Clinical Factors Relevant to the Recurrence of Atrial Tachyarrhythmia after Extensive Defragmentation Followed by Thoracic Vein Isolation
Kyoichiro Yazaki1, Masahiro Watarai1, Kenji Enta1, Mitsuru Kahata1, Asako Kumagai1, Koji Inoue1, Hiroshi Koganei1, Masato Otsuka1, Yasuhiro Ishii1
1Department of Cardiology, Cardiovascular Center, Ogikubo Hospital, Tokyo, Japan.
The efficacy of thoracic vein isolation (TVI), an approach to trigger atrial fibrillation (AF), for the management of AF has been established. Our goal was to identify the predictors for late recurrence of atrial tachyarrhythmias (ATAs), for which the patients and procedural and/or echocardiographic parameters were retrospectively analyzed. Although substrate modification in the atrium for the treatment of AF ablation remains controversial, the background associated with the outcome has not been fully investigated. We retrospectively studied 33 patients with paroxysmal AF and 21 with persistent AF undergoing defragmentation followed by TVI. We evaluated the late/early recurrences, defined as ATA at 3 months after/within the single procedure.
During a median follow-up period of 22 (11–37) months, 28 patients (52%) experienced a late recurrence. There was a higher incidence of late recurrences in the patients with disease durations of ≥12.4 months, which was the optimal cut-off point measured in the receiver operating characteristic curve analysis, or in those with left atrial diameter >50 mm or with earlier recurrences than the others (19% versus 72%, p=0.01; 0% versus 37%, p=0.02; or 13% versus 53%, p<0.0001 by the log-rank test, respectively). Moreover, there was a trend toward a higher atrial tachycardia (AT)-free rate in the patients with AF termination during the procedure (75% versus 54%, p=0.07 by the log-rank test). A multivariate analysis based on the Cox proportional hazard model showed that disease duration ≥12.4 months or early recurrence was highly associated with the outcomes (HR 3.72, 95%CI 1.42–12.79, p<0.006; HR 4.80, 95%CI 2.24–10.56, p<0.0001).
The AF duration and early ATA recurrence are the peri-procedural factors significantly relevant to the outcome after extensive defragmentation followed by TVI.
Key Words : Thoracic Vein Isolation, Atrial Fibrillation, Complex Fractionated Atrial Electrogram.
Correspondence to: KyoichiroYazaki, MD
Department of Cardiology, Cardiovascular Center, Ogikubo Hospital 3-1-24 Imagawa, Suginami-ku, Tokyo 167-0035, Japan.
Since ectopic beats observed within the pulmonary vein have been identified as the trigger for paroxysmal atrial fibrillation (PAF), pulmonary vein isolation (PVI) has become the first-line strategy for the treatment of paroxysmal AF. A second focal source for the trigger of AF is considered to be in the superior vena cava (SVC), which is derived from the embryonic sinus venosus and has arrhythmogenic properties. In the recent literature, it was reported that additional SVC isolation (SVCI) is one of the outcomes following atrial fibrillation ablation. In contrast, the complex fractionated atrial electrogram (CFAE) is a target for substrate modification in the atria, and the strategy involving this CFAE is called defragmentation. Although some researchers have demonstrated the efficacy of substrate modification, a large randomized-controlled trial refuted the concept that CFAE ablation plus PVI was superior to PVI alone. In addition, a recent meta-analysis also failed to exhibit significant improvements in the recurrence-free survival rate after PVI plus CFAE ablation compared with PVI alone. Therefore, it remains unclear whether CFAE marks the arrhythmia substrate, and its elimination results in relief from atrial tachyarrhythmias (ATAs). In addition, background characteristics of patients who have experienced ATA recurrences following PVI or SVCI plus CFAE ablation have not been fully investigated. Our strategy consisted of extensive defragmentation intended to eliminate the broad CFAE area before TVI, which was different from the previous protocol. We performed a retrospective analysis to reveal the background and predictors of ATA recurrence after this procedure.
A total of 75 patients who underwent catheter ablation for atrial fibrillation (AF) from December 2011 to October 2017 in our institute were considered for inclusion in this study. These patients were followed up for at least three months. Of these, 54 patients (33 patients with paroxysmal AF that spontaneously terminated within a week and 21 with persistent AF that lasted for more than a week), were retrospectively analyzed. The 21 remaining patients were excluded from the analysis because of non-inducibility of sustained AF during the procedure. These patients underwent PVI alone. Catheter ablation was needed for the presence of AF refractory to at least one anti-arrhythmic drugs (AADs), or the presence of intolerance to AAD for some reasons. All patients provided written informed consent. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Institutional Review Board and the Ethical Committee of Ogikubo Hospital.
Catheter Ablation Protocol
Before the ablation, multidetector computed tomography (MDCT) using a 64-slice CT scanner was performed for detailing the structure of the left atrium (LA) and ruling out an atrial thrombus. CT was performed using about 100 ml of contrast in tolerant patients and without contrast in patients with renal dysfunction. Transthoracic echocardiography was routinely performed within a month of the procedure. Anti-coagulation therapy was effectively started more than 1 month before the procedure and continued during catheter ablation. A decapolar catheter for mapping the coronary sinus was inserted via the jugular vein. SoundStar intracardiac echocardiography (ICE) (Biosense Webster Inc., Diamond Bar, CA, USA) was utilized for integrating geometric information with reconstructed CT images using CARTO (Biosense Webster Inc.), merged with a visual alignment technique. After a trans-septal puncture was made under ICE guidance, 2 long sheaths were introduced into the LA. Next, an initial 5,000 IU heparin was administrated, followed by repeated doses of heparin to maintain an activated clotting time (ACT) of more than 300 s at every 30-minute ACT measurement. The patients were deeply sedated with dexmedetomidine, by continuous administration for 10 minutes at a concentration of 6 µg/kg/h, followed by continuous infusion at 0.3−0.7 µg/kg/h. The sedation level was well controlled based on the value of the bispectral index. The ablation procedure consisted of CFAE ablation, followed by PVI and SVCI. We used a single Lasso catheter (Biosense Webster Inc.) as a mapping catheter, and a 3.5-mm open irrigated tipped catheter (ThermoCool or ThermoCool STSF; Biosense Webster Inc.) as an ablation catheter at a power of 30 W, with an upper temperature limit of 42 °C. The power was limited to 25 W during the SVCI. The catheter tip was irrigated with saline at a flow rate of 2 mL/min during mapping and at a flow rate of 8−15 mL/min during the ablation using the ThermoCool STSF, which was also irrigated at a flow rate of 2 mL/min during mapping and at a flow rate of 17−30 mL/min during ablation using ThermoCool. In patients with PAF, at the beginning of the procedure AF was induced using 10-beat high-frequency burst stimulation with a cycle length of 300 ms at a pacing output of 5 mA and 2 ms pulse duration via the coronary sinus (CS) catheter. This burst pacing was repeatedly conducted with 20 ms decrements until 2:1 atrial capture. If AF persisted, fast anatomical mapping and simultaneous detection of the CFAE area were initially performed using a multi-spline duodecapolar mapping catheter (PentaRay, Biosense Webster Inc.). On the other hand, in patients with PerAF, fast anatomical mapping and simultaneous detection of the CFAE area were initially performed using a PentaRay catheter. In both types of AF, the next ablation protocol was the same as that described below.
CFAE area was defined by the shortest complex interval (SCI) that CARTO employed for the detection of these areas, as previously described[1]. The successive peaks of two P-waves with amplitudes from 0.05 mV to 0.15 mV and intervals of 50–110 ms during a 2.5-s recording were counted. That number was identified as the interval confidence level (ICL), which was tagged with two distinct colors according to number, whose principle was described in a previous report[2]. We also manually tagged additional points that we determined as CFAEs. These points were visually continuous, low-voltage, and had multiple potential atrial signals. The CFAE ablation was conducted initially from the LA to the right atrium (RA) and coronary sinus (CS) and was continued unless AF was terminated or the CFAE area was entirely eliminated. When sinus restoration was achieved, re-induction was enacted by atrial burst pacing without isoproterenol or adenosine-triphosphate. CFAE ablation was performed again if AF was induced. When AF persisted despite the elimination of the entire CFAE area, intracardiac cardioversion was conducted. In case AF was converted to atrial tachycardia (AT), activation mapping was performed again, and subsequent catheter ablation was performed to eliminate AT.
PVI was performed after CFAE ablation, only when the potential in the pulmonary vein was observed. An isolation line was created in each pulmonary vein (PV) ostia, resulting in individual PVIs. The PVI was defined as the elimination of the PV potential, which was confirmed with a Lasso catheter. After the 30-minute waiting period following the creation of the isolation line, the absence of PV potential was reconfirmed. An exit blockade of the PV was confirmed by pacing from the Lasso catheter. Then, SVCI was performed. An isolation line was created 10 mm above the right atrium (RA)–SVC junction to avoid sick sinus syndrome. When the SVC potential was not recorded at the Lasso catheter, SVCI was not performed. Radiofrequency (RF)energy was delivered along the Lasso catheter in a point-by-point fashion using a maximum power of 25 W. The end point of the SVCI was the elimination of all SVC potentials recorded by the Lasso catheter and the absence of electrical conduction to the RA during pacing from the Lasso catheter located at the SVC. A 5-mA pacing was used to recognize the location of the phrenic nerve when ablating posterolateral aspects in the SVC. If the SVC potential remained at the point at which the right phrenic nerve was captured by pacing, very cautious RF application was started with an energy of 15 W, and the energy was increased by 5 W up to 25 W. At the end of the procedure, cavo-tricuspid isthmus ablation was performed in almost all of the cases, at the discretion of the surgeon.
Follow-up was based on outpatient clinic visits at 1 and 3 months and principally included one-day Holter monitoring via electrocardiography (ECG) before their visits. When the patient experienced palpitations or symptoms of ATA recurrence, Holter monitoring via ECG was again considered. After that, regular 1-to-3-month follow-ups were continued, which varied per the physicians’ discretion. In the first three months, the patients were treated with AAD in principle to prevent any early recurrence of ATA, with the exception of patients with concomitant diseases, such as sick sinus syndrome, severe hepatic disorder, or severe kidney disease. Whether AAD was discontinued after the blanking period was per the physician’s discretion, considering residual patient symptoms or recurrent ATA. Late recurrence was defined as ATA including AF, which lasted >30 s, and occurred outside the 3-month postoperative blanking periods, whereas early recurrences were those within the postoperative blanking period.
Categorical variables were expressed as mean ± standard deviation (SD) or median with [interquartile range (IQR)]. The student t-test and Wilcoxon test were used to compare the continuous variables between the two groups, and the Fisher exact test was used to evaluate the statistical differences in the categorical variables between the two groups. The long-term recurrence-free rate was evaluated using the Kaplan–Meier method, and statistical differences were confirmed using the log-rank test. All tests were considered statistically significant at a P-value less than 0.05. A Cox proportional hazard analysis was used to find the predictors for recurrence of ATA and to measure their hazard ratios (HRs) in a univariate or multivariate analysis. The area under the curve (AUC) found with receiver operating characteristic (ROC) curve analysis was used to assess the AF duration with respect to the ATA recurrence. The Youden index was used to identify the best cutoff value for the index of AF duration. All statistical analyses were performed using JMP® 13 (SAS Institute Inc., Cary, NC, USA).
In[Table 1], the baseline characteristics are presented for the two groups: recurrence of ATA (group A) or no recurrence of ATA (group B). In group A, the duration of AF, which was defined as the time from the first detection of AF to the ablation procedure, was significantly longer than those in the group B (42 months vs. 8 months, p = 0.016). There were no significant differences in patient sex, type of AF, CHADS2 score, left atrial diameter (LAD), or the prevalence of structural heart disease between the two groups. The number of patients who were resistant to AAD was 22 (92%) and 20 (74%) in groups A and B, respectively, whereas the rest of the patients showed AAD intolerance because of the presence of sick sinus syndrome or other reasons of discomfort, as described in[Table 1].
Table 1. Baseline characteristics
| Variables |
Recurrence |
No Recurrence |
|
(n=28) |
(n=26) |
| Mean age (yrs) |
62±10 |
58±11 |
| Male n (%) |
23(82) |
5(88) |
| Persitent AF, n(%) |
9(32) |
12(46) |
| AF Duration (months) |
42(14-56) |
8(5-39) |
| Comobidities |
|
|
| Hypertension, n (%) |
12(43) |
13(50) |
| Dibetes , n(%) |
4(14) |
5(19) |
| Structural Heart Disease |
10(36) |
7(27) |
| Coronary Artery Disease n(%) |
1(4) |
4(15) |
| Cardiomyopathy n(%) |
7(25) |
2(8) |
| Hypertension Heart Disease , n(%) |
0 |
1(4) |
| Valvular Heart Disease, n(%) |
2(7) |
0 |
| History of stroke , n(%) |
1(4) |
0(0) |
| Heart Failure , n(%) |
6(21) |
6(23) |
| CHADS 2 scrore |
|
|
| 0/1/2/3 n(%) |
11 (39)/11 (39)/5 (18)/5 (9) |
9 (35)/10 (38)/3 (12)/4 (15) |
| CHADS 2 VASc scrore |
|
|
| 0/1/2/3 n(%) |
6 (21)/9 (32)/6 (21)/7 (25) |
7 (27)/10 (38)/1 (4)/8 (31) |
| LVEF (%) |
59.7±11.6 |
58.6±11.6 |
| LAD (mm) |
42.2±6.9 |
42..2±5.8 |
| BNP (pg/ml) |
65.5(32.2-142.5) |
90.4(33.0-124.8) |
| Number of Ineffective ADD |
|
|
| 0/1/2/3 n(%) |
2 (8)/14 (58)/6 (25)/2 (8) |
6 (26)/8 (35)/6 (26)/3 (13) |
| AAD Treatment At Recurrence, n(%) |
24(86) |
15(58) |
* Value are mean ±SD, n(%),or median(IQR). AF=atrial fibrillation; CHADS2 Score=congestive heart failure, hypertension, age>75y,diabetes mellitus, prior stroke; LVEF= left ventricular ejection fraction LAD=left atria diameter ;BNP=Brain natriuretic peptide; ADD= Anti arrhythmic drug; ATA=atrial tachyarrhythmia
Procedural Characteristics
CFAE ablations were performed in all patients. PVI, SVCI, or CTI linear ablations were performed in 52 (96%), 29 (53%), or 49 (90%) patients, respectively. One patient experienced roof and bottom linear ablation, which resulted in posterior wall isolation because of the presence of non-PV foci from the posterior wall in LA without the use of adrenergic agents. AF termination was achieved in 28 (52%) patients during the procedure. There were no significant differences in procedure time, radiation exposure, or the number of applications in the two groups. In addition, there was no significant difference in the procedure duration between the patient with PAF and those with PerAF (299 ± 111 min versus 271 ± 49 min, p = 0.29). With regard to the peri-procedural complications, one patient experienced stroke after the procedure with tiny neural dysfunction. The patient had microinfarction in the occipital lobe and had temporary vision impairment, which was relieved after only one week of hospitalization. Another patient experienced coronary spastic angina in the right coronary artery during CTI linear ablation. However, there were no life-threatening adverse events in all patients as described in[Table 2].
Table 2. Procedural Characteristics
| Variables |
Recurrence |
No Recurrence |
P Value |
|
(n=28) |
(n=26) |
|
| Procedure Time (min) |
181±113 |
295±66 |
0.59 |
| Sinus Restorarion During the Procedure n(%) |
15(54) |
13(50) |
0.28 |
| Radiation exposure (mGy) |
1624(939-2363) |
1809(1430-3379) |
0.21 |
| Number of Application (times) |
67±21 |
72±22 |
0.39 |
| Details of ablation Procedure |
|
|
|
| CFAE ablation, n (%) |
28(100) |
26(100) |
0.39 |
| PVI, n (%) |
28(100) |
24(92) |
0.22 |
| SVCI, n (%) |
14(50) |
15(58) |
0.60 |
| CTI ablation, n (%) |
24(86) |
25(96) |
0.35 |
| Linerar ablation, n (%) |
1(4) |
0(0) |
1.00 |
| Complications |
|
|
|
| Pherenic Nerve Palsy, n (%) |
0(0) |
0(0) |
1.00 |
| Atrio-eshpagel fistula, n (%) |
0(0) |
0(0) |
1.00 |
| Cardiac Tamponade, n (%) |
0(0) |
0(0) |
1.00 |
| Bleeding, n (%) |
0(0) |
0(0) |
1.00 |
| Stroke, n (%) |
1(4) |
0(0) |
1.00 |
| Others, n (%) |
1(4) |
0(0) |
1.00 |
* Value are mean ±SD, n(%),or median(IQR). CFAE=Complex fractionated atrial electrogram; PVI=Pulmonary Vein Isolation; SVCI=superior vena cava Isolation; CTI=cavo-tricuspid isthmus
AF Duration and ATA Recurrence
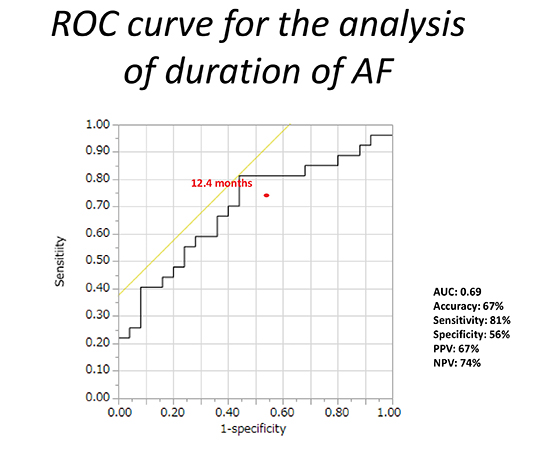
We evaluated the association between ATA recurrence and the duration of AF. When applying the ATA recurrence as a positive, the optimal cut-off point of the AF duration was 12.4 months by the ROC analysis. The AF duration predicted ATA recurrence with the AUC of 0.69, and when applying the ATA recurrence as a positive, the duration ≥12.4 months estimated 22 true positive, 14 true negative, 11 false positive, and 5 false negative, which resulted in the accuracy, sensitivity, specificity, positive predictive value, or negative predictive value of 67%, 81%, 56%, 67%, or 74%, respectively[Figure 1].
Figure 1. Receiver operating characteristic curves for atrial fibrillation-duration predictability indicated an area under the curve of 0.69. Optimal cut-off value for atrial fibrillation duration was 12.4 months, with an accuracy of 67%. ROC= receiver operating characteristics; AF= atrial fibrillation; AUC= area under the curve; PPV = positive predictive value; NPV = negative predictive value.
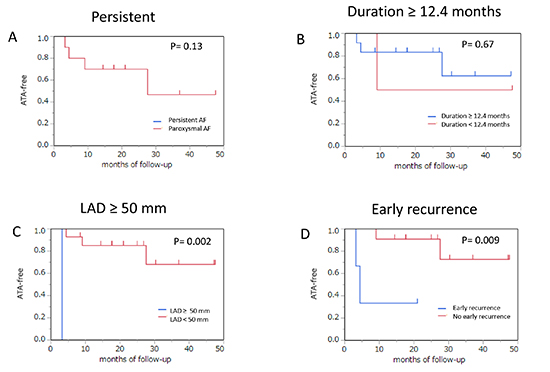
During a median follow-up of 22 months outside a blanking period of 3 months, 28 patients (51.9%) experienced ATA recurrence. Arrhythmia type of recurrence consisted of AF, AT, or both in 14 (26%), 9 (17%), or 5 (9%) patients, respectively. In a Kaplan–Meier event-free survival analysis, the entire ATA recurrence-free ratio was 61.0% and 46.8% at one year and two years, respectively. There was no statistically significant difference in the ATA-free rate between the patients with persistent AF (PerAF) and those with paroxysmal AF (PAF) (51% versus 35%, p = 0.88 by the log-rank test) [Figure 2A]. An AF duration ≥12.4 months, LAD ≥50 mm, or early ATA recurrence maintained sinus rhythm less frequently than that in the others (19% versus 72%, p = 0.01; 0% versus 37%, p = 0.02; or 13% versus 53%, p < 0.0001, respectively) [Figure 2B-D]. However, the AF duration ≥12.4 months and LAD ≥50 mm showed no difference in maintaining sinus rhythm in patients with PerAF (p = 0.41 and 0.48 by the log-rank test), whereas a significantly high recurrence rate was observed in those with PAF (p = 0.005 and 0.02 by the log-rank test). There were no significant differences in ATA recurrence between the patients with AF termination and those without during the procedure (35% versus 45%, p = 0.66 by the log-rank test), regardless of type of AF [Figure 3A]. However, the AT-free rate had a tendency to be greater in patients with sinus restoration during the procedure than the others (75% versus 54%, p = 0.07 by the log-rank test), whereas the AF-free rate was not significantly different between the two groups (72% versus 43%, p = 0.44 by the log-rank test) [Figure 3B,C]. At the end of the follow-up period, 24 patients (86%) in group A and 15 patients (58%) in group B still used AAD (p = 0.03). Although a small number of patients were evaluated, LAD ≥50 mm and early ATA recurrence lowered the late ATA recurrence-free rate (0% versus 68%, p = 0.002; and 33% versus 73%, p = 0.009 by the log-rank test) when including only patients without AAD [Figure 4].
Figure 2. The difference in atrial tachyarrhythmia-free survival is indicated between the patients with persistent atrial fibrillation (A), atrial fibrillation duration ≥12.4 months (B), left atrial diameter ≥50 mm (C), early recurrence of atrial tachyarrhythmia (D), and others. AF = atrial fibrillation; LAD = left atrial diameter.
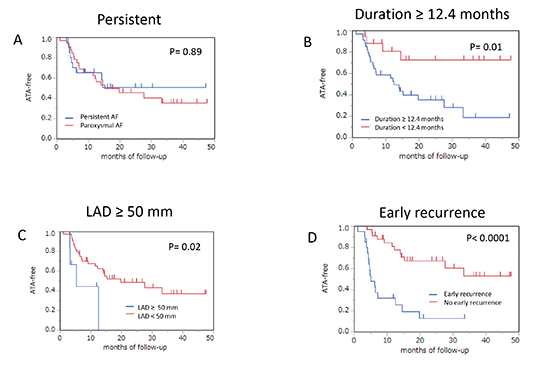
Figure 3. . In patients with AF termination during the procedure, ATA, AT, or AF-free survival rates are statistically superior to that in patients with failure of AF termination. The AT-free rate had a tendency to be greater if AF termination was achieved than the others
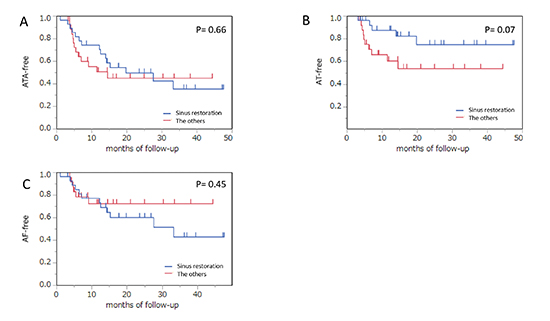
Figure 4. The difference in atrial tachyarrhythmia-free survival without AAD is indicated between the patients with persistent atrial fibrillation (A), atrial fibrillation duration ≥12.4 months (B), left atrial diameter ≥50 mm (C), early recurrence of atrial tachyarrhythmia (D), and others. AF = atrial fibrillation; LAD = left atrial diameter
The factors associated with ATA recurrence
In the univariate analysis, the following factors were associated with ATA recurrence: AF duration more than 12.4 months (HR: 3.72, 95% CI: 1.42–12.79, p = 0.006), and early recurrence of ATA (HR: 4.80, 95% CI: 2.24–10.56, p < 0.0001). Other factors, such as sex, AF termination, LAD, type of AF, or left ventricular ejection fraction, did not have an association with late ATA recurrence. The multivariate analysis demonstrated that AF duration or early ATA recurrence could independently predict late ATA recurrence (HR: 4.62, 95% CI: 1.71–16.23, p < 0.0015 and HR: 6.39, 95% CI: 2.86–14.65, p < 0.0001, respectively), when adjusting for these two covariates[Table 3].
Table 3. Risk Factors For Atrial Tachyarrhythmia Recurrence
| Variables |
Crude HR |
P Value |
Adjusted HR |
P Value |
|
(95%CI) |
|
(95%Cl) |
|
| Duration of AF <12.4 months |
3.72(1.42-12.79) |
0.006 |
4.62(1.71-16.23) |
<0.0001 |
| Sinus restoration during procedure |
0.84(0.40-1.81) |
0.66 |
|
|
| LAD >50mm |
3.30(0.94-9.09) |
0.06 |
|
|
| LAD >45mm |
1.33(0.60-2.86) |
0.48 |
|
|
| Early Recurrence of ATA |
4.80(2.24-10.56) |
<0.0001 |
6.39(2.86-14.65) |
<0.0001 |
| Female |
1.98(0.66-4.83 |
0.20 |
|
|
| Persistent AF |
0.94(0.40-2.04) |
0.88 |
|
|
| Left Ventricular ejection Fraction |
1.00(0.97-1.04) |
0.93 |
|
|
* LAD=Left Atrial diameter; AF=atrial Fibrillation; ATA=atrial Tachyarrhythmia; HR= hazard ratio.
In the 28 patients who experienced late ATA recurrence, 16 patients (57%) underwent a second procedure that consisted of CFAE ablation, followed by PVI and SVCI if needed. Electrical reconnection of the PV was observed in all the recurrent patients (100%), and they received successful PVI in the redo procedure. On the other hand, seven patients, who underwent SVCI in the first session, experienced the redo procedure. Four (57%) of them had electrical reconnection of SVC during the redo procedure. Four patients experienced ATA recurrences after the second procedure. Overall recurrence-free rate after a single or the second procedure was 70% (16 patients/54 patients).
In the present study, we elucidated a relatively long-term outcome after extensive defragmentation followed by TVI and revealed the background associated with the outcome. Approximately half of the patients with persistent AF/PAF experienced ATA recurrence, and about half of these patients underwent a second procedure, which resulted in a total recurrence-free rate of 70%. Our findings also suggested clinical factors relevant to the outcome, which included AF duration or the presence of early recurrence of ATA. These factors were independently associated with the outcomes following a single procedure.
Extensive Defragmentation Followed by TVI
Catheter ablation is the established approach for PAF. TVI, which includes PVI and SVCI, is the accepted procedure for not only PAF but also for PerAF because we know that the ectopic electrical activity from the PV or SVC is responsible for triggering and maintaining AF[3,4]. However, we could not clearly explain the underlying mechanism of the perpetuation of AF. CFAE, which was considered to be associated with AF maintenance, was a target for the AF ablation[5-7]. We initially performed extensive defragmentation because the CFAE area had decreased after RF application[8], and the efficacy of this strategy for rhythm control remained unclear in both types of AF. Currently, CFAE ablation is no longer accepted as an additional procedure to PVI, owing to the absence of overall benefit and the time-consuming nature of the procedure even for PerAF, revealed in some randomized-controlled studies[9-11] and meta-analyses[12],[13]. Indeed, our study demonstrated an entire recurrence-free rate of about 50% after a single procedure regardless of the type of AF. This recurrence-free rate was not superior to that of the PVI procedure alone, as reported previously[11]. Moreover, our strategy required long procedure times of 288 min. Therefore, we should necessarily optimize our strategy to increase the freedom from ATA-free rates after the single procedure – that is, more optimal CFAE areas should be explored, and the induction of AF should be more intensively intended with the injection of adrenergic agents.
Clinical Factors Relevant to the Outcome
Our findings suggested the two important clinical factors that allowed us to predict ATA recurrence following the procedure: long duration of AF and early recurrence of ATA. These factors were reasonably associated with the outcome following a single procedure. Moreover, AF termination during the procedure had a tendency to predict AT recurrence. In contrast, LA dimension, patient sex, or left ventricular ejection fraction, which was considered relevant to the outcome, in general, did not show a significant association with the outcome in our univariate analysis. The type of AF was generally associated with ATA recurrences following AF ablation. In the literature, non-paroxysmal AF was found to be an independent predictor for ATA recurrence, with a summarized risk ratio of 1.59 (95% CI, 1.38–1.82, p < 0.001)[14]. However, our results were inconsistent with those of the previous report. This is partially because of the strategy that mainly included substrate modification. It has been accepted that PVI reduces the CFAE area, which results in the avoidance of excessive RF application[8],[15],[16]. Hence, PVI was primarily the intended target in general. However, we performed CFAE ablations prior to PVI to conduct the broad substrate modification. In addition, electrical reconnection of the LA–PV was regrettably observed in all cases in the second session, specifically because the insufficient procedure to trigger elimination might result in poor outcomes. Considering these aspects, we speculated that strategic characteristics and incomplete PVI might contribute to the absence of a difference in the outcome between the two types of AF. In addition, we evaluated a small number of patients, which might have contributed to the same recurrence rate in both types.
In the present study, sinus restoration was not an independent predictor for estimating AF recurrence, but it did have a tendency to predict AT recurrence. To date, whether the stepwise approach provides better outcomes than PVI alone is still controversial[9],[17], and our results were in line with these reports.
Our results suggested the optimal cut-off duration of about 1 year and a higher late recurrence rate in patients with a history of a long duration of AF (>12.4 months) than that in the others. AF duration reflected the advanced structural and electrical remodeling in the atrium regardless of the presence of symptoms or difference in type of AF in general, mainly because of repeated stimulations of the LA, PVs, and extended substrate in the atrium[18],[19]. The last relevant factor was the presence of early ATA recurrence that was widely accepted as an indicator of the late ATA recurrence following AF ablation[20]. There was a strong association between the presence of early and late ATAs also in our study.
First, the strategy to predominantly eliminate the CFAE area and thereafter achieve TVI did not show better outcomes than the other approaches, as reported previously. More sophisticated approaches should be explored. Second, our study suggested the optimal cut-off of AF duration in the TVI with defragmentation procedure, which informed us of the optimal timing of AF ablation: earlier catheter ablation might lead to better outcomes. Third, our strategy suggested the superiority of AF termination during the procedure to the failure of that in terms of AT recurrence-free rate. One-year recurrence-free rate of AT in the patients with AF termination was 88%, while that in the patients without AF termination was 61%, suggesting that sinus restoration might contribute to the avoidance of the occurrence of secondary AT. Our strategy might predominantly modify the CFAE area responsible for the maintenance of AT, as described in the literature[21], although the AT mechanism was not clearly evaluated in this study. Fourth, in our study, seeking to perform durable PVI might result in better outcomes. In the present study, regrettably, electrical PV reconnection was observed in all cases undergoing the second procedure. In the past literature[22], an electrical reconnection of at least one PV was observed in all patients, and SVC-RA reconnection was observed in about half of the patients in the reoperation procedures, suggesting the difficulty of durable isolation. Finally, although our strategy required long procedure times and large amounts of radiation exposure as compared with the sole PVI procedure, life-threatening major complications were not observed in this study.
There are several unavoidable limitations to this study.First, this study has no control group for comparison. Therefore, the efficacy of this procedure was not truly evaluated. Second, this study was retrospectively conducted and included a relatively small number of patients, which could have introduced some selection bias. Third, non-negligible numbers of patients were treated with AAD during the follow-up period in group B, which might have affected the outcome. However, our results showed that there were more patients with AAD in group A than in group B, which suggested the possibility that the ATA might recur following the index procedure regardless of AAD use. Fourth, we only enrolled patients with inducible AF, and if not inducible, sole PVI was performed. This might contribute to selection bias, and it is speculated that high-risk patients who might have more substrate in their atria were included in this study. Finally, we performed AF inductions using atrial burst pacing without isoproterenol or adenosine-triphosphate infusion; that is, we could not truly confirm its non-inducibility, which is also a limitation of this study.
The AF duration and early ATA recurrence are the peri-procedural factors significantly relevant to the outcome after extensive defragmentation followed by TVI.