Outcomes Of Manifest Right Free Wall Accessory Pathway Ablation: Data From A Single Center
Matthew T. Brown, Soroosh Kiani, George B. Black, Marvin LR Lu, Neal Bhatia, Michael Lloyd, Anand Shah, Stacy Westerman, Faisal M. Merchant, Mikhael F. El-Chami
Right free wall (RFW) accessory pathways (AP) typically present anatomical challenges to ablation leading to high rates of procedural failure and recovery of AP conduction.
Patients with a diagnosis of Wolff-Parkinson-White Syndrome (WPW) and a manifest RFW AP undergoing an electrophysiology study (EPS) or an ablation at our center between 01/01/2008 and 08/01/2019 were identified from our databases using diagnosis codes and manual chart review.
Twenty-one patients with manifest RFW AP underwent EPS, all of which were targeted for ablation. Single procedure success rate was 19 / 21 (90.5%). Of the 19 successful cases, 4 (17.4%) patients were found to have recurrent right free wall pathway conduction at follow-up and each underwent a successful 2nd procedure (9.5%). Fluoroscopic and 3D electroanatomic mapping software was used in all cases to guide ablation. A 4 mm or 8 mm non-irrigated radiofrequency (RF) ablation catheter was used in 76% of cases while an 8 mm cryo-catheter was used in one case. More than one type of ablation catheter was used in four cases (16%). A steerable sheath was used in 68% of cases.
In a tertiary center, RFW AP ablation has high acute success (>90%) but approximately 21% of patients with initially successful ablation required a 2nd procedure for recurrence of pathway conduction. A combination of a large tip ablation catheter and a steerable sheath were used in most cases.
Key Words : Accessory Pathways, Right free wall pathways, Cryoablation, Radiofrequency ablation, Wolf Parkinson White.
Mikhael El-Chami, MD
Medical office Tower 12th Floor
550 Peachtree Street NE
Atlanta, Ga 30308
Catheter ablation of accessory pathways (AP) has proven to be a safe and effective first-line therapy for treatment of Wolff-Parkinson-White (WPW) Syndrome. Ablation success is, however, influenced by AP location. Right free wall (RFW) AP have higher rates of procedural failure and recurrence than their left-sided counterparts 1. Early ablation outcomes reported significantly greater radiofrequency (RF) applications, procedure durations, and fluoroscopy times with lower rates of success (75% to 85%) and higher rates of recurrence (14% to 35%) for RFW AP ablation 2-4. Poor catheter stability and tissue contact at the tricuspid annulus (TA) are responsible for these suboptimal outcomes 5, 6. Ablation via the superior approach (right internal jugular vein) 7, 8, incorporation of advanced diagnostic catheters 4, 9, 10, availability of electroanatomic mapping systems 11-13, and use of intra-cardiac echocardiography (ICE) guidance 14, 15 may improve the outcomes. In this manuscript, we present our center’s experience with RFW AP ablation.
A retrospective chart review of all patients undergoing electrophysiologic study (EPS) and ablation of manifest RFW AP at our institution between January 2008 and September 2018 was completed. Baseline demographics of all identified patients including age, gender, body mass index, comorbidities, medications, ejection fraction, and results of prior EPS were noted. Baseline electrocardiograms (ECG) were reviewed in addition to AP characteristics including pathway location, “malignant” vs. “non-malignant” properties, and effective refractory period (ERP). A malignant AP was defined as a:
a. Pathway with an ERP< 250 ms or
b. The presence of preexcited atrial fibrillation (AF) with an R-R interval < 250 ms or
c. The occurrence of unexplained syncope in patients with RFW AP.
Detailed analysis of catheter ablation techniques including access approach, procedure and fluoroscopy duration, mapping system and steerable sheath use, and ablation catheter type and energy were also recorded. Procedural success, recurrence, and complications were main outcomes of interest. Acute procedural success was defined as complete elimination of anterograde and retrograde conduction along the AP. The Emory University institutional review board (IRB) approved the study protocol.
Our EPS is typically a 3-catheter study utilizing: (1) high right atrial, (2) his bundle, and (3) right ventricular diagnostic catheters proceeding via right femoral venous access. A multi-electrode steerable catheter (Livewire™ St Jude Medical, Minneapolis, U.S.A) is at times used in lieu of the diagnostic right atrial catheter. Isoproterenol is used as needed to assess AP ERP and supraventricular tachycardia induction. Fluoroscopy and 3D mapping are used in all cases: NAVIX/ESI (St Jude Medical, Minnesota) was used in 24 procedures while CARTO (Biosense Webster, Irvine California) was used in the one remaining. Ablation catheter choice is subject to user preference and in most cases involved the combination of an 8 mm tip large curve non-irrigated RF ablation catheter (Blazer™ Boston Scientific, Marlborough, Massachusetts) through a steerable sheath (Agilis™ Abbott, Chicago, Illinois). However, a variety of alternative ablation catheters with and without a steerable sheath were also utilized including 4 mm tip non-irrigated RF ablation catheters (Blazer™ Boston Scientific, Marlborough, Massachusetts or Carto® Navistar® Biosense Webster, Irvine, California), 3.5 tip irrigated RF ablation catheters (Carto® ThermoCool® SF Biosense Webster, Irvine, California or FlexAbility™ Abbot, Chicago, Illinois), and 8 mm tip cryoablation catheter (FREEZOR MAX™, Medtronic, Fridley, Minnesota).
Indications for RFW AP Ablation:
Wolff-Parkinson-White (WPW) syndrome is defined as WPW ECG pattern and (1) the presence of palpitation, (2) documented spontaneous SVT, or (3) inducible SVT during EPS. During EPS, a RFW AP ablation attempt was performed (1) if the AP ERP was ≤ 250 ms, (2) if a SVT was inducible, or (3) if pre-excited atrial fibrillation with a short RR interval ≤ 250 ms was documented. In this study, each patient met at least one of these criteria and was therefore targeted for catheter ablation. Our ablation strategy relied first on localization of the earliest local ventricular signal along the tricuspid annulus during atrial pacing aiming to have maximal preexcitation. Also mapping during ventricular pacing or ORT was performed to localize earliest retrograde conduction in cases where mapping during maximal preexcitation proved challenging.
RF therapy was typically applied for 60 seconds at the effective site with additional lesions to follow unless no effect on AP conduction was observed after an initial 7-10 seconds. A power of 80 Watts was applied with an 8 mm tip large curve ablation catheter while a 35 to 40 Watts was used with an irrigated catheter. Cryo lesions were delivered at the effective site for 240 seconds with one additional lesion to follow unless no effect on AP conduction was observed within 30 seconds of lesion initiation.
Continuous data are expressed as the mean +/- standard deviation. Acute success rate refers to the ability to successfully ablate a patient’s RFW AP during the index procedure whereas total success rate accounts for all ablation procedure attempts for a given patient. The relation between patient/ablation characteristics and recurrence/failure was evaluated by using an unpaired Student t-test. All analyses were performed using IBM SPSS ver. 26 (2019; IBM SPSS Statistics for Macintosh, Version 26.0. Armonk, NY: IBM Corp).
Twenty-one patients underwent an EPS and ablation for symptomatic WPW syndrome patients with a RFW AP location. The mean age of patients was 39.3 +/- 13.7 (19 - 65) years with over two-thirds female and 7 with hypertension, 3 with coronary artery disease, and 3 with obstructive sleep apnea [Table 1].
Nearly all patients presented with palpitations (95.2%) and almost two-thirds had experienced syncope [Table 1]. The most common medical therapy prescribed prior to ablation was beta blockade with 11 patients on various agents, 1 using flecainide in combination with beta blockers, while 1 other patient was prescribed flecainide monotherapy. Nine patients were not on any medical therapy prior to ablation. Six patients (28.6%) had prior ablations before establishing with our center, one of whom had undergone 4 prior EP procedures.
Table 1. Patient Demographics
| Patient Characteristics |
Right Free Wall n=21 |
| Age [avg,years] |
39.3+/-13.7 [19-65] |
| Body Mass Index (kg/m2) |
30.6+/-7.0 [21-44] |
| Sex [% male] |
6/21 (28.6%) |
| Medical Conditions (n,%) |
|
| -Palpitations |
20 (95.2%) |
| -(pre)syncope |
13 (61.9%) |
| -Hyperlipidemia |
8 (38.1%) |
| -Hypertension |
7 (33.3%) |
| -Diabetes |
4 (19.0%) |
| -CAD |
3 (14.3%) |
| -OSA |
3 (14.3%) |
| -Asthma |
1 (4.8%) |
| Medicines (n,%) |
|
| -Beta Blocker |
11 (52.4%) |
| -None |
9(42.9%) |
| -Class IC AAD |
2(9.5%) |
| -CCB |
0 |
| Class III AAD |
0 |
| LVEF (%) |
56+/-8 [40-73] |
| Prior RF/Cryo Ablation |
6/21 (28.6%) |
Abbreviations: avg = average, CAD = coronary artery disease, OSA = obstructive sleep apnea, AAD = antiarrhythmic drug, LVEF = Left Ventricular Ejection Fraction, RF = Radiofrequency.
Right Free Wall Accessory Pathway Characteristics:
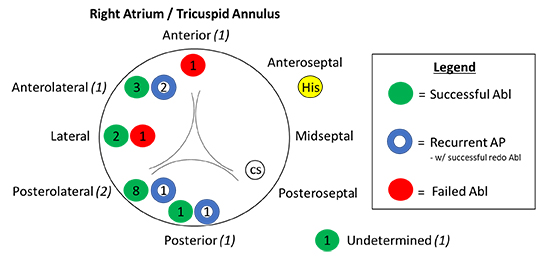
APs were localized fluoroscopically and with the aid of 3D mapping software and categorized in reference to their position about the TA: anterior (1), anterolateral (7), lateral (3), posterolateral (10), and posterior (3) with one undocumented site [Figure 1]. Five patients (23.8%) had malignant APs based on unexplained syncopal events (2), evidence of pre-excited atrial fibrillation with an R-R interval < 250 msec and or an AP ERP < 250 ms (1). The average AP Effective Refractory Period was 303 +/- 56 (210 – 440) msec.
Figure 1. Right Free Wall Accessory Pathway Location and Ablation Outcomes in 21 Patients Undergoing Index Ablation at Our Institution. Pathway Location (#) indicates number of patients with this type of pathway who had previously undergone EPS +/- ablation attempt at an outside institution. [Figure Adapted from Jan et al. BMC Cardiovascular Disorders (2020) 20:210]
Ablation Techniques and Outcomes:
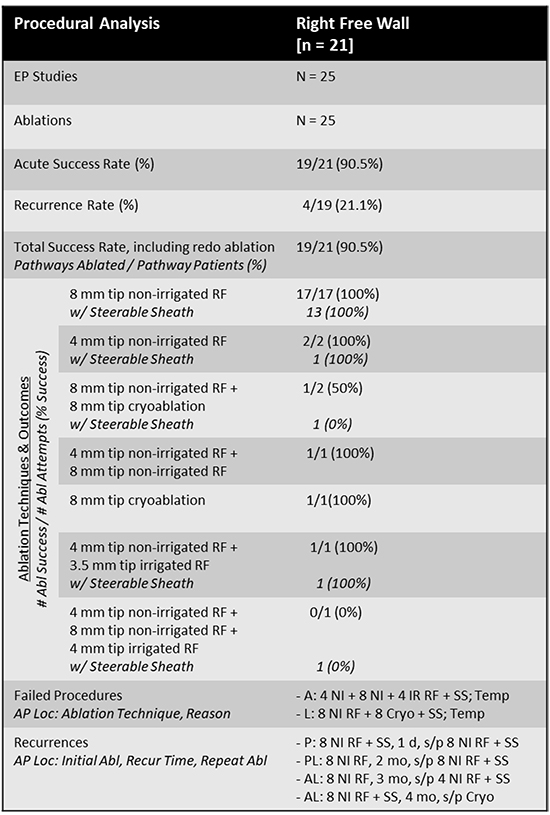
Ablation was acutely successful in 19/21 (90.5%) patients. Two patients had unsuccessful attempts at ablation during the index procedure, in both cases due to poor catheter stability and inadequate tissue contact. In one case, the AP was located in an anterior location and in the other was in the lateral RFW. [Figure 1]. Pathway conduction recurred in 4 out of 19 acutely successful cases (21.0%). All 4 patients underwent successful repeat attempt at ablation, resulting in a total of 25 EPS/ablation procedures in 21 patients. Procedures on average lasted 2:10 +/- 0:45 hours with 21.6 +/- 16 minutes of fluoroscopy time for 18 +/- 13 ablation lesions over a range of power 78.5 +/- 14 (50 to 100) watts. A variety of catheters were used, sometimes separately within the same procedure, to facilitate ablation success with all procedures proceeding via right femoral venous access. An 8 mm tip non-irrigated RF ablation catheter alone was used in 17 (68%) procedures and combined with a steerable sheath in 13 of those. A 4 mm tip non-irrigated RF ablation catheter alone was used in 2 (8%) procedures and combined with a steerable sheath in only one of those. An 8 mm cryoablation catheter alone was used in a single procedure (4%). The remaining 5 procedures utilized several of the above catheters in isolation [Table 2]. Three of those remaining procedures included use of a steerable sheath for a total of 17 (68%) procedures utilizing this technology. Two complications were observed: 1 pericardial effusion in a patient who underwent epicardial mapping and ablation of RFW and a posteroseptal (PS) AP as well as 1 access site hematoma.
Table 2. Electrophysiologic Study and Associated Ablation Techniques and Outcomes
Outcomes of Patients with Prior Ablation Attempts:
Six patients (28.6%) had previously undergone at least one EPS +/- ablation at outside institutions of variable RFW AP locations before establishing at our center. Four of these patients with 2 posterolateral, 1 anterolateral, and 1 undocumented RFW AP location underwent successful repeat ablation of at our site using an 8 mm tip non-irrigated RF ablation catheter with or without a steerable sheath. None have had signs of recurrence at follow-up. One of these six had already undergone 4 EPS with ablations at prior institutions and upon repeat attempt at our site was found to have both a posterior RFW AP and a PS AP necessitating extensive epicardial mapping before successful ablation of both sites. This posterior RFW AP then recurred a single day after the procedure, but was successfully ablated again with the same combination of an 8 mm tip non-irrigated RF catheter and steerable sheath the following month.
The final of these six patients had undergone an EPS as a child and on reattempt at our institution was found to have a non-malignant, anterior RFW AP. Attempt at ablation during this procedure failed to eliminate the AP despite use of both small (4 mm) and large (8 mm) tip non-irrigated catheters followed by 3.5 mm irrigated tip catheter with a steerable sheath.
Outcomes of Patients with No Prior Ablation Attempts:
Among the 15 patients without prior EPS/ablation attempts, three patients had recurrence of their RFW AP and one patient had a failed ablation.
The three patients with a recurrent pathway underwent successful repeat RFW AP ablation at 2, 4, and 11 months post-prior ablation. For a recurrent posterolateral RFW AP, an 8 mm tip non-irrigated RF catheter was used but this time combined with a steerable sheath. For two recurrent anterolateral RFW APs, one repeat ablation utilized an 8 mm tip cryoablation catheter while the other combined a 4 mm tip non-irrigated RF catheter with a steerable sheath. The failed ablation occurred in a patient with a lateral RFW AP despite attempts with both an 8 mm tip non-irrigated RF catheter and an 8 mm tip cryoablation catheter combined with a steerable sheath.
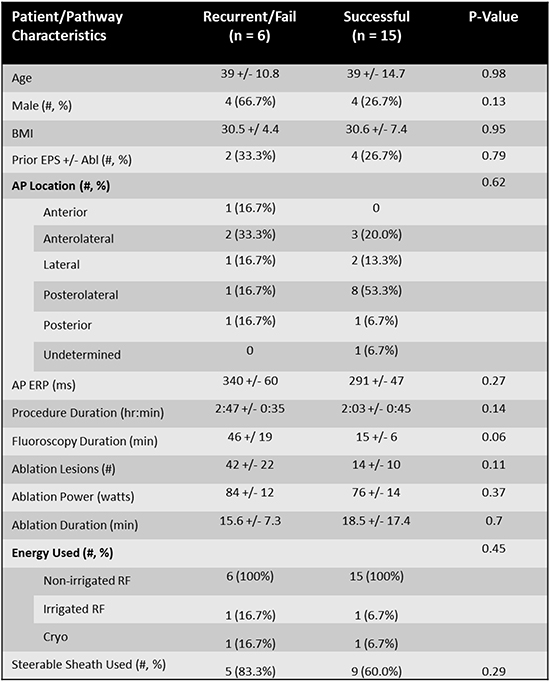
Comparing patients and AP ablation characteristics between recurrent/failed and successful RFW AP did not reveal any univariate predictors of AP recurrence or ablation failure. All patient and procedural characteristics were found to be similar [Table 3]. The median follow-up of these patients after index ablation at our center was 658 (IQR 127-1307) days with a range of 22 – 2828 days.
Table 3. Comparing Patient and Pathway Characteristics from those with recurrent or failed ablations to those with successful ablations at our institution
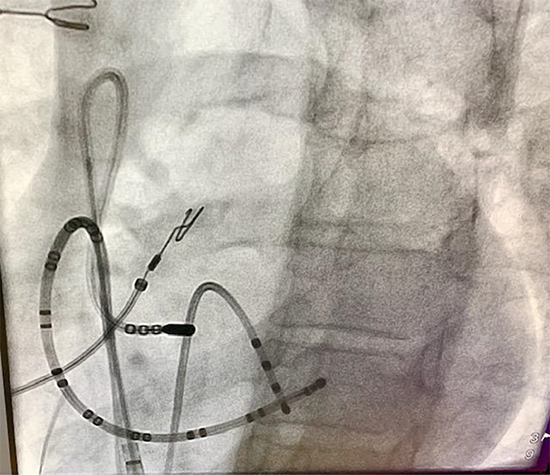
This is a single center study with a 21 patients undergoing 25 ablations for RFW AP. Almost 30% had failed an ablation in a different center indicating that these ablations are typically challenging procedures. The outcomes of these ablations in a contemporary setting demonstrate some improvements over historical reports (91% vs. 75-85%). Recurrence rates however remain concerning (20%) comparable to previously reported recurrence rates (14%-35%). Ablation of APs on the tricuspid annulus is fraught with challenges of catheter stability and poor tissue contact. The use of steerable sheath is helpful in achieving stability10. In this study ≈ 70% of procedures involved the use of a steerable sheath. The use of cryo catheter has the ability to adhere to the targeted ablation site and may overcome contact and stability problems encountered at the tricuspid annulus16. If the AP conduction is eliminated within 30 seconds, cryo application is typically continued for 240 seconds. In our practice, the use of cryo is often considered after failure of a large tip RF catheter. Its use as a first line ablation tool is uncommon mainly because of the higher recurrence rate with cryo therapy as compared to RF16. While the use of a superior approach (i.e. Right IJ) has been described to increase catheter stability and ablation success8, we have not adopted this approach in our practice. Forming a Loop with the catheter from a starting position in the RA/SVC junction and maneuvering the catheter to the tricuspid annulus [Figure 2] may accomplish similar stability to a superior approach. We have utilized this technique when the typical femoral approach has failed [Figure 2]. In our experience this approach works best with anterior RFW AP (AP located at 11 and 12 o’clock along the tricuspid annulus). The high recurrence rate seen in this study and in prior similar studies is probably related to the poor tissue contact and catheter stability, which result in reversible tissue damage. Another explanation for the higher failure rate of RFW AP ablation is the presence of multiple atrial insertions. Li et.al. reported on the presence of multiple atrial insertion sites in 10 patients with RFW AP. Ablation was successful only after targeting all atrial insertion sites17.
Figure 2. Catheter looped in the SVC down to the anterior tricuspid annulus to target a right free wall AP.
Several factors could explain the improved outcomes in this study compared to older published reports. The use of steerable sheath, large tip ablation catheters or irrigated catheters could help achieve more stability and a more effective temperature delivery. The availability of cryocatheter also provide another mean for success when traditional RF ablation fails. The use of intracardiac echocardiography could help overcome some anatomical barriers for success. The use of 3D mapping is paramount in localizing the area of interest especially in this setting where catheter stability is suboptimal.
Contact force catheter is a technology that could improve the outcomes of RFW AP ablation; an ablation that is fraught with challenges related to poor tissue contact and catheter stability.
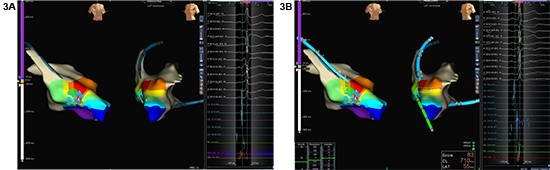
Figure 3. 3D mapping with ESI showing a lateral RFW AP with electrograms at the site of successful ablation (3A). Note disappearance of preexcitation after ablation (3B)
This is a single center study with a relatively small number of patients. Contact force catheter was not used routinely nor some advanced mapping techniques such as “open window” mapping. Nevertheless, ablation of RFW AP is relatively uncommon in adult patients and this study reports on contemporary ablation outcomes in patients with RFW AP.
Ablation of RFW AP in a contemporary setting is associated with good success rate but high recurrence rates. Future technologies and procedural technique should focus on means to increase catheter stability and effective temperature delivery.