The Importance of Atrial Fibrillation’s Associated Comorbidities as Clinical Presentation and Outcome Contributors
James A. Reiffel1
1Columbia Memorial Hospital and New York Presbyterian Westchester Division.
Atrial fibrillation (AF) has a heterogeneous clinical presentation. It can occur: (a) in the presence or absence of detectable heart disease, and, (b) with or without relatedsymptoms. Its prognosis in terms of thromboembolismand mortality is most benign when applied to young individuals (aged less than 60 years) without clinical orechocardiographic evidence of cardiopulmonary disease [termed “lone AF”]. However, by virtue of aging or because of the development of concomitant cardiovascular disorders, patientsmove out of the lone AF category over time, accompanied by increased risks for thromboembolism and mortality. Thus, underlying and/or associated comorbidities must play an important role in the presentation and consequences of patients with AF. While, no doubt, most clinicians likely appreciate that the majority of the AF patients they see have associated cardiovascular, pulmonary, metabolic, endocrinologic, genetic, and/or other disorders, it is not clear how much they appreciate that these disorders directly relate to the presenting symptoms and to the risks from AF in addition to their role as risk factors (or markers) for AF. This issue is the subject of this review manuscript.
Key Words : .
James A. Reiffel, M.D.
202 Birkdale Lane, Jupiter, FL 33458 USA
Atrial fibrillation (AF) has a heterogeneous clinical presentation. It can occur: (a) in the presence or absence of detectable heart disease, and, (b) with or without relatedsymptoms. Its prognosis in terms of thromboembolismand mortality is most benign when applied to young individuals (aged less than 60 years) without clinical orechocardiographic evidence of cardiopulmonary disease [termed “lone AF”]. However, by virtue of aging or because of the development of concomitant cardiovascular disorders, patientsmove out of the lone AF category over time, accompanied by increased risks for thromboembolism and mortality, Thus, underlying and/or associated comorbidities must play an important role in the presentation and consequences of patients with AF. While, no doubt, most clinicians likely appreciate that the majority of the AF patients they see have associated cardiovascular, pulmonary, metabolic, endocrinologic, genetic, and/or other disorders, it is not clear how much they appreciate that these disorders directly relate to the presenting symptoms and to the risks from AF in addition to their role as risk factors (or markers) for AF. This issue is the subject of this review manuscript.
According to the 2001 ACC/AHA/ESC Guidelines for the Management of Patients
With Atrial Fibrillation (AF): (1) “AF has a heterogeneous clinical presentation, occurring in the presence or absence of detectable heart disease or relatedsymptoms…The prognosis associated with AF in terms of thromboembolismand mortality is most benign when applied to young individuals (aged less than 60 years) without clinical orechocardiographic evidence of cardiopulmonary disease [descriptors for what has been termed “lone AF”]. These patients have a favorable prognosis with respect to thromboembolism and mortality. By virtue of aging or the development of cardiac abnormalities, however, patientsmove out of the lone AF category over time, and the risks of thromboembolism and mortality rise.”1
The subsequent versions of these guidelines from 2006 through 2019 do not expound further on these comments. Implied in this statement is the importance that the underlying and/or associated comorbidities play in the presentation and consequences of patients with AF. While, no doubt,most clinicians likely appreciate that the majority of the AF patients they see have associated cardiovascular, pulmonary, metabolic, endocrinologic, genetic, and/or other disorders, it is not clear how much they appreciate and remember when facing a patient that these disorders directly relate to the presenting symptoms and to the risks from AF in addition to their role as risk factors (or markers) for AF.
“Lone AF” Can Be Instructive
“Lone AF” (or, as it was originally called, lone auricular fibrillation) is a term that was coined almost a century ago to indicate AF in the absence of associated causative structural disorders. It has also been described as benign AF, idiopathic AF, functional AF, fibrillation of unknown origin, and fibrillation without heart disease.2 In that era, the diagnostic tools available to detect possible underlying cardiovascular (CV) disorders consisted of the medical history, detailed physical examination, electrocardiography, phonocardiography, Master’s 2-step exercise test, and chest x ray. Quite likelyNo doubt, some patients classified then as “lone AF” would now be reclassified using modern diagnostic methods. In fact, multiple studies during the past few decades have revealed histopathological changes in atrial tissue obtained from apparently “lone AF” – most commonly inflammation and/or fibrosis 3-9–which indicate that subclinical atrial alterations are often present in patients otherwise labelled“lone AF”. Additionally, generelated causes for some of the patients have also become apparent as has later in life hypertension in up to 40% or more.10,11 Moreover, despite the absence of structural heart disorders, some “lone AF” patients have recognized precipitants for their events, such as parasympathetic, sympathetic, alcohol, stimulant, or sleep-apneic triggers. Nonetheless, important clinical observations were made in apparently “lone AF” patients in decades past that can teach us important messages now. These, coupled with observations in patients with AF plus associated disorders can inform us of the multiple ways such disorders interact with/affect the presentations and outcomes of the very many myriadof patients we encounter with AF.
AF can be symptomatic or asymptomatic, even in the samepatient. Symptoms can vary with the ventricular rate, underlyingfunctional status, frequency/duration/burden of AF, and individual patientperceptions.1,12,13 Additionally, mostpatients with AF complain of palpitations, chest pain, dyspnea,fatigue, or lightheadedness.1,12,13 Less frequent are thromboembolic events, overt syncope, acute ischemic events, or acute heart failure. Importantly, none of these symptoms are specific for AF, as each may result from many disease states. Nonetheless, the question should be asked as to whether and which of these symptoms result primarily from atrial fibrillation versus from or synergistically consequent to underlying cardiovascular comorbidities. [Figure 1] The answer is important because for those that are substantially linked to the underlying comorbidities, elimination of AF, whether by drugs and/or ablation may not provide any certainty of relief -- a major point of focus behind this manuscript. It is through an examination of “lone AF” that answers may become apparent and hence the contributions of comorbidities best appreciated.
Figure 1. The relationship between atrial fibrillation burden, comorbidities, and consequences.
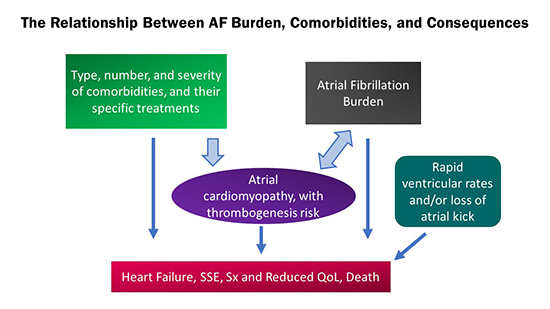
AF = atrial fibrillation. SSE = stroke and systemic embolism. Sx = symptoms. QoL = quality of life. Heart failure, thromboembolic risk, symptoms, reduced quality of life, and/or death may be directly consequent to the number, type, and severity of comorbidities present in a patient with or without AF, to the rapid ventricular rate in the presence of AF, and/or to the amount of AF present (AF burden). Both the comorbidities and the AF burden are co-contributors to the atrial cardiomyopathy present in AF patients, to the extent of the atrial cardiomyopathy, and to its thrombogenic potential.
Clinical Presentation of Lone AF
In almost all patients with AF, the immediately proximate instigators of AF are abnormalities that are present in the left atrium. These are anatomic, histopathologic, contractile, electrophysiologic, and endothelial.1,12,13 These atrial cardiomyopathic alterations are the result of mechanisms linked to underlying comorbidities and/or alterations due to AF itself via an atrial tachycardic myopathic effect 14-17[Figure 2]. In themselves, these atrial alterations do not directly produce systemic symptoms, although their etiologies can. For example, hypertension may result in LV hypertrophy with reduced diastolic compliance and increased LV pressures. These,in turn,may result in left atrial anatomic and functional alterations, increases in LA pressure, and consequent dyspnea. Accordingly, it is the elevated LV pressures that are the major factor leading to dyspnea, rather than dyspnea being just a result of LA alterations. Superimposed, dyspnea may worsen with impaired LA emptying and loss of AV synchrony if AF also develops in this physiologic setting. [Figure 1]In contrast, palpitations are likely the symptom that is most directly linked to AF itself. The sense of palpitations is a consequence of the change in pulse rate and rhythm that occurs with the development of AF. Palpitations are not noted by all AF patients, but when they are, they are a directresult of the arrhythmia. Other symptoms commonly seen in patients with AF, and perhaps most prominent during AF, such as dyspnea, chest discomfort, hypotensive- or reduced cardiac output-associated, require impairment of LV compliance, alterations in systolic output, ischemia, etc., as contributing mechanisms. If the above is all true, then “lone AF”should have palpitations as the major or only symptom – particularly if paroxysmal rather than chronic. This, in fact, is the case.
Figure 2. Atrial fibrillation and comorbidities may act in concert (synergistically) leading to poor outcomes in patients with atrial fibrillation
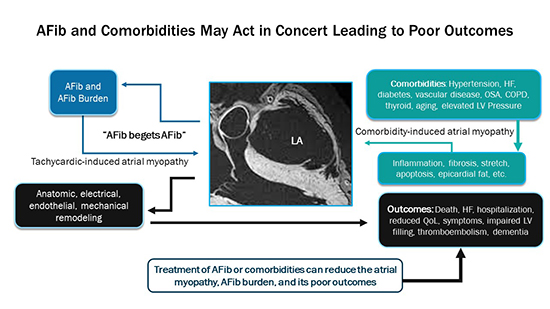
AFib = atrial fibrillation. HF = heart failure. OSA = obstructive sleep apnea. COPD = chronic obstructive pulmonary disease. LV = left ventricle/left ventricular. QoL = quality of life. Comorbidities present in patients with AF can produce histopathologic, anatomic, mechanical changes in the left atrium that result in an atrial cardiomyopathy. These changes, with associated electrophysiological alterations can result in the development of atrial fibrillation. Atrial fibrillation itself can contribute to and further worsen atrial cardiomyopathic changes, resulting in more atrial fibrillation (Afib begets Afib) which is a detrimental loop in the progression of this process. The altered atrial function (including endothelial dysfunction) can be prothrombotic, resulting in stroke and systemic embolism, as well as resulting in impaired ventricular filling and systolic function, higher pulmonary vascular pressures, and adverse outcomes associated there with.
In their 1954 manuscript titled Lone Auricular Fibrillation, Evans and Swann (2) reported on 20 patients with “lone AF” and found that: “Many of the patients were free from symptoms and the arrhythmia was found at a medical examination conducted for reasons unconnected with the heart…The single relevant complaint was palpitations, which were present in 11 of the patients.” “Shortness of breath was sometimes present, …but was always explained by aging, obesity, or commencing emphysema.” “Symptoms were absent in the other 9 patients.” This report was consistent with an earlier but smaller series published by Master and Eichert18 in which 5 naval officers, ages 23-48, were diagnosed with “functional paroxysmal auricular fibrillation.” All were without any evident associated cardiovascular disease. “Palpitation of the heart is the chief presenting symptom” was noted in each case; in two, there was a GI trigger and a sense of “distension.” In some, there was associated lightheadedness if standing and exerting. No other cardiovascular symptoms were reported. Six additional reports from 1930 to 1968 also report palpitations as the most common symptom in patients with “lone AF.” 19-24 Some had associated fatigue, “anxiety”, “weakness”, dizziness, sweating; some were asymptomatic. Mild dyspnea or chest discomfort was infrequently reported in these six papers.Not yet adequately investigated is any possible relationship between age and the awareness of palpitations in “lone AF”. If, with age, for example, a patient was to develop AV nodal conduction dysfunction in association with the electrical disorder causing AF (such as may occur in the brady-tachy syndrome patient), the ventricular rate during paroxysmal AF may slow, which might possibly alter the sense of palpitations.
While it is possible that in some of the above-reported patients, modern diagnostic methodologies might reveal mild structural heart disease or biopsies might reveal altered atrial histology, such findings would not change the observation that the overwhelmingly dominant presenting symptom, when one was present, was palpitations. Consequently, these finding suggest that when AF presents with substantial cardiovascular or pulmonary symptoms, underlying comorbidities are likely playing a role that should be addressed beyond rhythm control of the AF.
Consistent with these observations is the recent report regarding the presence of symptoms as predictors of monitor-detected subclinical AF (SCAF). In the recent REVEALAF trial, in which an inserted cardiac monitor (ICM) was used in a demographically enriched population to assess the frequency and characteristics of SCAF, 25 a substudy assessment of symptoms as predictors of detecting SCAF determined that only palpitations had an association with AF detection when controlling for other baseline symptoms (hazard ratio 1.61 (95%CI 1.12-2.32; p=0.011). 26 No other symptom studied, including dyspnea, chest pain, syncope, dizziness, or fatigue, was associated with an increased likelihood of SCAF detection. Yet, patients without detected SCAF had an even higher frequency of such other cardiovascular symptoms than those with detected SCAF.
The most feared clinical consequences of AF are thromboembolism and death. Heart failure (HF), impaired quality of life (QoL), need for hospitalization, costs are additional issues of concern. Here, again, “lone AF” can be contrasted with AF plus associated disorders, but with greater difficulty. The ACC/AHA/ESC practice guidelines 1,12 note that “the rate of stroke in patients with AF is related to coexistent cardiovascular disease. In a small,retrospective, population-based study in Olmsted County, Minnesota, over 3 decades, the 15-year cumulative stroke rate in people with lone AF (defined as those younger than age 60 years with no clinical history or echocardiographic signs of cardiopulmonary disease) was 1.3% per year.”1,12 In a larger, 30-yr follow up series reported by this same investigative center, “overall survival of the 76 patients with lone atrial fibrillation was 92% and 68% at 15 and 30 years, respectively, similar to 86% and 57% survival for the age- and sex-matched Minnesota population. Observed survival free of heart failure was slightly [but not significantly] worse than expected (p=0.051). Risk for stroke or transient ischemic attack was similar to the expected population risk during the initial 25 years of follow-up but increased thereafter (p=0.004), although confidence intervals were wide. All patients who had a cerebrovascular event had developed at least 1 risk factor for thromboembolism.” 27 They concluded that “Comorbidities significantly modulate progression and complications of atrial fibrillation. Age or development of hypertension increases thromboembolic risk.”
In accordance with the above, the ACC/AHA/HRS and ESC guidelines do not recommended anticoagulation for stroke prevention in patients with lone AF.However, they note that thromboembolic risk increases progressively as concomitant risk disorders increase [using the CHADS2 score where C = congestive heart failure, H = hypertension, A = age 75 yrs or above, D = diabetes, and S = prior stroke/systemic embolism or transient ischemic attack] and that elevated risk scores call for chronic oral anticoagulation. This strategy has not changed in concept during ensuing years. As noted by the 2020 ESC guidelines (11) common stroke risk factors are now summarized in the clinical risk-factor-based CHA2DS2-VASc score [C = congestive heart failure, H = hypertension, A = age 75 years or older, D = diabetes mellitus, S = stroke/systemic embolism/prior TIA, V = vascular disease, A = age 64-74, Sc = Sex category (female)]. These 2020 guidelines recommend no anticoagulation for a score of 0 in men or 1 in women. [Similarly, but not identically, the 2019 ACC/AHA/HRS guidelines recommend no anticoagulation for men with a score 0-1or women with a score 0-2.] However, higher scores come with recommendations for chronic oral anticoagulation, again driven by the presence of specific comorbidities. The particular difficulty with “lone AF” is the fact that aging does not protect these patients from developing the comorbidities that commonly appear in older populations, including hypertension, diabetes, vascular disease, obesity, and the like. Accordingly: (1) it is difficult to determine a true risk for thromboembolic events, or mortality, or heart failure associated with “lone AF”, as we would no longer call it “lone AF” if we encountered the same patient later in life, after hypertension and coronary artery disease, for example, were to have developed; (2) even in the “lone AF” population, as with the non-lone AF population, there is data to suggest that thromboembolic risk is greater for non-paroxysmal AF versus paroxysmal AF, and in association, the risk is greater in those that have or who develop atrial enlargement or atrial dysfunction as determined by imaging;10,11, 28, 29and,3 different series of “lone AF” patients that consist of different ages, different AF patterns, and different durations of follow up will accordingly find different rates of outcome events. For example, Scardi et al.30 reported yearly stroke rates in “lone AF” of 1.1% for paroxysmal AF but 16.3% for chronic AF (mean follow up period 10 years); Brand et al.31 reported stroke rates of 28% in “lone AF, in the Framingham data base but 7 of the 8 patients had chronic rather than paroxysmal AF (mean follow up period 30 years); Rienstra et al.32 reported stroke rates of 3% in the “lone AF” patients who participated in the RACE study (mean follow up period 2.3 years); and Kopecky et al.33 reported stroke rates in Olmsted County, MN of 4% in “lone AF” (mean follow up 14.8 years). Regardless of such differences, we should feel certain that comorbidities play an important role in the clinical presentation and prognosis associated with AF, whether they have a causative role in the development of AF and/or a modifying role in the consequences associated with AF.
Recently, the roles of AF and concomitant comorbidities re: thromboembolic risk have been further clarified with studies that have compared event rates in patients with lone AF, AF plus comorbidities, and comorbidities without AF. In 2009, Botto and colleagues 34 introduced this concept when they reported that stroke risk was dependent upon both AF presence and duration plus CHADS2 score. Using data from 568 patients with known AF and implanted pacemakers, they identified two subpopulations with markedly different risks of thromboembolic events: 0.8% vs 5% (p=0.035). The former corresponded to no device-detected AF and a CHADS2 score = 2 or less, or AF 5 minutes with CHADS2 score= 1 or less, or AF 24 hours with CHADS2 score = 0. In contrast, thromboembolic rates rose progressively as AF duration increased and/or as CHADS2 score increased in patients with a CHADS2 score of >2 to >5. In 2019, similar findings were reported in a larger series by Kaplan et al. (35)They determined rates of stroke and systemic embolism in 21,768 nonanticoagulated patients with implantable electronic devices capable of continuous AF monitoring using electronic health records. Maximal AF duration and CHA2DS2-VASc scores were assessed, with AF burdens categorized as no AF, AF 6 min-23.5 hours, AF >23.5 hours. “Both increasing AF duration (p<0.001) and increasing CHA2DS2-VASc score(p<0.001) were significantly associated with annualized risk of stroke and systemic embolism.” Stroke and systemic embolism rates were low in patients with a CHA2DS2-VASc score of 0 to 1, regardless of detected AF duration but rose to >1%/year in patients with a CHA2DS2-VASc score of 2 with >23.5 hours of AF and in those with a CHA2DS2-VASc score of 3-4 plus > 6 minutes of AF, and in patients with a CHA2DS2-VASc score of 5 or higher, even with no AF. Concordantly, Singleton et al 36 reported on observations from the REGARDS study (Reasons for Geographic and Racial Differences in Stroke) in which 28,253 patients were classified into 1 of 4 groups based upon the presence or absence of AF and the presence or absence of cardiovascular comorbidities. During 244,560 personyears of follow-up (median 8.7), 1206 strokes occurred.Compared with patients with neither AF nor cardiovascular comorbidities, there was no increased stroke risk among patients with AF alone (hazard ratio 1.23; 95% CI 0.62-2.18 [p=0.511]). However, patients without AF but with cardiovascular comorbidities had an increased risk of stroke (hazard ratio 1.77; 95% CI 1.48-2.18 [p<0.0001]) and specifically cardioembolic stroke (hazard ratio 2.34; 95% CI 1.48-3.90 [p=0.002]). These data sets are consistent with the impression that “lone AF” has a low risk of thromboembolism [likely because atrial pathophysiology is minimal – unless, perhaps, the AF is chronic] and that cardiovascular comorbidities contribute significantly to the thrombosis-prone atrial myopathy and hence embolic risk.
Patients with AF are prone to symptoms and to adverse outcomes. However, the type and severity of symptoms differ in patients with “lone AF” from those with AF plus cardiovascular comorbidities. Palpitations are the most prominent symptom in “lone AF” whereas the likelihood or severity of dyspnea, chest discomfort, hemodynamic impairment, and the like are increased in the presence of associated cardiovascular and related comorbidities. Similar symptoms, however, can occur in patients with cardiovascular disorders without accompanying AF, though they may be more pronounced when AF is also present, due to the effects of reduced ventricular filling time, loss of atrial contraction, and loss of AV synchrony consequent to the AF. Similarly, patients with “lone AF” appear to have a lower risk for major morbidity and death than patients with AF plus cardiovascular comorbidities. However, since such comorbidities may develop later in life, categorizing a patient as having “lone AF”does not mean that its more benign course will remain as such as the patient ages. Thus, continued vigilance and reevaluation of the AF patient is required during the course of clinical follow up – especially if a change in symptoms is noted. Finally, it now appears likely that simply classifying a patient as having or not having AF, whether lone or not, is no longer sufficient. We must also appreciate the presence/absence and importance of any associated left atrial cardiomyopathy – especially its key role in stroke risk. In such a construct, the role of AF as a risk factor for thromboembolism must encompass enough AF to contribute to the myopathic alterations that reach the threshold necessary for thrombogenesis. With lone AF, absent associated cardiomyopathies, such a threshold is rarely reached. Additionally, the length of AF runs and total AF burden must be more important than occasional relatively short episodes of paroxysmal AF. Simultaneously, the greater the number and severity of associated contributory comorbidities, the shorter might be the AF runs that become importantly contributory. Thus, notably, in this framework, the presence or absence of any AF-associated symptoms is not important with respect to atrial myopathic thrombogeneisis and stroke risk. Accordingly, it is not surprising that the very recent manuscript by the GARFIELD-AF registry investigators (which did not include patients with lone AF) noted that major outcomes (stroke, bleeding) do not differ between symptomatic and asymptomatic AF presentations. 37 However, adverse outcomes as well as symptomatic presentations are different in patients with versus without associated comorbidities, as well as which ones and their severity.