Efficacy, High Procedural Safety And Rapid Optimization Of Cryoballoon Atrial Fibrillation Ablation In The Hands Of A New Operator
Eberhard Scholz1, Patrick Lugenbiel1, Patrick A. Schweizer1,2, Panagiotis Xynogalos1, Claudia Seyler1,2, Edgar Zitron1,2, Rüdiger Becker1,3, Hugo A. Katus1,2, Dierk Thomas1,2
1Department of Cardiology, Medical University Hospital, Im Neuenheimer Feld 410, 69120 Heidelberg, Germany .2DZHK (German Centre for Cardiovascular Research), partner site Heidelberg/Mannheim, Im Neuenheimer Feld 410, 69120 Heidelberg, Germany.3Present address: Department of Cardiology, Angiology, Nephrology and Intensive Care, Klinikum Wolfsburg, Sauerbruchstr. 7, 38440 Wolfsburg, Germany.
Cryoballoon (CB) ablation is successful in eliminating atrial fibrillation (AF).
The purpose of this study was to assess procedural efficacy and safety of CB ablation performed by a newly trained operator.
Forty patients with documented paroxysmal AF (58 ± 11 years, 26 male) undergoing CB catheter ablation were prospectively enrolled.
Electrical pulmonary vein (PV) isolation was achieved in all patients (156 PVs). The primary end point (PV isolation using CB only) was reached in 31 patients (92% PV isolation, 144/156 PVs). In the remaining 9 patients (12 PVs), additional single point cryofocal ablations were required to achieve isolation of all veins (LSPV, n = 5; LIPV, n = 3; LCPV, n = 2; RSPV, n = 1; RIPV, n = 1). There was no vascular access complication, pericardial effusion/tamponade, stroke/transient ischemic attack, phrenic nerve palsy, acute PV stenosis, or atrioesophageal fistula. The procedure duration decreased with experience by 30% from 155 min during the first 10 procedures to 108 min (final 10 treatments). Similar effects were observed with fluoroscopy time (-57%; from 28 min to 12 min), dose area product (-66%; from 22 Gy x cm2 to 8 Gy x cm2), CB time in the left atrium (-24%; from 99 min to 75 min), and cryoenergy delivery time (-19%; from 83 min to 67 min), when comparing cases #1-10 to cases #30-40.
CB ablation of AF is effective and safe in the hands of a new operator. Procedure and fluoroscopy times decrease with user experience.
Key Words : Atrial Fibrillation, Catheter Ablation, Cryoballoon, Fluoroscopy, Learning curve.
Correspondence to: Dierk Thomas,
Department of Cardiology, Medical University Hospital Heidelberg,
Im Neuenheimer Feld 410, 69120 Heidelberg,
Germany.
Catheter ablation is effective in eliminating symptomatic atrial fibrillation (AF).1 Ablation strategies include pulmonary vein (PV) isolation, the creation of linear lesions, and ablation of complex fractionated atrial electrograms, autonomic ganglia, or electrical rotors.2-7 Advanced technologies such as remote robotic navigation (RRN), visually-guided endoscopic balloon ablation, and cryoballoon (CB) ablation have evolved to improve catheter manipulation, ablation efficacy, and procedural safety while reducing procedure times, fluoroscopy exposure and physical demands (including manual dexterity).8-14 In particular, CB ablation represents a simplified and commonly used technique that achieves anatomical PV isolation without the need for a three-dimensional mapping and navigational system. Additional advantages of the CB approach include the introduction of a dedicated inner-lumen circular mapping catheter (Achieve; Medtronic, Minneapolis, MN, USA) that serves as supporting guidewire and allows for real-time assessment of electrical PV isolation during ablation.15-19 Conversely, CB ablation is associated with a considerable prevalence of phrenic nerve palsy (PNP) that requires caution during cryoenergy application.8,14,20 Given the growing number of centers offering catheter ablation of AF and considering the increasing clinical use of CB technology for AF ablation, we sought to assess CB ablation in the hands of a newly training operator within an experienced high-volume electrophysiology environment. To this end, 40 patients presenting with symptomatic paroxysmal and drug refractory AF were prospectively enrolled, and acute ablation efficacy, procedural characteristics, and safety were evaluated.
The study protocol was approved by the ethics committee of Heidelberg University (Heidelberg, Germany), and the study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Written informed consent was obtained from all study patients. This study was registered at www.clinicaltrials.gov (NCT01448265). The procedural methods used in this investigation were standard-of-care during the operation of this study.
The study population consisted of 40 consecutive patients between 18 and 75 years referred for catheter ablation of symptomatic paroxysmal AF despite treatment with ≥1 antiarrhythmic drug at the Medical University Hospital of Heidelberg (Germany). Patients with prior AF ablation procedures, left atrial size ≥ 50 mm, left atrial thrombus, irregular PV anatomy as assessed by pre-procedural transesophageal echocardiography, left ventricular ejection fraction < 40%, moderate to severe valvular heart disease or previous heart valve replacement, or contraindication for anticoagulation medication were excluded from this study.
The primary operator (E.S.) had no previous experience in CB AF ablation, allowing for an unbiased learning curve assessment and review. Of note, the operator did exhibit expertise in the employment of focal catheter ablation, including radiofrequency ablation of patients with AF and other cardiac arrhythmias.
Electrophysiology Study Procedures
Oral anticoagulation (OAC) was discontinued prior to ablation until an international normalized ratio (INR) < 2 was reached. Bridging of OAC was performed using low-molecular weight heparin, and transesophageal echocardiography (TEE) was conducted within 24 h to assess PV anatomy and to exclude left atrial thrombus. TEE studies were performed using multiplane 5-MHz TEE probes. For visualisation of the PV anatomy a transducer angle of 0° was generally employed. When necessary, manipulation of the probe and use of Doppler color ultrasound were applied. No additional preprocedural cardiac imaging was done. Procedures were performed in the postabsorptive state under conscious sedation and analgesia with appropriate doses of midazolam, fentanyl, and/or propofol. Vital parameters such as blood pressure and oxygen saturation were monitored throughout procedure. Catheter entries were accessed via right and left femoral veins, respectively. A steerable diagnostic quadripolar catheter (Xtrem, ELA Medical, Sorin Group, Munich, Germany) was positioned in the coronary sinus. A fluoroscopy-guided, double transseptal approach using a Brockenbrough needle was then followed in all procedures. The surface ECG and bipolar endocardial electrograms were monitored continuously and stored using a digital amplification and recording system (LabSystem, Bard Electrophysiology Division C. R. Bard, Lowell, MA, USA). During the intervention, a continuous titrated infusion of heparin was maintained to achieve an activated clotting time (ACT) between 300 and 400 s.
Cryoballoon Ablation of AF
For CB ablation a steerable transseptal sheath (12 F, FlexCath™, Medtronic, Minneapolis, MN, USA) was introduced into the left atrium (LA) over a guidewire to steer the Arctic Front™ CB (28 mm diameter; Medtronic). The circular mapping catheter (20 mm diameter, Achieve™, Medtronic) was inserted through the lumen of the CB. An additional SL1 sheath (St. Jude Medical, St. Paul, MN, USA) was placed as part of the study protocol to allow for cross-over to regular CB guidewire and simultaneous placement of a circular mapping catheter if stable balloon positions could not be obtained. Transseptal sheaths were constantly perfused with heparinized 0.9% saline. After transseptal puncture, a patient weight-adjusted unfractionated heparin bolus was given. The CB was manoeuvred to all PV ostia. Balloon position and the degree of PV occlusion were evaluated by injection of radiopaque contrast agent diluted in 1:1 ratio with 0.9% saline. As a standard, two deliveries of cryoenergy were applied for 5 min per application to each PV, which was a commonly used dosage for the first generation CB. Before targeting the right-sided PVs, the steerable quadripolar catheter (Xtrem, ELA Medical, Sorin Group) was positioned in the superior vena cava for continuous phrenic nerve stimulation (10 V, 2.9 ms; ~50 mA) during cryoenergy application. Delivery of cryoenergy was terminated immediately upon loss of capture during phrenic stimulation. LA-PV and PV-LA conduction blockade (entrance and exit block) was confirmed by complete elimination of PV potentials or by dissociated electrical PV activity and by using pacing manoeuvres aiming for capture within the PV, respectively. If electrical PV isolation could not be achieved using the CB alone, additional single point cryofocal ablations were applied using a focal cryocatheter (Freezor Max®, Medtronic) until the respective PV was isolated.
Transthoracic echocardiography was performed immediately after the procedure and on the next day to exclude pericardial effusion. Following removal of sheaths and pressure taping, patients received unfractionated heparin targeting a partial thromboplastin time of 40 to 60 seconds until an INR of 2 to 3 was achieved by oral anticoagulation that was resumed 24 to 48 hours after the procedure. OAC was maintained for at least three months after the procedure and according to current guidelines thereafter. Prior to discharge, 12-lead ECG and 24-hour Holter ECG recordings were performed.
Continuous variables are expressed as mean ± standard deviation (SD). Procedural data are provided as median with 25th and 75th percentiles. For between-group comparisons, the unpaired Student’s t-test (two-tailed test) was used. Categorical variables are described as count and percentage. A P value <0.05 was considered statistically significant.
Between December 2011 and July 2013 a total of 40 patients (26 male, 14 female) suffering from paroxysmal atrial fibrillation (mean EHRA score, 2.5 ± 0.6) were prospectively included (Table 1). The mean time from AF diagnosis to catheter ablation was 61 ± 63 months. Prior to ablation, antiarrhythmic drug therapy had remained ineffective with 1.3 ± 0.8 antiarrhythmic drugs applied per patient. The mean left atrial diameter was 40 ± 3.9 mm, and structural heart disease was present only in a minority of the patients (Table 1).
Table 1. Patient Characteristics
|
Study population (n = 40) |
| Age (years; mean ± SD) |
58 ± 11 |
| Male (n; %) |
26 (65) |
| Body mass index (kg / m2; mean ± SD) |
29 ± 5.6 |
| Paroxysmal AF (n; %) |
40 (100) |
| Time since AF diagnosis (months; mean ± SD) |
61 ± 63 |
| Failed class I through IV AAD (mean ± SD) |
1.3 ± 0.8 |
| Failed class I and III AAD (mean ± SD) |
0.6 ± 0.7 |
| EHRA score (mean ± SD) |
2.5 ± 0.6 |
| CHA2DS2-VASc score (mean ± SD) |
1.4 ± 1.1 |
| Prior stroke or TIA (n; %) |
0 |
| Left atrial diameter (mm; mean ± SD) |
40 ± 3.9 |
| Mildly reduced LVEF (n; %) |
4 (10) |
| Hypertension (n; %) |
21 (53) |
| Concomitant heart disease (n; %) |
9 (23) |
| Coronary artery disease (n; %) |
6 (15) |
| Dilated cardiomyopathy (n; %) |
2 (5.0) |
| Hypertrophic cardiomyopathy (n; %) |
1 (2.5) |
| Diabetes mellitus (n; %) |
1 (2.5) |
| Chronic obstructive pulmonary disease (n; %) |
3 (7.5) |
AAD, antiarrhythmic drug; AF, atrial fibrillation; EHRA, European Heart Rhythm Association; LVEF, left ventricular ejection fraction; TIA, transient ischemic attack
Feasibility and Procedural Safety
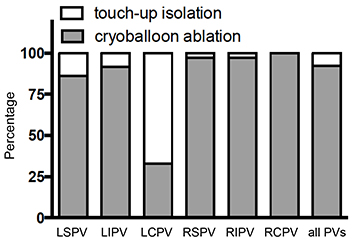
Within these 40 patients, 156 PVs were identified by angiography and successfully isolated during the ablation procedure (left superior (LS)PV, n = 37; left inferior (LI)PV, n = 37; right superior (RS)PV, n = 39; right inferior (RI)PV, n = 39; left common (LC)PV, n = 3; right common (RC)PV, n = 1). 144 (92%) of the veins could be isolated using the cryoballoon and circular mapping catheter setup
(Figure 1). For the isolation of the remaining 12 (8%) veins (9 patients), touch-up isolation using an additional cryofocal catheter was necessary. LSPV, LIPV, LCPV, RSPV and RIPV required 5, 3, 2, 1 and 1 touch-up cryoenergy deliveries, respectively
(Figure 1). A mean number of 11.6 additional energy applications/patient with a mean energy delivery time of 12.1 minutes were applied to achieve PV isolation. Among the group of 40 patients there were no reported complications with respect to vascular access site, pericardial effusion/tamponade, stroke, transient ischemic attack, phrenic nerve palsy, PV stenosis, and atrio-oesophageal fistula (Table 2).
Table 2. Complications
|
Number of patients |
| Vascular access complications |
0/40 (0%) |
| Pericardial effusion / tamponade |
0/40 (0%) |
| Stroke / transient ischemic attack |
0/40 (0%) |
| Phrenic nerve palsy |
0/40 (0%) |
| Acute pulmonary vein stenosis |
0/40 (0%) |
| Atrio-oesophageal fistula |
0/40 (0%) |
Figure 1. Successful Pulmonary Vein (PV) Isolation Using the Cryoballoon (CB) Technique The percentages of veins isolated using solely the cryoballoon and of veins requiring touch-up ablations are indicated for each PV. In 40 patients (156 PVs), a total of 12 PVs could not be isolated using the CB alone. Abbreviations: LSPV, left superior PV; LIPV, left inferior PV; LCPV, left common PV; RSPV, right superior PV; RIPV, right inferior PV; RCPV, right common PV
Performance of the Circular Mapping Catheter
PV isolation using the cryoballoon with the achieve mapping catheter has been performed similar to the technique described previously by Chun et al.17 The additional use of a regular guidewire for CB positioning was not required during the course of the study. Cryoenergy was delivered as soon as appropriate occlusion of the PV had been confirmed by angiography
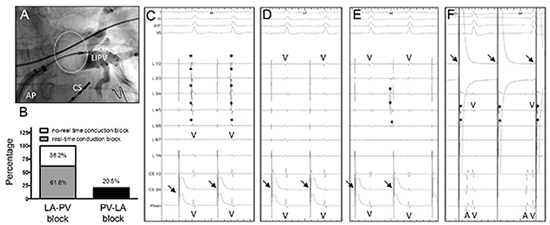
(Figure 2A). The circular mapping catheter was retracted towards the ostium of each PV prior to cryoablation to visualize PV spikes. Real-time LA-PV conduction block during energy delivery could be observed in 61.8% of isolated veins
(Figure 2B). In all other veins (38.2%), no PV spikes could be visualized prior to energy delivery, probably due to the distance between the mapping catheter and the PV ostium. In these cases, LA-PV conduction block was verified after ablation by withdrawing the mapping catheter to more proximal positions inside the PV ostium. An exemplary case before and after successful ablation is displayed in Figure 2C - 2F). In addition to LA-PV blockade, PV-LA conduction block was analysed by stimulating each vein close to the PV ostium using a rectangular voltage step (5 V, 1.9 ms; ~25 mA). PV capture was assumed when the stimulus was followed by a small sharp near field signal in the circular mapping catheter
(Figure 2f). Despite careful repositioning, PV capture and hence PV-LA conduction block could only be observed in the minority of cases (20.5%;)
Figure 2B.
Figure 2. Procedural Findings Obtained During Cryoballoon (CB) Ablation of Atrial Fibrillation (AF) (A) Angiogram of a left inferior pulmonary vein (LIPV) in anterior-posterior projection, revealing proper occlusion of the vein by the CB (B) Percentage of real-time conduction block verified using the circular mapping catheter. Left atrium (LA)-pulmonary vein (PV) conduction block (either in real-time or post ablation) could be documented in all cases, whereas PV-LA conduction block was obtained in 20.5% of the PVs only. (C-F) Typical electrogram recordings before and after isolation of a PV using the CB (C) Prior to ablation during electrical stimulation in the coronary sinus (indicated by arrows), PV spikes (indicated by asterisks) are clearly visualized by the circular mapping catheter right before a ventricular far-field signal (V) (D) After successful isolation, PV spikes are eliminated unmasking the ventricular far-field signal (V) in all poles of the circular mapping catheter (E) A dissociated PV signal (indicated by asterisks) was observed occasionally, indicating complete conduction block between the PV and the LA (F) Electrical stimulation between the first and second electrode of the circular mapping catheter (indicated by arrows) results in PV capture reflected by a small PV signal following the stimulation artefact (indicated by asterisks). Successful PV stimulation not exerting atrial electrical activity demonstrated PV-LA conduction block. A, atrium; AP, anterior-posterior projection; CS, coronary sinus catheter; V, ventricle
New Operator’s Learning Curve
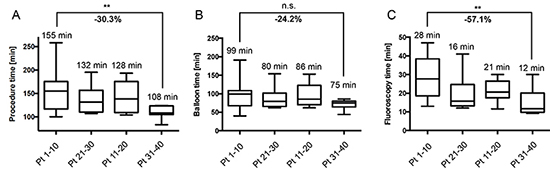
As a main goal of the present study, procedural data including procedure time, balloon time and fluoroscopy time were determined to assess the learning curve of a newly trained operator. When comparing median procedure times of the first ten to the last ten patients of the series, a reduction by 30.3% from 155 to 108 minutes (P < 0.01) could be observed
(Figure 3A). For the balloon time a reduction by 24.2% was apparent
(Figure 3B). However, this reduction was not statistically significant (P = 0.09). With median values of 28 minutes for the first ten and 12 minutes for the last ten procedures, fluoroscopy time could be significantly reduced by 57.1% (P < 0.01; Figure 3C). Similar effects were observed for fluoroscopy dose area product (65.6% reduction; from 22.4 Gy x cm2 to 7.7 Gy x cm2) and cryoenergy delivery time (-19.4%; from 82.5 min to 66.5 min), albeit without statistical significance (P > 0.05). Interestingly, conversion to touch-up isolation did not negatively correlate with increasing experience. With in the first, second, third and fourth group of ten patients, 3, 1, 7 and 1 touch-up isolations were necessary, respectively.
Figure 3. Procedural Data, Analysed in Four Subgroups of Ten Consecutive Patients Among the Study Cohort of 40 Patients (A) Median procedure times decreased by 30.3% between the first and the last ten patients. (B) Similarly, balloon time revealed a reduction from 99 to 75 minutes (-24.2%). (C) Median fluoroscopy time showed a marked decrease from 28 to 12 minutes (57.1% decrease). Median, minimal and maximal as well as the 25th and 75th percentiles are displayed for the first, second, third, and forth subgroup of patients, respectively. Pt, patient; ***P < 0.01; n.s., not significant (P > 0.05)
We demonstrate high acute efficacy and safety of CB AF ablation when performed by a newly trained CB operator. Furthermore, rapid technical optimization was illustrated by improvement of procedure duration (-30%), ablation time (-21%), and fluoroscopy time (-57%) when the first 10 procedures where compared with the final 10 patients in the present cohort of 40 study subjects. This work provides incremental information regarding efficacy and safety of CB ablation over previous studies that were mostly carried out by electrophysiologists with significant prior CB technology experience. Here, feasibility of CB ablation was apparent early during application of the technology by a first-time user in a real-world setting.
Comparison of a New Operator’s CB Ablation Procedures with Previous Clinical Experience
During the first 40 cases, acute success (i.e., electrical PV isolation) was reached in 100% of cases, which is in line with first-generation CB data provided by a meta-analysis (98%).21 The CB and circular mapping catheter alone were sufficient to achieve isolation in 92% of the veins in the present work, compared with 93% in the combined literature.21 These findings highlight that effective isolation was feasible already during early use of the CB. Currently, PV isolation rates using only the CB are as high as 99-100% when the second generation balloon is used.22-27
The circular mapping catheter reliably served as guidewire for CB positioning in all cases. Real-time visualization of PV signals during ablation was possible in 62% of veins, which is similar to ongoing experience with the second generation CB (53-76%)22,23,28 and exceeds earlier results with the first generation balloon (47-55%).15-17,19,22,23
Median procedure times decreased from 155 to 108 minutes, which appears acceptably short when compared with 108 to 371 minutes duration in other reports using the first generation CB.11 By contrast, the ablation procedure is even quicker when the second generation CB (Arctic Front AdvanceTM, Medtronic) is used (93-135 min).22,23,25 Similar to procedure duration, fluoroscopy times improved from 28 min to 12 min during the learning curve. The first-generation CB previously required a mean fluoroscopy time of 46 min (range, 20 to 95 min), indicating efficient application of the technology in the present study.21 The operator in the present study achieved fluoroscopy times equivalent to those currently observed with procedure-optimized use of the improved second generation CB (12-25 min).22,23,1,27
We observed an exceptionally low 0% complication rate despite first-time CB ablation by the operator in the present study setting. Importantly, this experience included no reported cases of phrenic nerve damage. This result suggests that within an experienced environment CB ablation may be not only rapidly but also safely integrated into an operator’s clinical ablation routine. In prior studies, the first generation CB has been associated with moderate complication rates that were mainly carried by PNP (6.4%; although only 3.5% PNP was reported with the 28 mm balloon used here), vascular access complications (1.8%), pericardial effusion or tamponade (1.5%), and embolic events (myocardial infarction, stroke, TIA; 0.6%).21 Of note, the second generation CB is linked to PNP in 2-20% of cases and to 2.4% vascular access complications.22,24,25,29
The primary goal of the study was to assess early characteristics and a learning curve associated with CB ablation of AF. Therefore, the number of patients was limited to. In addition, analyses were confined to procedure-related parameters, and postprocedural outcome data were not acquired. The strict focus on acute safety and efficacy provides immediate support for decision making with respect to strategic planning of AF ablation programs. It is important to recognize that the operator had significant previous expertise in radiofrequency ablation of AF and was therefore skilled in obtaining transseptal access and in left atrial catheter manipulation. These prerequisites may have facilitated rapid implementation of CB ablation into the electrophysiologist’s clinical routine. Thus, results of first-time CB use may vary among other operators depending on their individual experience with AF ablation.
Since the conduction of this study technical and procedural standards have evolved further. Currently, single transseptal access and the second generation CB are routinely used, and periablation anticoagulation management has been modified. Furthermore, a strategy employing single energy applications per vein is increasingly embraced, and individual ablations times have been shortened to include 3 min applications/vein.27,30 Considering these procedural simplifications that are now widely accepted, it is reasonable to assume that CB ablation procedure times and learning curve may be shorter in current practice compared to the present study.
CB ablation of AF is feasible and effective when performed by an operator without prior experience with this technology. Extremely low complication rates suggest safety of the procedure. Procedure duration as well as fluoroscopy and energy delivery times decrease rapidly during the first 40 procedures. Thus, the CB in combination with a circular mapping catheter appears to be an appropriate tool for AF ablation in the hands of a newly trained operator, provided that backup expertise is ensured.
E. Scholz received speaker’s honoraria from St. Jude Medical and travel compensation from Boston Scientific. P. A. Schweizer received travel grants from Medtronic and from St. Jude Medical. E. Zitron received speaker’s honoraria from Medtronic and from St. Jude Medical. D. Thomas received research support, speaker’s honoraria and travel compensation from Medtronic as well as speaker’s honoraria and travel compensation from St. Jude Medical.