Left Atrial Strain Predicts Pro-Thrombotic State in Patients with Non-Valvular Atrial Fibrillation
Matteo Cameli1, Stefano Lunghetti1, Giulia Elena Mandoli1, Francesca Maria Righini1, Matteo Lisi1, Valeria Curci1, Cristina Di Tommaso1, Marco Solari1, Dan Nistor2, Annalaura Gismondi1, Marta Focardi1, Roberto Favilli1, Sergio Mondillo1
1Department of Cardiovascular Diseases, University of Siena, Siena, Italy.2Department of Internal Medicine V, University of Medicine and Pharmacy Targu Mures, Romania.
Atrial fibrillation (AF) has a high prevalence in the population and it is responsible for up to the 25% of the strokes in elderly people. The aim of our study was to assess the correlations of left atrial (LA) functional parameter, global peak atrial longitudinal strain (PALS), derived from speckle tracking echocardiography (STE), with transesophageal echocardiography (TEE) findings in patients with persistent AF undergoing TEE before electrical cardioversion or ablation procedures.
79 patients (58 males, 21 females) with persistent AF waiting for cardioversion were included in the study. The patients underwent conventional two-dimensional (2D) and 2D speckle tracking echocardiogram. PALS were measured in all subjects. Patients were divided into two groups according to the presence of reduced LA appendage (LAA) emptying velocity (<25 cm/s) and/or thrombus in the LAA at TEE examination.
Patients with reduced LAA emptying velocity and/or thrombus at TEE examination showed a significantly higher LA volume and increased E/E’ ratio. 4-chamber, 2-chamber and global PALS were significantly lower in patients with reduced LAA emptying velocity and/or thrombus (6.8 ± 2.0% vs. 27.5 ± 5.4%, P < 0.0001; 8.6 ± 3.5% vs. 29.4 ± 7.1%, P < 0.0001; 7.9 ± 3.2% vs. 28.5 ± 6.1%, P <0.0001, respectively). Among all variables analyzed, global PALS demonstrated the highest diagnostic accuracy (AUC of 0.92) and, with a cut-off value less than 8.1%, good sensitivity and specificity of 87% and 94%, respectively, to predict LAA thrombus and/or reduced LAA emptying velocity.
Among all parameters derived from transthoracic echocardiography, global PALS was the best predictor of LAA thrombus and/or reduced LAA emptying in patients with persistent non-valvular AF undergoing TEE before electrical cardioversion or ablation procedures.
Key Words : atrial fibrillation, transesophageal echocardiography, left atrial strain, speckle tracking echocardiography..
Correspondence to: Dr. Matteo Cameli, MD, PhD
Department of Cardiovascular Diseases, University of Siena.
Policlinico “Le Scotte”, Viale Bracci 1, 53100 Siena, Italy.
Atrial fibrillation (AF) is a major risk factor for systemic embolic events (SEE), accounting for up to 25% of all strokes in the elderly [1]. Ischemic strokes in AF are often more severe portending a higher risk of death and long-term disability. The risk of stroke and SEE is not homogeneous among AF patients as it is influenced by the presence of various clinical individual risk factors. The stroke risk in AF patients is graded as a continuum rather than as a categorization as assessed through the use of several risk scores[2],[3]. Recently, the CHADS2 scoring system has been expanded into the CHA2DS2-VASc score including additional clinical risk factors [4]. Left ventricular (LV) dysfunction, as assessed through imaging modalities, is the only non-clinical risk factor acknowledged in these scores, although many other findings provided by cardiac imaging may be potentially useful in further refining the individual thromboembolic risk [2]. Despite this emerging evidence, the role of transthoracic echocardiography (TTE) is currently restricted to the assessment of LV systolic function (as a component of CHADS2 and CHA2DS2-VASc scores) while transesophageal echocardiography (TEE) is still the gold standard for the evaluation of left atrial appendage (LAA) thrombus in candidates to cardioversion or ablation procedures [5].
There is a growing body of evidence suggestive of the association between CHA2DS2VASc score and TEE findings of high embolic risk, such as LA thrombus and spontaneous echo contrast (SEC) [6],[7]. The presence of LA or LAA thrombus is a powerful predictor of increased risk and stands as the only absolute contraindication to cardioversion or ablation; conversely SEC is associated with a high incidence of thrombus formation and thromboembolic events, but there is no consensus that such procedures are contraindicated in patients without coexistent LA or LAA thrombus identified [8],[9]. TEE, however, is semi-invasive and not so widely available, hence, given the central role of LA and LAA size, morphology and function, a non-invasive imaging modality capable of providing complete information also on these variables, beyond LV dimensions and function and heart valves, would be of great clinical interest and utility.
Speckle tracking echocardiography (STE), a TTE technique that uses standard B-mode images for the analysis of myocardial deformation, born for the analysis of LV function [10],[11] has extended its application to the study of LA [12],[13]. Considering the limitations of classical indices of LA function, assessment of LA strain by STE may represent a relatively rapid and easy-to-perform technique to explore LA function, due to its semi-automated and angle-independent nature and to its off-line processing [12]. Accordingly, the parameter of LA longitudinal strain (PALS) has been consistently reported as the first parameter useful for functional analysis of LA [12],[14],[15],[16],[17].
The aim of this study was to assess the correlations of LA deformation using STE with TEE findings in patients with persistent AF undergoing TEE before electrical cardioversion or ablation procedures.
All patients with persistent AF referred to our Echo Laboratory for a pre-procedural (electrical cardioversion or ablation) TEE from September 2015 to March 2016 were screened as potential candidate to the study. Common indications for TEE were: TEE-guided strategy of electrical cardioversion for AF ≥48 h of duration or with duration unknown, suboptimal anticoagulation in the previous 3 weeks in subjects scheduled for elective cardioversion, routine evaluation as per local protocol before AF ablation procedures. Eligible patients underwent both a TEE and conventional complete two-dimensional (2D) TTE and 2D speckle tracking analysis of LA.
The main exclusion criteria were AF duration <48 h, heart transplantation, valve repair or presence of prosthetic heart valve, more than mild mitral valve stenosis, previous closure of the LAA, presence of relevant valvular disease, technical inability to perform either TEE or STE analysis (i.e. inappropriate endocardial border definition of the LA).
All the patients provided their written informed consent for the participation to the study that complied with the declaration of Helsinki. The study was approved by our local ethic committee.
Echocardiographic evaluation
Echocardiographic studies were performed using a high-quality echocardiograph (Vivid 7, GE, USA), equipped with a 2.5 MHz transducer. Subjects were studied in the left lateral recumbent position. Measurements of LV and LA dimensions, LV ejection fraction, and diastolic LV filling velocities were made in accordance with current recommendations of ASE [18]. LV ejection fraction (EF), measured using Simpson’s method, was used as a standard index of global LV systolic function. M-mode measurements of mitral annular plane systolic excursion (MAPSE) was performed by placing the cursor perpendicular to the lateral site of the annulus [19] and was used as an index of LV longitudinal function.
Regarding LA dimension and size, the following measurements were obtained: LA AP diameter, measured by M-mode from the parasternal long- axis view, LA area, measured using planimetry in TTE apical four-chamber and two chamber views, and LA volumes using the area-length method, from the apical four and two chamber views. LA volumes were subsequently indexed by body surface area (BSA). Care was taken to exclude the pulmonary veins and LA appendage from the LA tracing.
For speckle tracking analysis, apical four- and two- chamber views images were obtained using conventional two dimensional gray scale echocardiography, during breath hold and with a stable ECG recording. Care was taken to obtain true apical images using standard anatomic landmarks in each view and not foreshorten the left atrium, allowing a more reliable delineation of the atrial endocardial border. Three consecutive heart cycles were recorded and averaged. The frame rate was set between 60 and 80 frames per second. The analysis of files recorded was performed off-line by a single experienced and independent echocardiographer, who was not directly involved in the image acquisition and had no knowledge of other echocardiographic parameters representing LV, LA and valvular structure and function, using a commercially available semi-automated two-dimensional strain software (EchoPac, GE, Milwaukee, USA). As previously described [12], and as stated in the current ASE/EAE Consensus [20], LA endocardial border is manually traced in both four- and two-chamber views, thus delineating a region of interest (ROI), composed by 6 segments. Then, after the segmental tracking quality analysis and the eventual manual adjustment of the ROI, the longitudinal strain curves are generated by the software for each atrial segment. Peak atrial longitudinal strain (PALS), measured at the end of the reservoir phase, was calculated by averaging values observed in all LA segments (global PALS), and by separately averaging values observed in 4- and 2-chamber views (4- and 2-chamber average PALS). In patients in whom some segments were excluded because of the impossibility of achieving adequate tracking, PALS was calculated by averaging values measured in the remaining segments. The reproducibility and the feasibility of the speckle tracking measurement of LA longitudinal strain has been previously reported by studies conducted in our echocardiographic laboratory [12],[13].
Regarding the TEE, images were acquired with a multiplane transoesophageal probe (2.9–7.0 MHz). LA thrombus was diagnosed by the presence of an echo-dense mass in the left atrium or the LAA. SEC was diagnosed by the presence of characteristic dynamic smoke-like swirling echoes in the LA or the LAA, distinct from background white noise caused by excessive gain [22], and was classified according to the classification (1 to 4+) proposed by Fatkin et al. [23]. Dense SEC was defined as grade 3+ or 4+. LAA flow velocities were assessed with a pulsed Doppler sample placed 1 cm from the entry of the LAA into the body of the LA. Maximum emptying and filling velocities were estimated from an average of five well-defined emptying and filling waves. Patients with maximum emptying and filling velocity ≤ 25 cm/s were classified as having low flow velocities.
Continuous variables are expressed as mean ± SD. A P value <0.05 was considered statistically significant. Pearson’s correlation coefficients were calculated to assess the relationships between continuous variables. Sensitivity and specificity were calculated using standard definitions, receiver operating characteristic curves were constructed and the area under the curve was calculated for the prediction of LAA thrombus and/or reduced LAA emptying velocity.
Analyses were performed using the SPSS (Statistical Package for the Social Sciences, Chicago, Illinois) software Release 12.0.
Among 86 patients screened, 79 (58 males) met eligibility criteria for the study. Five patients were excluded because of valvular AF (more than mild mitral stenosis and mitral valve replacement), and two because of impossibility to perform TEE (difficulties in intubation).Included patients were categorized in 2 groups according to the presence or absence of reduced LAA emptying velocity (<25 cm/s) and/or thrombus in the LAA at TEE examination. [Table 1] [Table 2]show the main clinical and echocardiographic characteristics of the two study groups.
Table 1. Main clinical data
|
LAA velocity < 25
|
LAA velocity >25 |
p Value |
|
(cm/s) and/or |
(cm/s), no |
|
|
thrombus |
thrombus |
|
|
(n = 28) |
(n = 51) |
|
| Clinical data |
|
|
|
| Age |
71.5 ± 9.2 |
70.1 ± 8.3 |
0.22 |
| Gender (% female) |
38 |
35 |
0.23 |
| Body Mass Index (Kg/m2) |
23.6 ± 4 |
22.3 ± 5.2) |
0.53 |
| Hypertension (%)
|
75 |
77 |
0.41 |
| Diabetes mellitus (%) |
29 |
27 |
0.33 |
| Hypercholesterolemia (%) |
64 |
68 |
0.25 |
| Current smoker (%) |
29 |
31 |
0.38 |
| Previous TIA/stroke (%) |
29 |
6 |
0.001 |
| CHA2DS2-VASc score |
3.5 ± 2.2 |
2.9 ± 2.1 |
0.01 |
| CHADS2 score |
3.1 ± 0.9 |
2.3 ± 1.1 |
0.02 |
| Medical therapy |
|
|
|
| VKA o NOAc (%) |
82 |
80 |
0.40 |
| Antiplatelet therapy (%) |
18 |
20 |
0.35 |
TIA: transient ischemic attack; VKA: vitamin-K antagonist therapy; NOAc: new oral anticoagulant therapy.
Table 2. Echocardiographic data
|
LAA velocity < 25
|
LAA velocity >25 |
p Value |
|
(cm/s) and/or |
(cm/s), no |
|
|
thrombus |
thrombus |
|
|
(n = 28) |
(n = 51) |
|
| Echocardiographic data |
|
|
|
| LA diameter (mm) |
45.2 ± 9.2 |
46.2 ± 8.6 |
0.55 |
| LA area (cm2) |
26.2 ± 7.2 |
24.6 ± 6.4 |
0.45 |
| LA volume (ml) |
47.2 ± 15.2 |
39.8 ± 13.5 |
0.01 |
| LV End-diastolic diameter (mm)
|
52.6 ± 6.1 |
53.1 ± 6.4 |
0.69 |
| LV End-systolic diameter (mm) |
37.2 ± 3.2 |
37.2 ± 3.2 |
0.82 |
| LV End-diastolic volume (ml) |
92.3 ± 18.1 |
96.5 ± 17.6 |
0.84 |
| LV Ejection Fraction (%) |
53.1 ± 7.3 |
53.2 ± 8.3 |
0.59 |
| LV relative wall thickness (cm) |
0.39 ± 0.19 |
0.35 ± 0.11 |
0.42 |
| LV† mass/BSA (g/m2) |
100.3 ± 10.2 |
96.5 ± 7.7 |
0.61 |
| CHADS2 score |
3.1 ± 0.9 |
2.3 ± 1.1 |
0.02 |
| E/E’ratio (cm/s) |
14.3 ± 7.1 |
11.6 ± 7.3 |
0.01 |
| MAPSE (mm) |
13.5 ± 4.6 |
13.8 ± 4.8 |
0.74 |
| 2-chamber PALS (%) |
8.6 ± 6.3 |
29.4 ± 8.1 |
< 0.001 |
| 4-chamber PALS (%) |
6.8 ± 5.5 |
27.5 ± 7.6 |
< 0.001 |
| Global PALS (%) |
7.9 ± 6.0 |
27.5 ± 7.6 |
< 0.001 |
LA: left atrium; LV: left ventricle; E: early transmitral flow velocity; E’: early diastolic mitral annular velocity; MAPSE: tricuspid annular plane systolic excursion; PALS: peak atrial longitudinal strain.
No significant differences were observed between groups regarding age, sex, body mass index and cardiovascular risk factors. The incidence of previous episodes of stoke and/or TIA was higher in patients with reduced LAA emptying velocity and/or thrombus, accounting for a significantly higher CHA2DS2VASc score.
There were no significant differences in anti-thrombotic therapy before TEE examination between the two groups.
As for echocardiographic data, there were no significant differences in any of LV function parameters between the two groups.
The patients with evidence at TEE examination of reduced LAA emptying velocity and/or thrombus showed a significantly higher LA volume and increased E/E’ ratio.
Moreover, 4-chamber, 2-chamber and Global PALS were significantly lower in patients with reduced LAA emptying velocity and/or thrombus (6.8 ± 2.0% vs. 27.5 ± 5.4%, P < 0.0001; 8.6 ± 3.5% vs. 29.4 ± 7.1%, P < 0.0001; 7.9 ± 3.2% vs. 28.5 ± 6.1%, P < 0.0001, respectively).
[Figure 1]
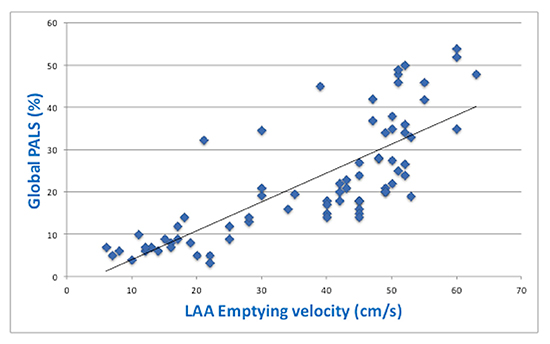
Moreover, close positive correlation between global PALS and the value of LAA emptying velocity was found (r=0.82, p<0.0001;[Figure 2].The correlation analysis was then performed after adjustment for age, severity of mitral regurgitation, LV EF and severity of diastolic dysfunction (different conditions that could determine a reduction of PALS), obtaining again a good grade of correlation between LA strain and LAA emptying velocity (r=0.77, p<0.0001).
Figure 1. Differences of the values of 4-ch PALS, 2-ch PALS and Global PALS in patients with reduced LAA emptying velocity/thrombus or normal LAA emptying velocity/No thrombus.
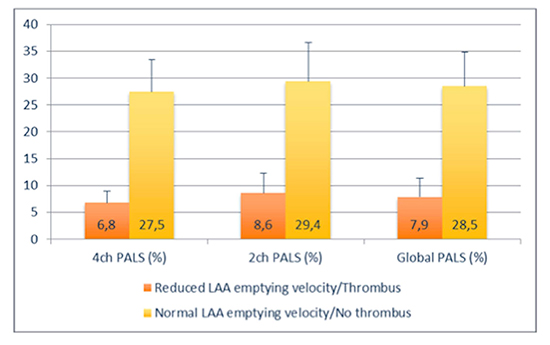
Figure 2. Correlation between global peak atrial longitudinal strain (global PALS) and LAA emptying velocity.
[Figure 3] and
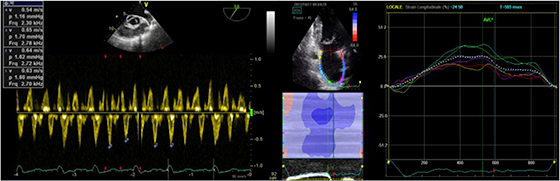
[Figure 4]display a representative case of each study group: the patient with evidence of reduced LAA emptying velocity at TEE examination shows a low value of PALS at STE analysis while the patient with normal LAA emptying velocity has higher PALS.
Among all transthoracic echocardiographic variables analyzed, global PALS demonstrated the highest diagnostic accuracy (AUC of 0.92) and, with a cut-off value less than 8.1%, good sensitivity and specificity of 87% and 94%, respectively, to predict LAA thrombus and/or reduced LAA emptying velocity.
Figure 3. Left panel: pulsed Doppler measurement of LAA emptying velocity (<25 cm/s) at TEE; right panel: measurement of PALS by STE in the same patient.
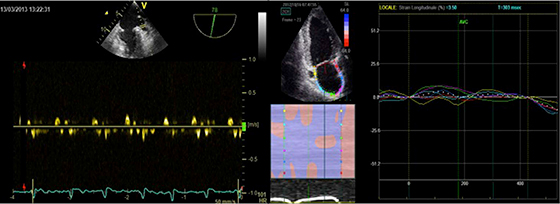
Figure 4. . Left panel: pulsed Doppler measurement of LAA emptying velocity (> 25 cm/s) at TEE; right panel: measurement of PALS by STE in the same patient.
Our results showed the correlation between reduced LA longitudinal strain, assessed by STE, with the presence of LAA reduced emptying velocity and/or thrombus on TEE examination in patient with persistent AF candidate to cardioversion or ablation. We were able to demonstrate a close positive correlation between reduced LA deformation in AF and echocardiographic findings linked to thromboembolism and stroke. Moreover, among all transthoracic echocardiographic variables analyzed, global PALS demonstrated the highest diagnostic accuracy (AUC of 0.92) to predict LAA thrombus and/or reduced LAA emptying velocity.
Two-dimensional TEE provides excellent characterization of the LA appendage and LA morphology because of the anatomic proximity of these structures to the esophagus. However, TEE findings are part of a dynamic process: LAA thrombus disappears either because of embolization or adequate anticoagulation; SEC appears or increases over time alongside with dilatation, fibrosis, and progressive atrial dysfunction or stunning; and LA appendage flow decreases with time as AF episode duration progresses or increases some weeks after an effective cardioversion. Therefore, a single TEE examination is like a single photograph of the LA milieu and may not be fully representative of past and future changes at this level. TTE with STE study given its noninvasiveness offers the advantage of being potentially used for serial assessments and evaluation of the patient devoid of the limitations, mainly tolerability, of TEE. Traditional TTE-derived parameters (LA diameter, LA area and LA volume), play a minor, if any, role in current risk scores since only LV ejection fraction <40% is included as a surrogate for congestive heart failure consistent also with the observation that a normal LV ejection fraction has been associated with the absence of LAA thrombus at TEE [24]. STE as an adjunct to standard TTE may allow for a more complete analysis of the LA thus providing valuable information on the pro-thrombotic state of LA and LAA deemed critical in some clinical settings of the decision-making in AF patients.
It has been widely demonstrated that AF is associated with tissue as well as overall atrial chamber structural remodeling in both animal models and humans [25]. This remodeling process is intrinsic to the pathophysiology of the arrhythmia and constitutes the substrate required for its maintenance [26]. Replacement of healthy atrial tissue with fibrotic tissue in AF can lead to reduced atrial contractile function and blood stasis and can potentially be mechanistically linked to the process of thrombus formation [5]. Moreover, atrial fibrosis may constitute tissue injury that can also contribute to the thrombogenesis cascade and this is supported by the finding that some AF patients have thrombi outside the LAA [27]. In addition, in a recent study, Akoum et al. [27] demonstrated that atrial fibrosis quantified using magnetic resonance imaging (MRI) appears to be an independent predictor of thrombus formation.
For these reasons, the measurement of functional echocardiographic parameters might provide more insights to the major site of thromboembolism in patients with AF.
Kuppahally et al. [28] demonstrated that LA wall fibrosis, as assessed by delayed enhancement-MRI, is inversely related to the strain and strain rate in patients with persistent AF and that patients with persistent AF had significantly greater delayed enhancement (as a marker for fibrosis) and decreased strain and strain rates, compared with patients with paroxysmal AF, supporting the concept that progressive remodeling occurs once AF develops[29].
In one of our investigations [30], we showed a strong relationship between the extent of LA fibrosis, assessed by and the value of LA strain, showing the higher sensibility of this new index respect to other echocardiographic parameters of LA size and function. Moreover, a recent prospective study demonstrated also the strong and independent power of global PALS to predict cardiovascular events [31]. Nonetheless, patients with permanent AF and history of stroke seem to have lower peak systolic LA strain rates assessed using STE during the reservoir phase compared with matched controls with no history of stroke [32].
Providência et al. [33] hypothesized that speckle tracking derived strain and strain rate could be of interest to evaluate the risk of LA stasis among patients with AF, reporting lower values of peak positive strain in patients with LAA thrombus or sludge.
Some main limitations of this study need to be acknowledged. Sample size was small and the findings need to be confirmed in larger cohorts, until then the results are hypothesis generating only. Exclusion criteria, mainly those regarding concomitant valvular disease, were rather restrictive but, in our opinion, necessary to avoid introduction of numerous selection biases. No data may be inferred with respect to the potential impact of anticoagulation therapy since patients were included regardless of the anticoagulation regimen (ranging from non-anticoagulated to effectively anticoagulated). Finally, the measurement of global PALS requires more capability and is contingent on the presence of adequate apical views; optimal 4 and 2-chamber apical view is required, permitting an easy delineation of endocardium border and, at the same time, avoiding LA foreshortening and auricular visualization. However, in this study, the feasibility was excellent at 95%. An additional limitation is that we used the current software for LV analysis to study the LA pattern strain because a dedicated software for LA analysis has not yet been released.
In conclusion, even if TEE remains the gold standard for the exclusion of LAA thrombus, global PALS, this new promising parameter derived from TTE, might have a role in thromboembolic risk assessment of patients with non-valvular AF. Further prospective studies are necessary to define its additive role in clinical practice.