quality Index (PSQI).11
|
Adjusted OR: 3.34 (95%CI:1.99-5.61)in poor sleep (PSQI >5) patients
adjusted for: age, sex, body mass index, hypertension, diabetes, heart rate
|
| Khawaja, 2013, USA |
18,755
AF:1,468
Non AF:
17,287
|
mean:68 ±8.6 |
Prospective cohort study
population-based
US male physicians( PHS)
|
Incident AF was ascertained through yearly follow-up questionnaires. |
For self-reported sleep duration. duration: ≤6, 7, and ≥8 hours. categories of sleep duration. |
adjusted hazard ratios (95% CI) for AF were 1.06 (0.92–1.22) in sleep<=6 h, 1.0 (sleep =7h), and 1.13 (1.00–1.27) in sleep >=8
adjusted for: age, sex, exercise frequency, body mass index, alcohol consumption, smoking, hypertension, diabetes, dyslipidemia, sleep apnea, snoring, coronary heart disease, congestive heart failure
subgroup analysis: in sleep apnea patients:
2.26 (1.26-4.05) in sleep<=6 h, 1.0 (sleep =7h), and 1.34 (0.73-2.46) in sleep >=8
|
| Kokubo, 2014, Japan |
6,780
AF:244
Non AF: 6536
|
30 to 84 years old |
Prospective cohort study
population-based
(Suita study)
|
Incident AF including: AF was present on/ AF was indicated as a present illness by either annual questionnaire responses or medical records. |
Sleep duration was classified into 4 categories: <6, 6 and 7, 8, and ≥9 hours levels. |
Compared with 6 and 7 hours sleep duration, the adjusted HRs (95% CIs) of incident AF were 1.58 (1.18-2.13) for who slept less than 6 hours.
adjusted for: age, sex, body mass index, alcohol consumption, smoking, hypertension, diabetes, dyslipidemia
|
| Lee, 2017, Taiwan |
64,421 cases with insomnia and 128,842 control
AF:
1,674 (2.6%) cases in patients with insomnia 2,925 (2.3%) in non-insomnia group
Non AF:
62,747 cases in patients with insomnia 125,9170 in non-insomnia group
|
18-40 : Insomnia(14,402), Non-insomnia(28,804) | 41-65: Insomnia(32,621), Non-insomnia(65,242) | ≥65: Insomnia(17,398), Non-insomnia(34,796) |
retrospective National Cohort Study
population-based
|
Incident Atrial fibrillation (ICD-9-CM codes 427.31) is defined as the diagnosis of atrial fibrillation in an outpatient department and admission. |
patients with a diagnosis of insomnia 3 times [based on (ICD-9-CM) code 780.52] within one year |
HR = 1.33, 95% CI: 1.25-1.41 in patients with insomnia
Matched cohort on age, sex, index date, comorbidity
|
| Qiaofeng Song, 2017, China |
n=87,693
AF:322
Non AF:
87,371
|
50.54 (18-98) |
prospective cohort study
population-based
|
Incident
Atrial fibrillation diagnosis was made on a standard 12-lead electrocardiogram and via self-reported history
|
self-reported responses to
the question, were categorized into three groups: short (≤6 hours), average (7 hours), and long (≥8 hours).
|
adjusted hazard ratios (95% CI) for AF were 1.07 (0.75-1.53) in sleep<=6 h, 1.0 (sleep =7h), and 1.5 (1.07-2.10) in sleep >=8
adjusted for: age, sex, education, body mass index, alcohol consumption, physical activity, smoking, hypertension, diabetes, dyslipidemia, sleep apnea, snoring, myocardial infarction, uric acid, and high sensitivity C-reactive protein.
|
Among the six studies included in this systematic review, two studies were conducted in China, and one in the USA, Turkey, Taiwan, and Japan each. Three studies assessed AF using a self-reported questionnaire during the follow-up period [13,31,32] and four studies used ECG to evaluate AF [15,32-34]. Sleep duration per day was assessed by a self-reported questionnaire in four studies[13,15,31,32] while other studies used Athens Insomnia Scale (AIS-8) [32], ICD-9-CM code 780.52 [33], and Pittsburgh Sleep Quality Index (PSQI) [34].
Sleep duration per day was classified into three categories (≤6, 7 and ≥8 hours) in two studies [13,15] and four categories (<6, 6-7, 8 and ≥9 hours) in one study [31]. Mean and standard deviation of sleep duration per day were reported by two studies [13,32]; also, one study reported the frequency of insomnia [32].
Two population-based cohort studies reported that sleep duration of ≥8 hours was associated with an increased risk of AF. Also, in one study, this association was of borderline significance as adjusted hazard ratios (aHR) for AF were 1.06; 95% CI: 0.92-1.22 for sleep duration of ≤6 and 1.13; 95%CI: 1.00-1.27 for sleep duration of ≥8 hours compared to 7-hour sleeping. Another study reported aHR= 1.07, 95% CI:0.75-1.53 and aHR= 1.5, 95% CI: 1.07-2.10 for sleep duration of ≤6 and ≥8, respectively compared to 8 hours of sleeping per day [13,15].
In four studies, shorter sleep durations were associated with an increased risk of AF. Also, in a study conducted in Japan, an aHR= 1.58, 95% CI: 1.18 -2.13 was found for sleeping durations of less than 6 hours and increased risk of AF compared to sleeping durations of 6-7 hours per day [31]. In a population-based study conducted in China, patients with AF had shorter sleeping durations compared to those without AF (6.7± 1.3 hours vs. 7.2± 1.2 hours; P < 0.05) [32]. In another population-based cohort study conducted in Taiwan, patients with insomnia were at a higher risk of AF (aHR= 1.33, 95% CI: 1.25-1.41) as compared to patients without insomnia [33].
Sleep duration in a Turkish population-based cohort was lower in patients with AF than the individuals without AF (PSQ score: 1.4±1.1 in subjects without AF vs. 2.00±1.1 in patients with AF, p<0.001) [34]. In a sub-group of patients with sleep apnea, the risk of AF was higher in patients who slept for ≤ 6 hours compared to those with 7-hour sleep duration (aHR= 2.26, 95% CI: 1.26-4.05) [13].
To the best of our knowledge, this study is the first systematic review assessing possible association between sleep duration and AF. We found that both short and long durations of sleep were associated with AF, with shorter durations being associated with a higher risk. Four studies out of the six identified studies, reported that shorter sleeping duration was associated with increased risk of AF, while two studies reported that longer sleeping duration was associated with increased AF risk. In the subgroups, patients with sleep apnea showed a similar association while patients without sleep apnea did not exhibit such an association.
The link between sleep duration and AF is still unclear. Sleep deprivation was associated with alterations in ECG parameters including signs of AF. Moreover, sleep deprivation was associated with P-wave dispersion, QT dispersion and P-wave duration, which are also predictors of AF [35,36]. Some experimental studies showed that short duration of sleep, as defined by short-term forced sleep deprivation, negatively affected endocrinological, immunological and metabolic systems [37,38]. Sleep deprivation in healthy adults was shown to be associated with left atrial early diastolic strain rate reduction which is also known as “left atrium functional impairment” [39]. Furthermore, insomnia is regarded as a risk factor that increases the probability of the development of cardiovascular diseases through autonomic dysregulation and induction of inflammation pathways [40,41].
In the MESA (Multi-Ethnic Study of Atherosclerosis) Sleep Study, longer duration of slow wave sleep (SWS) was associated with lower odds of AF before and after adjustment for apnea-hypopnea index (AHI) [42]. The association between SWS time and AF emphasizes the importance of the sleep duration in sleep health. SWS is defined as the status with the highest parasympathetic activity [43]. Sleep was associated with high parasympathetic and low sympathetic activities, which might, in turn, be associated with AF occurrence [44].
In this systematic review, both short and long sleep durations were associated with a higher risk of AF in different studies, suggesting a U-shaped association between sleep duration and AF. Nonetheless, residual confounders such as depression or anxiety disorders, which are associated with cardiovascular risk, may play an important role in this relationship [45]. Moreover, differences among the subjects of the study populations in terms of shorter or longer sleep durations, might be due to a predisposing depression [46-49]. The U-shape associations between sleep duration and cardiovascular disease (CVD), CVD risk factors and all-cause mortality were reported earlier [50,51].
It was proposed that longer sleep duration which is regarded as a deviation from normal physiological aging, may put such individuals at higher risk of premature death [52].
The studies included in this review used different categories for sleep duration; therefore, it was not possible to perform a meta-analysis based on the categories reported in the studies. Thus, we decided to report sleep durations as short and long sleep categories as well as insomnia and non-insomnia based on studies that were included. Also, since “categories” were reported and discussed instead of exact duration of sleep, it was not possible to draw a conclusion with a cut off for public health.
This study is the first systematic review to assess the association between sleep duration and atrial fibrillation, and highlighted the impact of sleep health on cardiac rhythm. Our study has some limitation as follows: long term monitoring was not used in any of the studies; sleep apnea could have contributed to the AF burden; longer sleep duration being associated with higher AF burden could just be an association and not a causative phenomenon since there is a possibility that AF itself could cause fatigue or that medications used to treat AF could produce side effects leading to higher sleep duration. One of the studies [31] included in the systematic review was only published as an abstract, and reported by The Suita study, a population-based cohort conducted in the Japanese population [53].
Authors acknowledge the help and support provided by Clinical Research Unit, Mashhad University of Medical Sciences, Mashhad, Iran.
Authors have no conflict of interest to declare.
Unhealthy sleep duration defined as sleeping for less than 6 hours or more than 8 hours per day, is associated with an increased risk of AF. Further studies are warranted to ascertain the potential association between sleep duration and AF risk as well as the optimal sleep duration that is associated with the least risk of AF.