Contact Force and Atrial Fibrillation Ablation
Ullah W1, Schilling RJ1, Wong T2
1Cardiology Research Department, Barts Heart Centre, St Bartholomew’s Hospital, Barts Health NHS Trust, London, UK.
.2NIHR Cardiovascular Biomedical Research Unit, Institute of Cardiovascular Medicine and Science, The Royal Brompton and Harefield NHS Foundation Trust and Imperial College London, UK.
.
Catheters able to measure the force and vector of contact between the catheter tip and myocardium are now available. Pre-clinical work has established that the degree of contact between the radiofrequency ablation catheter and myocardium correlates with the size of the delivered lesion. Excess contact is associated with steam pops and perforation. Catheter contact varies within the left atrium secondary to factors including respiration, location, atrial rhythm and the trans-septal catheter delivery technology used. Compared with procedures performed without contact force (CF)-sensing, the use of this technology has, in some studies, been found to improve complication rates, procedure and fluoroscopy times, and success rates. However, for each of these parameters there are also studies suggesting a lack of difference from the availability of CF data. Nevertheless, CF-sensing technology has been adopted as a standard of care in many institutions. It is likely that use of CF-sensing technology will allow for the optimization of each individual radiofrequency application to maximize efficacy and procedural safety. Recent work has attempted to define what these optimal targets should be, and approaches to do this include assessing for sites of pulmonary vein reconnection after ablation, or comparing the impedance response to ablation. Based on such work, it is apparent that factors including mean CF, force time integral (the area under the force-time curve) and contact stability are important determinants of ablation efficacy. Multicenter prospective randomized data are lacking in this field and required to define the CF parameters required to produce optimal ablation
Key Words : Catheter Ablation, Atrial Fibrillation, Contact Force.
Correspondence to: Tom Wong
The Royal Brompton and Harefield NHS Foundation Trust
Sydney Street, London, SW3 6NP. United Kingdom
Traditionally, operators were reliant on indirect measures of the contact between the tip of the ablation catheter and myocardium, such as the fluoroscopic appearance of the catheter and tactile feedback, to guide ablation. With the advent of contact force (CF)-sensing catheters, this is no longer the case, as these data are now directly measured and available in real-time during a procedure. In this review, we present the preclinical work correlating CF and lesion sizes, the factors determining catheter contact in the human left atrium (LA), the impact of CF-sensing on the atrial fibrillation (AF) ablation procedure, and review work focused on establishing optimal CF parameters for ablation.
Catheter-Based Contact Force Sensing Technologies
The first of the catheter-based technologies to obtain a CE mark (2009) was the TactiCath® (St Jude Medical Inc., St Paul, MN, USA). This catheter uses three optical fibers between the second and third electrodes of the catheter and an elastic polymer catheter tip1. The latter undergoes micro-deformations in response to contact which changes the wavelength of reflected infrared light transmitted by the optical fibers. The magnitude and orientation of the contact force may then be derived from the wavelength of reflected light at the three fibers.1
The most recently CE marked of the contact force sensing technologies is the ThermoCool® SmartTouch™ Catheter (Biosense Webster Inc., Diamond Bar, CA, USA). Here the catheter the tip electrode is mounted on a precision spring permitting a small amount of electrode deflection. By measuring this deflection using location sensor coils at the proximal end of the spring, the system can calculate the force being exerted (and its orientation) using the known characteristics of the spring. The SmartTouch catheter integrates with the Carto3 (Biosense Webster Inc.) electroanatomic navigation system. During a case, the magnitude of contact force and its vector are displayed in real-time on the Carto3 display screen as well as the contact force waveform (Figure 1).
Figure 1. Display of contact force data measured by the SmartTouch Catheter on the Carto3 screen Displayed is a left atrial geometry created using the Carto3 electroanatomic navigation system. Highlighted are the contact force, contact force vector and the contact force waveform for the contact between the catheter and the myocardium
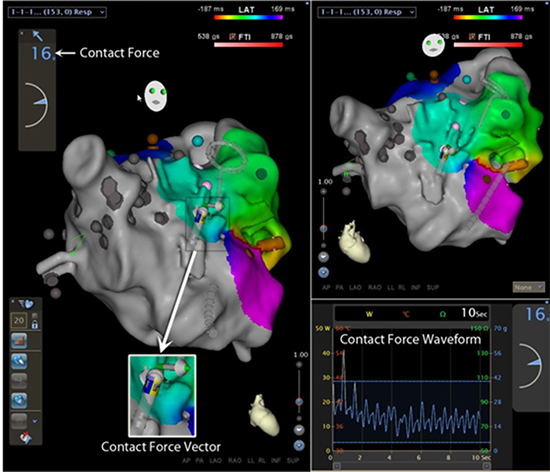
For the SmartTouch catheter, the reported sensitivity reported by the manufacturer is less than 1g of contact force. Data has been published for the Tacticath, where a comparison has been made between the measurements made by the catheter and a calibrated balance: this demonstrated the measurements by the catheter were highly sensitive and accurate (mean error ≤1g).1
Contact Force and Ablation
In vitro work with non-irrigated catheters using temperature-controlled ablation demonstrated a linear relationship between CF and lesion size, with the largest difference between no contact and being in contact with tissue.2 Over a wide range of contact forces (0-400N), there was an increase in lesion size with increasing CF (as long as the electrode tip temperature was maintained at the pre-set value and contact was maintained).2 The increase of lesion size with force is small such that a in a study comparing 10g to 20g of contact, no difference in lesion size was found.3 Because the power output of non-irrigated catheters is controlled by the catheter temperature the effect of changes in contact force is minimized. Poorer contact entails more power output to reach the same temperatures2 and since lesion size relates to temperature at the electrode tip,4 similar lesion sizes are therefore produced.
Conversely, if power controlled ablation is used, there is a significant relationship between lesion size and tissue contact.5 High contact and increased power is associated with an increase in steam pops and impedance rises following rapid, excessive electrode temperature rises.5 Irrigated ablation is not temperature controlled and thus lesion size is significantly greater at a force of 30g compared to 10g in vitro.6 Even with temperature-controlled power-limited ablation, in an in vitro model, where fluid flow external to the catheter is utilized to cool the catheter tip, lesion size increases with catheter contact before a plateau is attained.7
These proof of principle studies used catheter rigs where catheter tip force was extrapolated from the force loaded onto the catheter. Similar technologies in vivo demonstrate that increased CF is associated with larger and more transmural lesions8 but also more steam pops.9 These systems have the limitation that force measurement is not as accurate because of friction between the catheter and the sheath which varies with sheath deflection. Irrigated tip force-sensing catheters have further examined the impact of different levels of CF on lesion size and also the interplay of this variable with the ablation power. Increased CF is associated with increased tissue temperature at 3 and 7 mm away from the ETI, and is associated with increased lesion size at a given level of CF, as is increasing ablation power.1 Increasing CF is also associated with an increased incidence of thrombus formation at the electrode edge, especially with increasing power, and a similar relationship is also apparent for steam pops.1,10
Within the beating heart, even within the fibrillating atrium, it would be expected that contact will be dynamic rather than static. Such dynamic contact has been simulated in an in vitro setting where constant (static) contact has been found to produce the largest lesions while variable and intermittent contact produce progressively smaller lesions.11 Therefore, the dynamic quality of contact between the electrode and tissue is also of great importance in the efficacy of lesion formation. A measure termed the force time integral (FTI) has therefore been proposed as a measure of catheter contact during ablation. This is the area under the force-time curve and incorporates both the variation in CF during an ablation and also ablation duration. The FTI has been found to correlate with the lesion volume in in vitro experiments.11
Contact Force in The Left Atrium
Better electrode-tissue contact results in a greater proportion of the delivered power contributing to the resistive heating of the tissue, rather than wasted in the blood stream. Consequently, this can lead to larger, more likely transmural lesions but also increases the risk of complications through excessive tissue heating. In an ex vivo porcine heart study, a force of 417±167g could perforate un-ablated left atrium, while ablation reduced the force needed to perforate.12 A further study of in vivo porcine hearts found that the lowest CF recorded to cause perforation was 77g, with a force of 158.4 ± 55.4 needed to perforate unablated left atrium.13 A study in patients undergoing AF ablation demonstrated that the actual contact forces exerted on the myocardium vary significantly among operators during mapping and ablation when they were asked to maintain what they perceived (without CF-sensing technology) to be ‘good contact’.14 This included multiple high force events defined as the contact force exceeded 100g for 200ms, with six of the thirty four patients having over 40 such instances.14 These episodes occurred during catheter manipulation as well as ablation. A further study where operators also blinded to CF measurements mapped around the pulmonary veins specifically found a significant variation in the CF by location, with points taken around the left veins having lower CF than the right side, and the LA/left atrial appendage ridge points having the lowest CF.15
One would assume that having access to CF data would therefore reduce the risk of complications such as cardiac perforation, as high force episodes could be reduced. A retrospective study including 557 patients undergoing AF ablation demonstrated that the use of CF-sensing catheters was associated with a significant reduction in the rate of major complications (2.1 vs 7.8%, p=0.01) including cardiac perforation (0 vs 3.3%, p=0.021).16 However other studies have shown no reduction in complications with CF-sensing data available.17–19
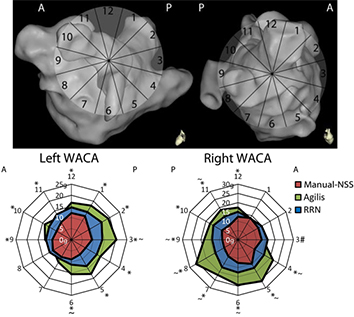
There are areas around the pulmonary veins which are more resistant to electrical isolation20 and have a higher frequency of both acute and chronic reconnection.21 The intervenous ridges and pulmonary vein-left atrial appendage ridge are important areas in this respect.20,21 The reason for this locational variation in the efficacy of ablation may relate variation in wall thickness and texture,22 resulting in differences in the compliance of different regions,23–26 thus changing the quality of the contact with the catheter. While availability of CF data could affect the CF applied by the operator during LA mapping and ablation, there are other additional factors which also play a role in determining contact in the LA. Respiratory motion may affect CF, with lesions delivered during apnea having a higher average contact force and force time integral than those during ventilation.27 During wide area circumferential ablation, the CF varies by location, with lower forces on the left side and anteriorly.26,28 In those ablated with the operator blinded to CF data, the CF is significantly lower during ablation around the LA/left atrial appendage ridge compared with cases where CF data was available.28 The type of trans-septal sheath used to deliver the ablation catheter during ablation, whether manual non-steerable or steerable also affected the distribution of contact forces around the WACA (Figure 2).29
Figure 2. Distribution of contact force in the right and left WACA Upper figure demonstrates a clock face scheme for segmenting each WACA, left pulmonary veins on the left, right pulmonary veins on the right. Lower figure demonstrates radar plots of the distribution of contact forces during initial pulmonary vein encirclement for 60 patients with persistent AF using different trans-septal sheaths. The outer edge represents the WACA location according to the clock face segmentation. The further from the center of the plots, the higher the median contact force in grams A=Anterior; P=Posterior; Manual-NSS=Manual Non-steerable sheath group; RRN=Remote Robotic Navigation *=Significant difference between Manual-NSS and steerable sheath groups (p<0.05); #=Significant difference between RRN and Manual-NSS groups (p=0.009); ~= Significant difference between RRN and Agilis group (p<0.05); +=Significant difference between indicated groups (p<0.05) (Reproduced from Ullah et al,29 Journal of Cardiovascular Electrophysiology, Wiley, with permission)
The quality of contact between the catheter and myocardium is not just reflected by the mean CF but also by the stability of the contact force waveform, quantified as the CF variability (CFV) (Figure 3).26
Figure 3. Contact force variability Contact force (CF) waveform over time at two mean contact forces – the contact force variability (CFV) is the difference between the mean trough and peak CF. The mean CF is indicated by the solid line while the mean peak and trough CFs are indicated by the dotted lines. On the left of the image is a waveform at low mean CF with low CFV, while on the right is a waveform at higher CF with higher CFV (Reproduced from Ullah et al,26 Journal of Cardiovascular Electrophysiology, Wiley, with permission)
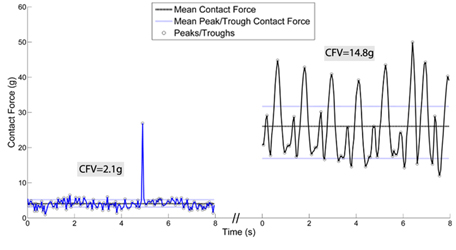
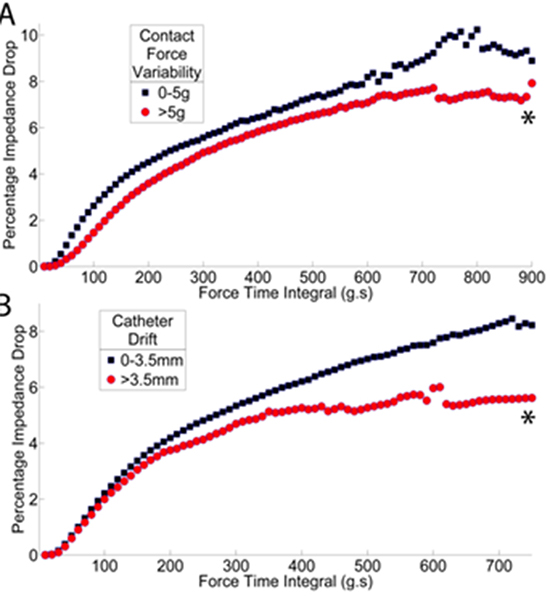
The greater the mean CF applied, the greater the CFV. This may be because at low CF the myocardium has a large capacity for stretching and can buffer sources of variability such as cardiac motion from affecting contact with the catheter; at higher CF, CFV increases, suggesting that less of this variability is buffered by the tissue and more transmitted to the catheter tip. Other factors have also been found to increase the CFV: sinus rhythm rather than AF (presumably because of increased cardiac movement); stiffer robotic sheaths, and LA location. Apnea has also been found to be associated with a reduction in the variability of the applied CF.27 This variability of the CF is of relevance to ablation as there is evidence that for the same FTI, a higher CFV can result in a lower ablation efficacy (Figure 4).26
Figure 4. Factors affecting impedance drop during ablation (A) Contact Force Variability and (B) Catheter drift. Each point is the mean of at least 10 ablations. *p<0.0005 (Reproduced from Ullah et al,26 Journal of Cardiovascular Electrophysiology, Wiley, with permission)
Impact of CF-Sensing on Clinical AF Ablation
One single center 38 patient prospective randomized study has been published assessing the impact of CF-sensing on AF ablation. A non-randomized prospective single-arm study in which 160 patients were ablated at 21 sites has been also published30 as well as prospective, non-randomized studies enrolling around 20-30 patients in the CF-sensing arm and comparing with a non-CF sensing group.28,31–34 Aside from these, multicenter registries,16,17,19 including 200-600 patients have also been published.The results from these studies are discussed below.
A limitation in all but two18,28 of the two arm studies published to date is that there has been some variation in the equipment used in the two study groups, namely the ablation catheter or mapping systems. This could introduce bias based on the handling characteristics of the CF-sensing catheter itself compared with non-CF sensing catheters, rather than its ability to measure CF. Moreover, the use of CF-sensing generally requires the most up to date iterations of electroanatomic navigation systems. In non-CF sensing groups, some of the cases could conceivably be performed using older systems - previous studies have demonstrated differences in procedural parameters between different mapping systems35 and different versions of the same mapping system.36
The use of CF-sensing catheters has been described to be associated with a reduction in fluoroscopy times,17,19,32,34 though in two studies fluoroscopy times were found to be longer in the CF-sensing group.16,31 Procedure times were also shorter in the CF-sensing arms of the above studies,17,32–34 other than in one study where it was significantly longer where CF data were available.31 In the randomized trial, procedure but not fluoroscopy times were shorter when CF data were available to the operator.18
While the above procedural parameters are of importance, a key factor determining the utility of CF-sensing is clinical efficacy. Success rates have been compared in some of the publications to date.17–19,31,32,34 In some cases, there was an improvement in success rates with the use of CF-sensing.17,19,31,32 In one of these studies, a benefit in terms of success rates was only observed for patients in PAF rather than persistent AF.19 In another study, exclusively examining persistent AF patients, an improvement in ablation success rates was only observed when CF-sensing was used with the remote robotic navigation system and not when used with manual ablation.17 Other studies, including the prospective randomized one, have not observed an improvement in success rates when CF-sensing was used.18,34
CF-sensing catheters have also been found to be associated with a reduction in procedure times in hybrid epicardial (using a bipolar RF catheter)/endocardial AF ablation procedures, in a study comparing the data with a historical cohort where a non CF-sensing catheter was used for the endocardial ablation.37 On comparison with the second generation cryoballoon, in a multicenter non-randomized study of 376 patients with PAF, the procedure times for CF-sensing catheters are significantly longer without a difference in fluoroscopy or overall complication rates, and with no difference in success rates at 18 months.38
Clearly, further, larger prospective randomized and preferably multicenter trials are needed to clarify the impact of CF-sensing on procedure parameters and success rates.
Optimizing CF Parameters During Ablation
An important point with regard to the success rates from AF ablation procedures is that it is unlikely that simply having CF-data available will improve outcomes: it more likely that using the data to maintain optimal ablation CF will make the difference. In this respect it is interesting that in the SMART-AF trial, cases where the operator had maintained the CF within their self-determined optimal range ≥80% of the time were associated with a significant improvement in success rates, with such procedures over 4 times as likely to be successful than those where this was not the case.30 This then raises the important question as to what the optimal CF parameters for ablation should be.
Contact force during ablation predicts acute wide area circumferential ablation (WACA) reconnection in patients with PAF, with sites of pulmonary vein reconnection having a lower average contact force27,28 and FTI during ablation.27 At 3 months’ follow up, segments within a WACA line ablated with a minimum force time integral below 400g.s had a greater chance of reconnection among PAF patients.39 At 12 months of follow up, the average contact force, FTI and incidence of low CF during ablation are predictive of procedural success in PAF patients.40
The aim of a radiofrequency application during AF ablation is the generation of a transmural lesion. This results in a persistent barrier to electrical conduction or the elimination of a driver. At a procedural level, this is best reflected by an improvement in the single procedure success rate for the ablation. Clearly, this is the most relevant outcome measure clinically. CF parameters have been compared between cases with and without a recurrence of symptoms by Reddy et al.,40 (Table 1), with higher CFs during ablation observed in those without recurrence. An improvement in success rates may not necessarily mean that the ablation procedure has been more efficient: this would be reflected by a reduction in the procedure length, for example. In order to make procedures more efficient, the aim should be for every radiofrequency application to be contributory to the success of the procedure. This would lead to shorter procedures and potentially less risk of complications. Moreover, suboptimal applications may lead to short term procedural success but long-term failure – by causing tissue edema and an incomplete transmural lesion. Consequently, it is useful to be able to assess the efficacy of individual radiofrequency applications.
Table 1. Clinical studies assessing ablation efficacy with respect to catheter contact force: methods used to assess efficacy and cut off values for effective ablation
| Author |
Number of Patients |
Operator Blinded to CF |
AF Subtype |
Method to judge ablation efficacy |
Suboptimal Ablation |
Effective Ablation |
| Reddy40 |
32 |
No |
PAF |
12 months recurrence of symptoms |
CF <10g; FTI<500g.s |
CF>20g
FTI>1000g.s |
| Haldar28 |
40 |
In half of cases |
35% PAF |
Acute PV reconnection in a 7 segment model per PV pair |
CF 14.5g |
CF 19.6g |
| Kumar27 |
12 |
Yes |
PAF |
Acute PV reconnection in a 5 segment model per PV pair |
LPV: CF 9g, FTI 173g.s RPV: CF 11g, FTI 282g.s |
LPV: CF 20g, FTI 436g.s RPV: CF 24g, FTI 609g.s |
| Kumar47 |
20 |
Yes |
PAF |
EGM criteria
for transmurality46 |
|
CF>16g, FTI >404g.s |
| Neuzil39 |
40 |
Yes |
PAF |
PV reconnection at 3 month protocol-driven restudy in a 5 segment model per PV pair |
CF 15.5g Minimum CF 3.6g Minimum FTI 118g.s |
FTI>400g.s CF 19.5g Minimum CF 8.1g Minimum FTI 232g.s |
| Ullah29 |
60 |
No |
Persistent AF |
Reconnecting segments in a 12 segment model per PV pair at redo procedure (median 8 months from index procedure) |
CF 11.5g FTI 231g.s |
CF 12.5g FTI 231g.s |
| Sohns53 |
6 |
No |
PAF |
MRI-defined scar in 5mm2 zone |
|
>1,200g.s |
EGM=Electrogram; PV=Pulmonary Vein; CF=Contact Force; FTI=Force Time Integral; LPV=Left Pulmonary Vein; RPV=Right Pulmonary Vein
In preclinical studies, the efficacy of an individual ablation is relatively straightforward to judge as histological lesion dimensions are available.2,41 Lesion histology is not available for clinical studies though, and therefore alternative measures of the effect of ablation are used. Classically, the attenuation of the electrogram has been used to judge the efficacy of an individual ablation. Unipolar atrial electrogram attenuation has been found to be associated with transmurality of ablative lesions.42 Significantly more amplitude reduction in the bipolar signal during sinus rhythm and AF with transmural lesions is seen in vitro, with a reduction of ≥ 60% having a high specificity for lesion transmurality.43 In clinical studies, an ≥80% reduction in electrogram amplitude has been targeted.21,44 Electrogram attenuation has been found to correlate poorly with CF applied during ablation.26,45 Changes in sinus rhythm electrogram morphology have been shown to be predictive of transmurality of ablation lesions in a porcine model by Otomo et al.,46 In this case, for unipolar signals a loss of a negative deflection was associated with transmurality, while in the case of bipolar signals, the changes associated with the latter were dependent on the orientation of the catheter to the myocardium. One group has used these criteria for bipolar signals to judge ablation efficacy and found that CF parameters were sensitive and specific for identifying transmurality based on electrogram parameters.47 Another group found no relationship between CF parameters and transmurality as suggested by the above electrogram morphology changes.26
The most commonly employed model for assessing ablation efficacy is reconnection of the WACA lines27,28,39 (Table 1). In this approach, the ipsilateral WACA is divided into five to twelve segments and efficacy is based on whether that segment reconnects or not. The disadvantage here is that target parameters for individual radiofrequency applications are being assessed based on the response of a region, quite often with overlapping lesions, to ablation. In most of these studies, operators were blinded to CF measurements (Table 1): such blinding serves to exaggerate the differences between ineffective and effective ablations as a lack of knowledge of CF allow for a greater range of CF to be applied and therefore makes it difficult to establish where the actual threshold for effective ablation lies. Based on these studies, a mean ablation CF of at least 15g and FTI of >400gs would appear to be associated with a reduced risk of an ablation being in a reconnecting segment.
While histological lesion parameters are not available for clinical cases, work has been done using cardiac MRI to attempt to image ablation lesions. McGann et al., described a methodology for imaging LA scar using delayed enhancement MRI (DE-MRI) following pulmonary vein isolation, and the burden of LA scar they observed correlated with arrhythmia recurrence.48 This group went on further to demonstrate that areas of DE-MRI enhancement correlate with areas of electrical scar (R2=0.57) and that DE-MRI imaging could be used to identify breaks in the pulmonary vein isolation lines.49 In a blinded analysis using pre- and post-ablation MRI images, another group found that investigators were able to identify ablated LA myocardium in only 60% of cases, with a poor ability to distinguish ostial from circumferential ablation lesions.50 This contrasts with another report in which ablated myocardium could be identified in 100% of cases on DE-MRI.51 These findings suggest MRI may be useful in determining the sites of ablation lesions but the difference in the reported reliabilities may relate to the signal intensity thresholds being used to assign scar on MRI. To address this, recent work has correlated macroscopic scar volumes with DE-MRI imaging scar volumes in the right atria of 8 swine: based on this, DE-MRI signal intensity thresholds have been proposed which allow the best approximation of the macroscopic scar volume.52
Contact force parameters have been compared with MRI-imaged atrial scar by Sohns et al.53 Table 1. In this study of six patients, the FTI of ablation was correlated with DE-MRI scar. In order for this comparison to occur though, the FTI was not examined from the perspective of a single radiofrequency application, but in a subdivision of 1cm zones. Increasing FTI above 1,200g.s was associated with a significant increase in the proportion of a 5mm2 region of myocardium exhibiting DE-MRI scar (below this FTI value, the increase in the scar burden in that zone with an increase in FTI was small). This study therefore raises the possibility of using cardiac MRI to assess ablation efficacy clinically. The drawback here though is that the efficacy is being assessed at a MRI-zone level (albeit a small zone) rather than an individual radiofrequency application. This therefore relies on extremely accurate registration of each radiofrequency application between the electroanatomic navigation system and the MRI being used to judge scar. Moreover, this method is unable to account for any overlap in applications (for example through catheter drift even during a putative static application). It may be for these reasons that the threshold for effective ablation is much higher in this study compared with the other work presented in Table 1.
An alternative approach to clinically assess the efficacy of each individual radiofrequency application based on the impedance drop/FTI relationship has also been used.26,45 For persistent AF patients, the relationship was found to be logarithmic with a plateau at 500g.s.45 This method was also used to investigate the impact of contact force variability and catheter drift on the efficacy of ablation (Figure 4).26 Based on this work, maximal efficacy is provided by parallel catheter contact with CFV ≤5g, catheter drift ≤3.5mm and there is no benefit in terms of biophysical efficacy from ablation beyond 500g.s.
Such detailed optimisation of catheter contact during ablation is now possible with the introduction of automated lesion marker placement software such as the Visitag upgrade for Carto.3 In a study by Anter et al.,54 such an algorithm was used and found to be associated with lower rates of acute pulmonary vein reconnection but not improved success rates at 6 months. The limitation of that study though was that only catheter displacement and impedance drop were used by the annotation algorithm. The incorporation of CF parameters could be used to further refine the targets for ablation. A randomized trial where one group was ablated with optimised CF targets and the other without CF sensing could be conducted to definitively prove the utility of CF-sensing to the ablation procedure.
The availability of real time catheter-based CF-sensing holds great potential for improving the safety and success rates of AF ablation procedures by reducing suboptimal and excessive CF during ablation. Optimal CF parameters for ablation remain to be established, and one would hope that their adoption would help to optimize each individual radiofrequency application, improving procedural efficacy. Multicenter prospective randomized data are lacking in this field and are required to definitively prove the argument for the adoption of this technology and the CF thresholds required during ablation.