Effect of Non-fluoroscopic Catheter Tracking on Radiation Exposure during Pulmonary Vein Isolation: Comparison of Four ablation systems
Yoshihisa Naruse1, Fehmi Keçe1, Marta de Riva1, Masaya Watanabe1, Adrianus P. Wijnmaalen1, Reza Alizadeh Dehnavi1, Martin J Schalij1, Katja Zeppenfeld1, Serge A. Trines1
1Department of Cardiology, Leiden University Medical Center, Leiden, the Netherlands.
A novel non-fluoroscopic catheter tracking system (Mediguide) can be used in combination with a 3D mapping system for atrial fibrillation (AF) ablation. However, the benefit on radiation exposure of the Mediguide system compared to other ablation systems is unknown.
We retrospectively enrolled consecutive 73 patients (51 men; 59±11 years; 60 paroxysmal AF) undergoing pulmonary vein isolation by the same operator. Radiation time, radiation effective dose, procedure time, AF recurrence after ablation, and procedure-related complications were compared among 4 different ablation systems.
Mediguide was used in 16 patients (group A), CARTO™ in 17 (group B), Cryoballoon in 30 (group C), and Multi-electrode Pulmonary Vein Ablation Catheter (PVAC) in 10 (group D). Although procedure time was shorter in patients with Cryoballoon (median 110 [interquartile range 99-120] min) and PVAC (123 [112–146] min) compared to those with Mediguide (181 [168–214] min) and CARTO (179 [160–195] min) (P<0.001), radiation exposure time and effective dose were decreased in patients with Mediguide compared to the other ablation systems (A: 5 [3–6] min; B: 14 [11–16] min; C: 14 [11–18] min; D: 20 [16–24] min, P<0.001 and A: 1.1 [0.8–2.0] mSv; B: 2.5 [1.3–3.8] mSv; C: 2.0 [1.4–2.5] mSv; D: 1.7 [1.4–3.6] mSv, P=0.015, respectively). AF recurrence rates and procedure-related complications were comparable among the 4 groups.
The Mediguide system reduces radiation exposure compared to other ablation systems without increasing AF recurrence or procedure-related complications.
Key Words : Radiation exposure, Atrial fibrillation, Ablation, Non-fluoroscopic Catheter Tracking System.
Correspondence to: Dr. Serge A. Trines
Department of Cardiology, Leiden University Medical Center, postal zone C-05-P, PO Box 9600, 2300RC, Leiden, the Netherlands
Atrial fibrillation (AF) is the most prevalent arrhythmia with an increasing incidence related to the aging of the population[1]. Radiofrequency catheter ablation of AF has become a successful treatment option for patients with both paroxysmal and persistent AF and pulmonary vein isolation (PVI) is the cornerstone of AF ablation[2]. Although 3-dimensional electro-anatomical mapping systems (EAMS) are routinely used to facilitate catheter navigation, conventional fluoroscopy is still needed for intracardiac catheter manipulation throughout the procedure. Therefore, to minimize radiation exposure for both patients and physicians, alternative catheter tracking systems are needed.
A novel non-fluoroscopic catheter tracking system (Mediguide Technology, St Jude Medical, St Paul, MN) can be used in combination with an EAMS (NavX-Ensite Velocity, St Jude Medical, St Paul, MN) in PVI, which enables continuous visualization of multiple catheter positions in pre-recorded cine loops[
3-
5]. Although a recent study showed that the use of Mediguide in combination with Ensite Velocity during PVI reduces radiation exposure compared to Ensite Velocity only,3 no study has compared the radiation exposure during PVI using the Mediguide/Ensite Velocity versus single-shot devices. The aim of our study was to investigate whether radiation exposure can be reduced using Mediguide system during PVI as compared to other ablation systems including a conventional EAMS (CARTO3, Biosense Webster, Diamond Bar, CA), Cryoballoon catheter (Arctic Front Advance, Medtronic, Minneapolis, MN), and multi-electrode pulmonary vein ablation catheter (PVAC-GOLD, Medtronic, Minneapolis, MN)
We retrospectively enrolled 524 patients who underwent radiofrequency catheter ablation of drug refractory AF in the Leiden University Medical Center between January 2014 and June 2016. Out of 524 patients, 189 patients with prior PVI, 99 patients who had additional ablation targets besides PVI and 163 patients treated by an electrophysiology fellow were excluded from this study to eliminate other factors influencing radiation time (e.g. complexity of the ablation, experience of the operator). The final study population comprised 73 patients[Figure 1].
Figure 1. Study design. Patients excluded from the analysis are indicated by arrows directed to the right. EP indicates electrophysiological; and PVI, pulmonary vein isolation.

The patients were classified in 4 groups according to the type of ablation system used during the procedure. The Mediguide system in combination with NavX-Ensite Velocity was used in 17 patients, CARTO 3 in 22, Cryoballoon in 48, and PVAC-GOLD in the remaining 10 (Figure 2). The selection of the ablation system depended on the physician’s decision. Primary endpoints were radiation exposure time, radiation effective dose and procedure time (from puncture to removal of sheaths) and secondary endpoints were AF recurrence after PVI and procedure-related complications. Data on age, sex, body mass index, cardiac risk factors and medication were collected. Transthoracic echocardiograms were reviewed for left atrium diameter, left ventricular ejection fraction, and valvular disease. All patients were treated according to our standard clinical protocol and provided informed consent. The Dutch Central Committee on Human-related Research (CCMO) permits use of anonymous data without prior approval of an Institutional Review Board, if the data are obtained for patient care and if the data do not contain identifiers that could be traced back to the individual patient
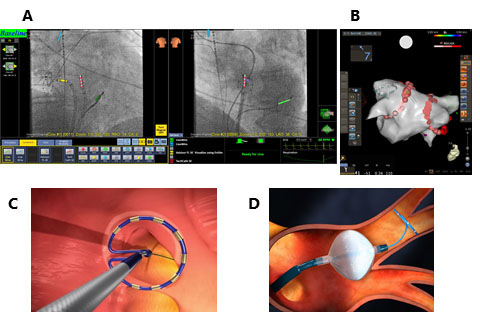
Figure 2. Images of 4 ablation systems. A: Mediguide, B: CARTO, C: Cryoballoon (by courtesy of Medtronic Japan Co., Ltd.), and D: PVAC (by courtesy of Medtronic Japan Co., Ltd.).
Non-fluoroscopic Catheter Tracking System and Fluoroscopic Settings
The Mediguide technology has been described previously[6]. In brief, the Mediguide system is installed on a Siemens fluoroscopy system (Siemens Healthcare GmbH, Erlangen, Germany). The transmitter unit is integrated with the fluoroscopy detector of the X-ray imaging system and is able to create the electromagnetic field in alignment with the fluoroscopic field of view. The sensor-equipped catheter can be either visualized on conventional fluoroscopy or tracked non-fluoroscopically on pre-recorded fluoroscopy or cine loops.
For all four ablation systems, the pulse rate of the fluoroscopy system was set at 7.5 pulses per second for live X-ray (Dose of 10nGy/p, Pulse width of 8.0ms, copper filter of 0.6–0.9mm) and the frame rate was set at 10 frames per seconds for cine-angiocardiography (Dose of 0.17 microGy/p, pulse width of 6.4ms, copper filter of 0.1–0.2.mm).
In all patients, a 320-slice Computer Tomography (Aquilion ONE, Toshiba Medical Systems, Otawara, Japan) was performed prior to the ablation to visualize the anatomy of the PVs and to guide the procedure[7]. All patients were on uninterrupted oral anticoagulation at the time of the ablation. Intravenous heparin was administered to maintain an activated clotting time of 300 to 400 seconds throughout the procedure.
Mediguide and CARTO procedures were performed using an EAMS (Navx-Ensite Velocity System and CARTO 3), an irrigated 3.5-mm ablation catheter (Coolpath Duo, St Jude Medical, St Paul, MN, or Thermocool [n=19] or Thermocool Smart Touch Surround Flow [n=3], Biosense Webster, Diamond Bar, CA), and a 10-polar circular mapping catheter (Lasso 2515, Biosense Webster, Diamond Bar, CA). Point-by-point ipsilateral PVI was performed. Radiofrequency power was delivered at 25W for a maximum duration of 30 seconds per application at the roof and the posterior left atrial wall and 30W at the anterior left atrium.
Cryoballoon ablation was performed with a 28-mm cryoballoon catheter (Arctic Front Advance, Medtronic, Minneapolis, MN) and an integrated circular mapping catheter (Achieve, Medtronic, Minneapolis, MN). The cryoballoon was advanced towards each PV to achieve occlusion verified by contrast injection. Cryoablation was performed with a single application of 240 sec in all PVs but the right superior PV (180 sec), resulting in PVI. To prevent phrenic nerve palsy, continuous phrenic nerve stimulation (cycle length of 2000ms and output of 20mA) was performed during ablation of the right PVs. A temperature probe was inserted in the esophagus to monitor the luminal esophageal temperature (Sensitherm, St Jude Medical, St Paul, MN). If loss of phrenic nerve capture or decrease in the luminal esophageal temperature below 18°C occurred, ablation was immediately interrupted by the “double stop” technique[8].
PVAC ablation was performed with the PVAC-GOLD catheter at a setting of 2:1 bipolar to unipolar energy. PVs were electrically isolated by targeted ablation of each PV antrum.
The endpoint of the ablation was PVI, defined as the presence of bidirectional conduction block from the atrium to the PVs and vice versa. Confirmation of bidirectional conduction block at least 30 minutes after successful PVI was routinely performed. Sinus rhythm was restored by cardioversion at the end of the procedure when needed.
Follow up and Definition of AF Recurrence
All patients were followed 3, 6 and 12 months after the procedure with a 12-lead ECG, 24 hour Holter monitoring and an exercise tests. Patients were encouraged to obtain ECG documentation in case of recurrent symptoms. Recurrence was defined as evidence of AF, atrial tachycardia or atypical flutter on a 12-lead ECG or lasting ≥30 seconds on Holter monitoring. A 3 month post-procedural blanking period was applied[9].
Continuous variables were expressed as means ± standard deviation or medians (IQR). Differences among 4 groups for continuous variables were determined by analysis of variance or Kruskal-Wallis test according to the data distribution with or without normality, and post hoc analyses were performed with Bonferroni test. All categorical variables were presented as the number and percentage in each group and were compared by a Fisher’s exact test. A comparison of the probability of the freedom from AF among 4 groups was performed using Kaplan-Meier survival analysis with log rank test. “Time 0” for the survival analyses was the date of PVI. Multiple linear regression analysis was used to determine independent predictors of radiation effective dose. Variables that achieved statistical significance (P<0.05) or were close to significance (P<0.1) in the Spearman's rank correlation coefficient were included in the multiple linear regression analysis. A P value <0.05 was considered statistically significant. All analyses were performed with R (The R Foundation for Statistical Computing, Vienna, Austria, version 3.1.1)
Demographic and Clinical Characteristics among 4 groups
Demographic and clinical characteristics are summarized in [Table 1]. The mean age of the patients was 59±11 years and 70% were male. Persistent AF was observed in 13 (18%) patients. The mean left atrial diameter was 41±5 mm. There was no significant difference in baseline clinical and echocardiographic characteristics among the 4 groups [Table 1].
Table 1. Demographic and Clinical Characteristics among 4 groups.
| Variable |
All
(n = 73)
|
Mediguide
(n = 16)
|
CARTO
(n = 17)
|
Cryoballoon
(n = 30)
|
PVAC
(n = 10)
|
P-value
|
| Age, years |
59 ± 11 |
61 ± 11 |
54 ± 15 |
61 ± 9 |
62 ± 9 |
0.142 |
| Male gender, n (%) |
51 (70%) |
13 (81%) |
13 (77%) |
19 (63%) |
6 (60%) |
0.492 |
| Body mass index, kg/m2 |
26 ± 3 |
26 ± 3 |
26 ± 3 |
27 ± 4 |
27 ± 3 |
0.768 |
| Persistent AF, n (%) |
13 (18%) |
5 (31%) |
4 (24%) |
2 (7%) |
2 (20%) |
0.177 |
| Risk factors |
|
|
|
|
|
|
| Hypertension, n (%) |
35 (48%) |
9 (56%) |
9 (53%) |
12 (40%) |
5 (50%) |
0.708 |
| Hyperlipidemia, n (%) |
20 (27%) |
3 (19%) |
5 (29%) |
8 (27%) |
4 (40%) |
0.696 |
| Diabetes mellitus, n (%) |
3 (4%) |
1 (6%) |
1 (6%) |
1 (3%) |
0 (0%) |
0.850 |
| Smoking, n (%) |
26 (36%) |
3 (19%) |
8 (47%) |
11 (37%) |
4 (40%) |
0.383 |
| Chronic kidney disease, n (%) |
7 (10%) |
1 (6%) |
2 (12%) |
2 (7%) |
2 (20%) |
0.605 |
| Ischemic heart disease, n (%) |
6 (8%) |
0 (0%) |
3 (18%) |
2 (7%) |
1 (10%) |
0.311 |
| Echocardiographic findings |
|
|
|
|
|
|
| LA diameter, mm |
41 ± 5 |
42 ± 6 |
42 ± 7 |
40 ± 4 |
43 ± 5 |
0.335 |
| LV ejection fraction, % |
65 ± 11 |
60 ± 12 |
64 ± 14 |
66 ± 8 |
70 ± 9 |
0.096 |
| Valvular disease, n (%) |
2 (3%) |
0 (0%) |
1 (6%) |
1 (3%) |
0 (0%) |
0.705 |
| Medication |
|
|
|
|
|
|
| Statin, n (%) |
18 (25%) |
3 (19%) |
6 (35%) |
7 (23%) |
2 (20%) |
0.687 |
| ACE-I/ARB, n (%) |
30 (41%) |
8 (50%) |
9 (53%) |
10 (33%) |
3 (30%) |
0.429 |
| Β-blocker, n (%) |
42 (58%) |
10 (63%) |
8 (47%) |
17 (57%) |
7 (70%) |
0.666 |
| Class I AAD, n (%) |
30 (41%) |
5 (31%) |
7 (41%) |
13 (43%) |
5 (50%) |
0.794 |
| Sotalol, (%) |
15 (21%) |
5 (31%) |
3 (18%) |
6 (20%) |
1 (10%) |
0.594 |
| Amiodarone, n (%) |
8 (11%) |
2 (13%) |
1 (6%) |
2 (7%) |
3 (30%) |
0.189 |
| Warfarin, n (%) |
59 (81%) |
10 (63%) |
15 (88%) |
24 (80%) |
10 (100%) |
0.091 |
| NOAC, n (%) |
14 (19%) |
6 (37%) |
2 (12%) |
6 (20%) |
0 (0%) |
0.091 |
Values are reported as the mean ± standard deviation or n (%). AAD indicates anti-arrhythmic drug; ACE-I, angiotensin converting enzyme inhibitor; AF, atrial fibrillation; ARB, angiotensin receptor blocker; LA, left atrium; LV, left ventricle; and NOAC, new oral anticoagulant.
Primary and Secondary Endpoints
Procedure time was shorter for the Cryoballoon (median 110 [IQR 99–120] min) and PVAC groups (123 [112–146] min) compared to Mediguide (181 [168–214] min) and CARTO (179 [160–195] min, P<0.001 by Kruskal-Wallis test)[Figure 2]. However, of importance, patients in the Mediguide group experienced a shorter radiation time (5 [3–6] min) compared to CARTO (14 [11–16] min), Cryoballoon (14 [11–18] min), and PVAC (20 [16–24] min) groups (P<0.001)[Figure 3]. Furthermore, radiation effective dose differed significantly among the 4 groups (1.1 [0.8–2.0] mSv in Mediguide; 2.5 [1.3–3.8] mSv in CARTO; 2.0 [1.4–2.5] mSv in Cryoballoon; and 1.7 [1.4–3.6] mSv in PVAC, P=0.015) [Figure 4]. Multiple linear regression analysis identified a low body mass index (P=0.001), short left atrial diameter (P=0.004), and the use of the Mediguide system (P=0.001) as independent factors associated with a decrease of radiation effective dose [Table 2].
Figure 3. Procedure time among 4 groups.* P<0.001 vs. Mediguide, † P<0.001 vs. CARTO, ‡P<0.01 vs. Mediguide, § P<0.01 vs. CARTO
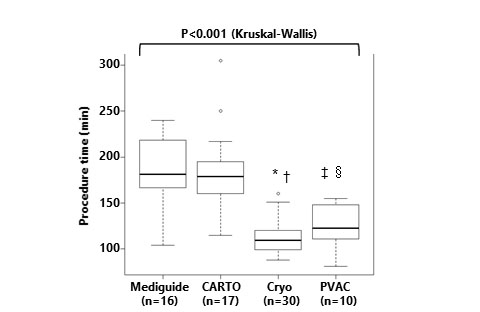
Figure 4. Radiation time among 4 groups.* P<0.001 vs. CARTO, † P<0.001 vs. Cryo, ‡P<0.001 vs. PVAC
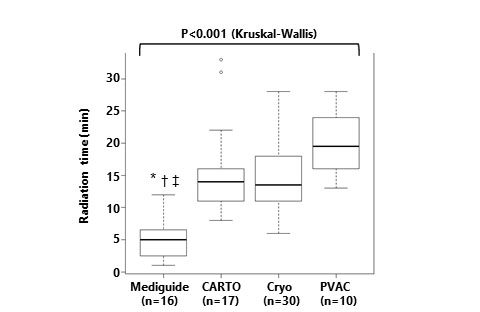
Figure 5. Radiation effective dose among 4 groups.* P<0.05 vs. Cryo
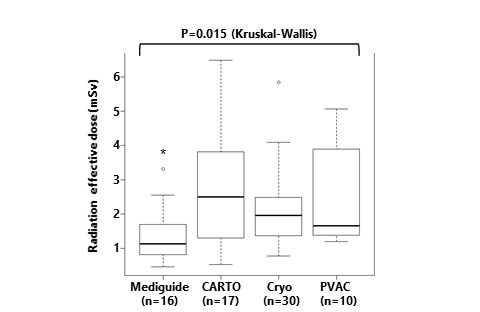
Table 2. Multiple Linear Regression Analysis of Variables Associated with Radiation Effective Dose.
| Variable |
Beta |
Standard error |
T value |
P-value |
| (Constant) |
-0.434 |
1.227 |
-3.535 |
<0.001 |
| Left atrial diameter |
0.074 |
0.025 |
2.968 |
0.004 |
| Body mass index |
0.139 |
0.041 |
3.366 |
0.001 |
| Mediguide usage |
-1.077 |
0.322 |
-3.341 |
0.001 |
| R2 = 0.381 (P<0.001) |
|
|
|
|
Procedural related complications did not differ among the 4 groups [Table 3]. None of the study patients experienced a stroke or transient ischemic attack. Transient phrenic nerve palsy occurred in 1 patient of the Cryoballoon group (P=0.693). Two patients (1 in the Mediguide group and 1 in the CARTO group) had pericardial bleeding requiring percutaneous drainage (P=0.476).
During a mean follow-up of 11±4 months, 23 (32%) patients had AF recurrence. Kaplan-Meier curve demonstrated that there was no significant difference in the recurrence of AF among the 4 groups (P=0.753 by log-rank test)
Table 3. Procedural related complications among 4 groups.
| Variable |
All
(n = 73)
|
Mediguide
(n = 16)
|
CARTO
(n = 17)
|
Cryoballoon
(n = 30)
|
PVAC
(n = 10)
|
P-value
|
| Stroke/TIA, n (%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (0%) |
1.0 |
| Cardiac tamponade, n (%) |
2 (3%) |
1 (6%) |
1 (6%) |
0 (0%) |
0 (0%) |
0.476 |
| Phrenic nerve palsy, n (%) |
1 (1%) |
0 (0%) |
0 (0%) |
1 (3%) |
0 (0%) |
0.693 |
| Permanent, n (%) |
0 (0%) |
0 (0%) |
0 (0%) |
0 (%) |
0 (0%) |
1.0 |
| Transient, n (%) |
1 (1%) |
0 (0%) |
0 (0%) |
1 (3%) |
0 (0%) |
0.693 |
Values are reported as n (%). TIA indicates transient ischemic attack.
The main findings of the present study are as follows: (1) patients undergoing a first PVI by an experienced operator with the use of Mediguide in combination with Ensite Velocity had a shorter radiation time and received less radiation effective dose compared to those undergoing PVI with a stand-alone nonfluoroscopic system (CARTO) or a single-shot device (either PVAC or Cryoballoon); (2) a lower body mass index, a shorter left atrial diameter and the use of Mediguide were independently associated with a lower radiation effective dose; (3) procedure time was significantly shorter when a single-shot device was used (Cryoballoon or PVAC); and (4) the prevalence of procedure-related complications and the incidence of AF-recurrence during follow-up did not differ among groups.Since the patient population of this study was relatively healthy (mean LA diameter of 41mm and 82% of patients had paroxysmal AF), the effect of non-fluoroscopic catheter tracking on radiation exposure would have been even more pronounced if sicker patients are examined.
Importance of reducing radiation exposure during ablation procedures
To minimize radiation exposure is important for both patients and physicians. Statistically significant increases in brain cancer and leukemia have been reported with doses as low as 30 mSv in children[10]. Previous studies estimated that a radiation exposure of 53 to 60 minutes during radiofrequency catheter ablation would result in 0.7 to 1.4 excess fatal malignancies per 1000 women and 1.0 to 2.6 per 1000 men[11]. Roguin et al. reported the occupational radiation exposure-induced brain and neck tumors among 31 physicians performing interventional procedures. In this group, approximately 85% of the brain tumors were located in the left cerebral hemisphere, while brain tumors are normally evenly distributed between both hemispheres. The fact that the brain is relatively unprotected and that the left side of the head is known to be more exposed to radiation could explain these findings[12].
Impact of use of a Non-fluoroscopic Catheter Tracking System during AF ablation on radiation exposure
In the early 2000’s, several studies reported mean radiation times of 57–130 minutes in patients undergoing fluoroscopy-guided PVI[11,13]. The advent of three-dimensional non-fluoroscopic EAMS (CARTO and Ensite) resulted in a significant reduction in radiation exposure during AF ablation[14],[15]. Fluoroscopy-guided single shot devices (PVAC/Cryoballoon) were subsequently developed to reduce procedural duration[16-18] However, the beneficial effect of single shot devices on reducing radiation exposure remains controversial[16-18]. The usage of a novel non-fluoroscopic catheter tracking system (Mediguide technology) in combination with an EAMS (Navx Ensite Velocity) has enabled operators to further minimize radiation exposure compared to use of an EAMS only[3]. However, no data are available comparing radiation exposure during PVI performed with a single shot device or with the Mediguide technology.
In line with previously reported, in our study, procedural time was shorter when PVI was performed with a single shot device (cryoballoon or PVAC) than when point-by-point ablation was performed using an EAMS (Carto or Ensite). However, and, of importance, radiation time were shorter when the combination of Mediguide/Ensite was used for ablation than when either a single shot device or the Carto system alone were used.
Although single shot devices create larger ablation lesions with a single application resulting in reduced procedure time, they rely on fluoroscopy to verify catheter positions resulting in a relatively high radiation dose in spite of short procedure time. On the other hand, point-by-point ablation using a conventional EAMS requires a large number of catheter manipulations, which may increase radiation dose as well, even when the procedure is performed by an experienced operator (as it was the case in our study). These factors may explain why radiation exposure did not differ among the CARTO, Cryoballoon, and PVAC groups.
Our findings demonstrate that a technology which enables the visualization of catheters on prerecorded Cine loops can successfully help to reduce radiation exposure during AF ablation. Currently, the CARTO system has also a module for integrating fluoroscopy with EAMS (UNIVU) that has proven to reduce radiation exposure[19].
In addition to the use of Mediguide technology, body mass index and LA diameter were independently associated with radiation dose during PVI. It is well known that obesity is associated with an increase in radiation dose[20]. However, the association between LA size and radiation dose has not been reported so far. We speculate that in patients with an enlarged LA, catheter manipulation and catheter contact might be more difficult. Furthermore, in the presence of the larger PV antra, more ablation lesions might be needed to achieve PVI, both factors resulting in prolonged radiation time and dose.
Although the risk of carcinogenesis with the reported radiation exposure levels during PVI might be low, it is prudent to follow the principles of keeping the radiation dose “as low as reasonably achievable” not only for patients but also for physicians[21]. Our findings indicate that the Mediguide technology can contribute to follow this policy without an increase in complication and AF recurrence. In addition, the Mediguide system might enable operators to perform PVI without wearing a lead apron as the necessity of fluoroscopy is rare after pulmonary venography. This may result in a decrease in operator fatigue and musculo-skeletal problems
The present study has several limitations. First, our study had a retrospective design with a relatively small sample size and should be considered as a hypothesis-generating study, not a conclusive trial.Moreover, the systems were not randomized but selected by operator’s decision; that may have caused a bias. However, in our center the selection of mapping systems for an individual procedure is rather arbitrary and depends more on availability of different cathlab rooms than on patient characteristics. In addition, our study population was relatively homogeneous, since we excluded patients with prior PVI, additional ablation besides PVI, and patients treated by an electrophysiological fellow. Nonetheless, further prospective and ideally multicenter randomized studies with larger sample sizes are necessary to confirm our results. Second, the cost of the system itself and the increased costs associated with Mediguide sensor-equipped tools may be a drawback for implementation of this system. Third, the reported findings come from an experienced operator at a high-volume referral center and may therefore not be applicable to smaller less experienced centers.Forth, contact force sensing catheters were only used in three CARTO procedures in this study. The systemic use of contact sensing catheters may be able to reduce procedure and radiation time in the CARTO and Mediguide groups. Finally, radiation dose could have been more reduced if lower pulse and frame rates were applied. In the version of the Mediguide software used in the procedures described in this article, the system did not work with a framerate below 7.5 f/s. In the current version of the software, this restriction is solved.
The use of Mediguide system in combination with Ensite Velocity during PVI reduces radiation exposure compared to other ablation systems without increasing AF recurrence or procedure-related complications. Therefore, physicians should consider the usage of new fluoroscopy integrating technology in an EAMS (Mediguide or CARTO UNIVU) to minimize radiation exposure.