Current Role And Future Prospects Of Magnetic Resonance Imaging In The Field Of Atrial Fibrillation Ablation
Pilar Cabanas-Grandío1, Felipe Bisbal2
1Complexo Hospitalario Universitario de Vigo, Vigo (Spain).2Hospital Germans Trias i Pujol, Badalona (Spain).
Atrial fibrillation (AF) is the most common arrhythmia in clinical practice, and catheter ablation of AF has become a first-line treatment for symptomatic drug-refractory AF. However, this is a complex procedure and recurrences after a single ablation procedure are frequent. New technologies are needed to simplify the procedure and improve results, and cardiac magnetic resonance (CMR) has emerged as a useful tool to stratify the risk of recurrence, guide the ablation procedure, and monitor the post-ablation remodeling process. This review summarizes the current role of CMR in the field of AF ablation and offers a perspective on its future potential.
Key Words : Atrial Fibrillation, Ablation, Magnetic Resonance Imaging, Remodelling, Late Gadolinium Enhancement.
Correspondence to: Felipe Bisbal Van Bylen
Arrhythmia Unit-Heart Institute (iCor)
Hospital Universitari Germans Trias i Pujol
Carretera Canyet, 08916 Badalona
Atrial fibrillation (AF) is the most common sustained arrhythmia in clinical practice and is associated with impaired quality of life, increased morbidity and mortality, and socioeconomic burden, especially because of its association with a high rate of hospitalizations, heart failure, and thromboembolic events.1-3 Catheter ablation of AF has become a first-line treatment for symptomatic drug-refractory AF, and could be considered a first-line therapy for symptomatic paroxysmal AF.4 As technologies, techniques, and experience have improved, the popularity of catheter ablation continues to escalate. However, AF ablation is a complex and time-consuming procedure, and reported success rates remain suboptimal.4 Therefore, both industry and electrophysiologists engage in continuous development efforts to improve AF ablation results. In this setting, cardiac magnetic resonance (CMR) emerges as a useful tool. It can improve patient selection, guide the ablation procedure, and assess post-ablation left atrial (LA) remodeling.
Role Of Pre-Procedural CMR In Risk Stratification
Pulmonary vein isolation (PVI) is the most common technique in catheter ablation of AF and aims to electrically isolate the pulmonary veins (PVs), the major source of ectopic beats that can initiate AF.5 However, despite the enormous development in this field, the long-term efficacy of PVI remains limited. Several studies have demonstrated clinical variables that may predict long-term PVI success, such as paroxysmal AF compared to persistent or permanent AF.6 However, clinical features seem not to be enough to identify patients who will remain in sinus rhythm at long-term follow-up. Therefore, alternative tools to predict long-term procedural success are desirable.
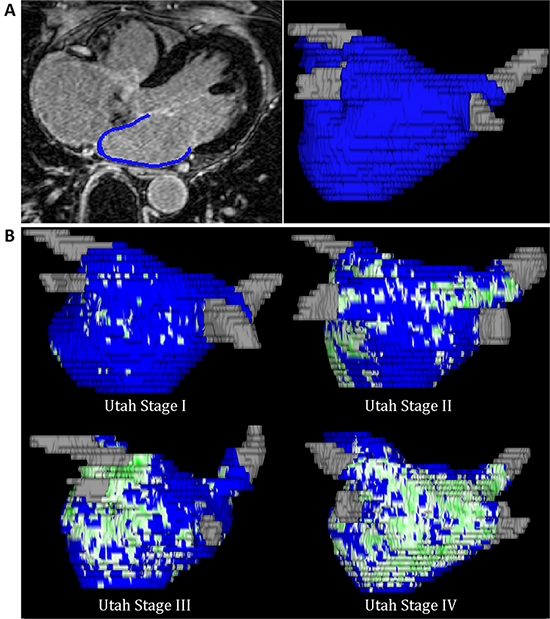
Late gadolinium enhancement CMR (LGE-CMR) has demonstrated accuracy in detecting fibrosis in the left ventricular myocardium in ischemic and non-ischemic cardiomyopathies.7-9 Over the past few years, improved LGE-CMR spatial resolution has allowed the identification of fibrosis (scar) around PVs and at the left atrial (LA) wall after PVI procedures.10 However, the current challenge of LGE-CMR studies is to identify the native fibrosis of the LA, which represents the potential substrate for AF and could predict ablation results. Recently, results from a single-center study11 showed that extensive LGE predicts poor response to catheter ablation therapy for AF. The study included 386 patients and quantified LA LGE before a first PVI procedure using the previously defined Utah structural remodeling (SRM) stages of LA wall LGE: stage I (<10%), stage II (10%–20%), stage III (21%–30%), and stage IV (<30%) (Figure 1). Patients with Utah stage IV had higher chances of recurrent AF at 1 year (71%) than patients with Utah stages I, II and III (21%, 29% and 34%, respectively). After adjusting for several clinical variables, stage IV was associated with the highest risk of recurrence (HR, 4.89 compared with SRM stage I; p<0.0001). These results seem to be consistent across centers, according to the reported data of a multicenter, international study.12 Marrouche et al12 found that for recurrent arrhythmia, the unadjusted risk of recurrence per each 1% increase in LA fibrosis was 6% (HR 1.06 [95% CI, 1.03-1.08]; p<0.001) and the estimated unadjusted cumulative incidence of recurrent arrhythmia by day 325 was 15.3% for stage 1, 32.6% for stage 2, 45.9% for stage 3, and 51.1% for stage 4, with similar results after adjusting for clinical covariates. However, one important limitation was that all image post-processing and fibrosis quantification was performed in a single core laboratory. Further multicenter studies in which recruitment, image acquisition, and post-processing are performed at each site are needed to confirm these promising results. Despite these findings, there are several limitations in the application of LGE-CMR to characterize the tissue of the LA wall and pulmonary veins: limited spatial resolution, the difficulty to avoid motion-related artifact, and the complex quantitative analysis of LA LGE. Future research may help to overcome these limitations and permit the use of LGE-CMR to identify patients at high risk of procedural failure.
Figure 1. (A) DE-CMR axial slice with the segmented LA wall (left panel) and the resulting three-dimensional reconstruction of the LA wall (Coreview, Marrek Inc, Salt Lake City, Utah, USA) (right panel). (B) Utah classification of the LA remodeling based on the percentage of enhancement of the LA wall volume: <10% (Utah I), 10–20% (Utah II), 20–30% (Utah III), and <30% (Utah IV). DE-CMR, delayed enhancement cardiac CMR; LA, left atrium. Reproduced from Bisbal F et al. Heart 2014;100:1975-1984 with permission of BMJ Publishing Group Ltd.
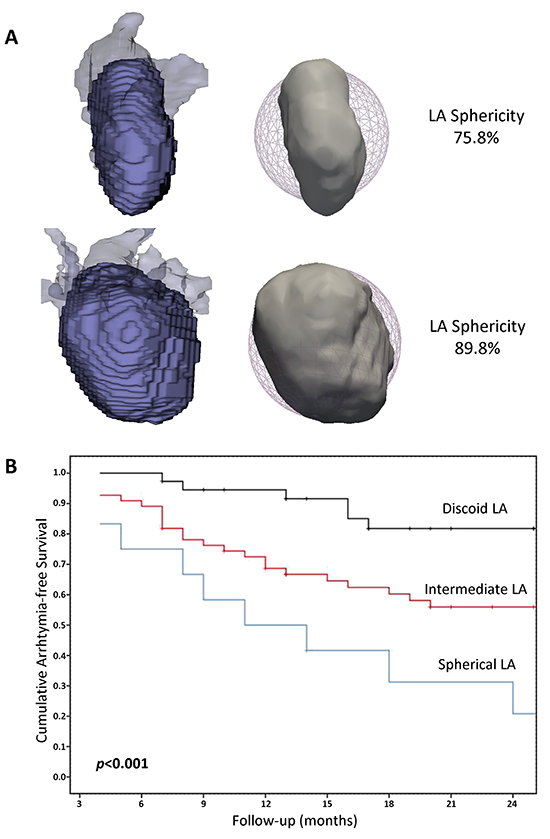
The use of CMR angiography allows the assessment of LA dimensions and shape: LA enlargement is associated with AF6 and LA diameter and LA volumes are common methods to determine LA size; however, these measurements have limited value when assessing the remodeling process in patients with AF. Recently, LA sphericity has been proposed as a new shape-based parameter to assess LA remodeling.13 This new approach quantifies the similarity of the LA geometry to a perfect sphere, a parameter that has shown independent predictive value for AF recurrence after ablation, exceeding that of LA size. Patients with a spherical LA had 11-fold higher probability of recurrence at one-year follow-up, compared to patients with a normal (discoid) LA shape (Figure 2). LA sphericity had better sensitivity and specificity values than LA volume, as determined by ROC analysis, and was the only remodeling parameter that remained significant in the multivariate analysis, suggesting that LA sphericity might be an earlier marker of adverse remodeling. The application of this parameter to pre-procedural risk stratification could help to identify those patients unlikely to respond to ablation, improving the risk-benefit ratio.
Figure 2. (A) Right lateral projection of the LA three-dimensional reconstruction (pulmonary veins and LA appendage shown in transparency) and the integrated sphere that best fitted the LA (shown as a mesh) of a patient with discoid LA (upper panel) and another with spherical LA (lower panel). (B) Kaplan-Meier curves displaying the cumulative arrhythmia-free survival of patients with discoid, intermediate, and spherical LA. LA, left atrium. Reproduced from Bisbal F et al. Heart 2014;100:1975-1984 with permission of BMJ Publishing Group Ltd.
Role Of CMR During Af Ablation Procedure: Usefulness To Guide The Procedure
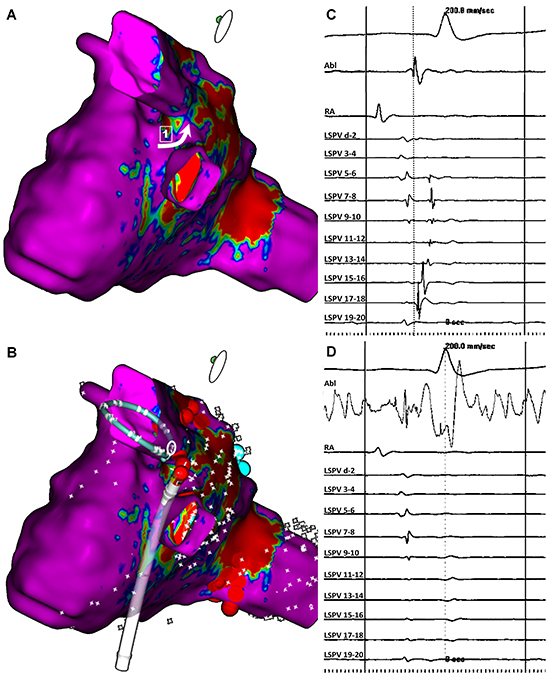
Pre-procedure CMR angiography and computed tomography of the LA are often used to define the LA anatomy and to assess the location and anatomy of the PV antra. These imaging modalities facilitate the interpretation of LA anatomy during mapping and are often used for image integration. The goal of AF ablation is to achieve permanent PVI. Additional testing to determine complete and durable isolation includes adenosine administration and pacing on ablated tissue; however, recurrences are still frequent. Recovery of conduction between the PVs and the LA has been postulated as the main mechanism for AF recurrence14 and this reconnection seems to be related to gaps in the previous encircling lesions around the PVs.15 LGE-CMR allows the identification of LA scarring after PVI and, therefore, the identification of ablation gaps. We recently reported our first experience with a CMR-guided ablation of gaps in repeat AF ablation procedures. The study showed the feasibility of CMR to identify previous ablation lines and gaps, but most importantly, this was the first study showing that LGE-CMR can successfully guide repeated ablation procedures by targeting the anatomical gaps of circumferential antral lesions and additional linear lesions.16 We evaluated 15 patients undergoing repeated AF ablation. A pre-procedure LGE-CMR was performed to create a 3D reconstruction where a pixel signal intensity map was projected and color-coded, generating a CMR model that was imported into the navigation system and merged with the LA electroanatomic map to guide the ablation of CMR gaps, defined as any discontinuity of previous ablation lesions (scar zone). All of the electrically reconnected PVs had CMR gaps. The CMR-guided ablation led to re-isolation of 95.6% of reconnected PVs and conduction block through the roof line in all patients (Figure 3). In light of this pilot study, CMR seems to be useful in planning repeated ablation procedures by assessing the number, length, and location of gaps, as well as successfully guiding catheter positioning to target them without the need of circular multipolar catheters. Because of its clinical implications, further research to assess the utility of CMR guidance for targeting of gaps is warranted.
Figure 3. Example of a CMR imaging-guided ablation of gaps leading to left superior pulmonary vein reisolation. A. Left atrial lateral projection of the DE-CMR model showing a gap at the carina (white arrow). B. Lasso catheter into left superior pulmonary vein and ablation catheter located in the gap (white circle shows D-2 Lasso dipole position). C. ThermoCool Smart Touch and Lasso catheter (Biosense Webster) electrograms before ablation show PV potentials. The gap in CMR correlates with D-2 Lasso dipole position (white circle), showing earliest PV potential (dotted line). D. Lasso catheter electrograms after radiofrequency application. Reproduced with permission of Elsevier from Bisbal F et al. J Am Coll Cardiol Img;2014;7:653-663
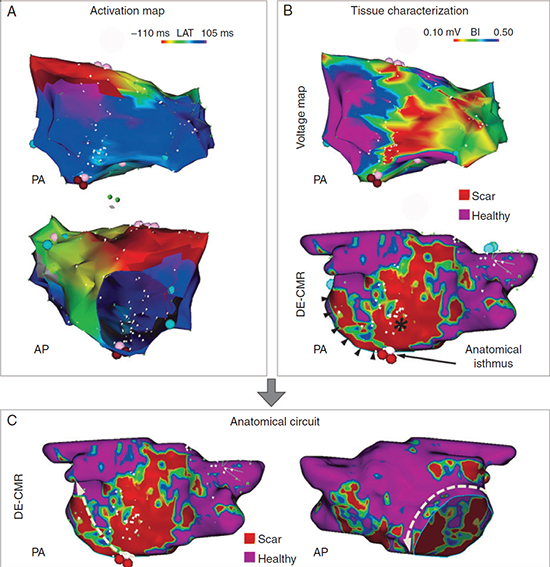
The role of CMR to guide ablation of supraventricular arrhythmias might not be confined to repeat AF ablation procedures. We recently reported the usefulness of CMR to characterize the substrate and guide ablation of complex left atrial arrhythmias, such as left atrial macroreentrant tachycardias.17,18 CMR permitted pre-procedural identification of the anatomical isthmus, which allowed procedural planning, limited mapping to the area of interest, and limited energy delivery (Figure 4).
Figure 4. Panel A. Activation map showing a counterclockwise perimitral activation sequence. Panel B. Substrate characterization with voltage map (upper part) and LGE-CMR (lower part). LGE-CMR, but not voltage mapping, identified an anatomical isthmus at the inferior aspect of the left atrium, between a large, scarred area at the posterior wall (asterisk) and the inferior mitral annulus (black arrowheads). The integrated CMR allowed catheter positioning and limited mapping at this site; a continuous, fractionated signal –highly suggestive of a protected, slow-conducting isthmus– was identified. Radiofrequency application at this site (red dots in panels A, B and C; 120 seconds) led to termination of the arrhythmia. Reproduced with permission of Oxford Journals from Bisbal F et al. Europace 2015;17:186
Real-Time CMR: The Future Of The Electrophysiology Lab?
Integration of CMR into the EP lab has recently been introduced. The purpose of the MRI-EP suite is to avoid radiation exposure by both patient and operator. Further, accurate definition of cardiac structures and arrhythmic substrates such as scar and fibrosis, visualization of catheter position, and the possibility to monitor the efficiency of radiofrequency applications by observing the lesion formation makes this technology very attractive.
Initial experiences with ablation of arrhythmias using real-time CMR guidance indicate that it seems to be feasible and safe.19,20 The development of this technology would address some of the current limitations of ablation procedures, such as procedural radiation exposure and therapeutic efficacy.21 Radiation exposure has been significantly reduced with the use of electroanatomic mapping systems; however, the use of interventional CMR would completely eliminate radiation exposure. Efficacy could be improved by identifying areas of edema with incomplete isolation; advances and improvements in real-time CMR guidance might be useful to detect gaps during the index procedure despite transient electrical isolation due to edema or non-transmural lesions. This would be a very important step toward improving the long-term outcome of AF ablation. However, despite these potential improvements, the implementation of a CMR suite is still a challenge. Several limitations must be overcome: the need for compatible equipment (12-lead ECG, catheters, wires, and ablation systems) and improved signal filtering to allow reliable signal interpretation, as well as the guarantee of patient safety: current defibrillation systems are not safe inside the CMR.
Role Of CMR After Af Ablation: Reverse Remodeling And Assessment Of Lesion Formation
Post-Procedural Remodeling
CMR and computed tomography are often used post-ablation when complications, such as pulmonary vein stenosis, are suspected. CMR provides additional information about LA remodeling after the ablation. Several studies have shown a decrease in LA size, diameter, and/or volume after AF ablation.22,23 We recently published the effect of PVI on LA reverse remodeling evaluated by LA sphericity.24 LA reverse remodeling was defined as the improvement in LA sphericity (spherical reverse remodeling) or volume reduction (volumetric reverse remodeling). The study evaluated 102 patients, of which 42% presented spherical reverse remodeling and 69% volumetric reverse remodeling. A trend towards a higher proportion of spherical reverse remodeling was observed in patients with no recurrence, compared to those with recurrences (46.2% vs 32.4%, respectively). A significantly greater proportion of patients with paroxysmal AF had spherical reverse remodeling, compared to those with persistent AF (50.8% vs 29.3%, p=0.03). Indeed, patients with persistent AF showed post-procedural worsening of LA sphericity (81.9% vs 82.9%, p=0.018). Reverse spherical remodeling was found in a lower proportion of patients, compared to volumetric remodeling. The reduction of volume could be related to both scarring and LA function recovery, while LA sphericity could be more related to LA structural recovery. These findings suggest LA reverse spherical remodeling may be more sensitive to identify favorable remodeling after ablation than is LA volumetric remodeling.
Lesion Formation And Gap Identification
Studies about lesion formation after AF ablation have shown controversial results regarding the ability of LGE-CMR to identify gaps in ablation lesions. Recently, Harrison et al25 reported the inability of LGE-CMR to reliably predict sites of electrical conduction after PVI. They found a weak point-by-point relationship between LGE-CMR and endocardial voltage in patients undergoing repeat AF ablation. In contrast, our group reported high rates of PV re-isolation (95.6%) when targeting the gaps identified by LGE-CMR, which ultimately implies good agreement between techniques.16 These conflicting results might be explained by several differences in the CMR image acquisition, image post-processing, and procedural technique. Compared to our study, Harrison et al created maps with a significantly lower density of points, mean 338 ± 210 (range 51-901), compared to 808 ± 159 (range 520-1004) in our study (p<0.001). This fact could limit the voltage information in areas of interest. Additionally, in their study, LGE-CMR was acquired with a greater voxel size (1.25x1.25x4mm) than has been reported by other groups (1.25x1.25x2.5mm),16,26 which could limit the assessment of smaller anatomical gaps.
LGE-CMR can identify LA fibrosis and ablation-induced scarring. Akoum et al27 reported a subanalysis of the DECAAF trial that studied the effect of residual LA fibrosis and scarring on AF catheter ablation outcomes. They found that the overall pre-ablation fibrosis was 18.7 ± 8.7%. The overall ablation-induced scar after PVI averaged 10.6 ± 4.4%. The calculated residual fibrosis, defined as pre-ablation fibrosis not covered by ablation scar, was 15.8 ± 8%. Baseline LA fibrosis and residual fibrosis and residual fibrosis were independently associated with AF recurrence (HR 1.09, 95% CI 1.06-1.12, p<0.001 and HR 1.09, 95% CI 1.05-1.13, p<0.001, respectively), while PV encirclement and overall scar were not. Therefore, native LA fibrosis, identified by CMR, could represent a target for ablation in AF patients; further research is needed to confirm this hypothesis.
Pre-Procedural Assessment Of Left Atrial Structural Remodeling
Two features of LA remodeling assessed by CMR have been associated with AF ablation procedure failure: native LA fibrosis and geometry (sphericity). Pre-procedural quantification of both MRI-derived remodeling parameters may identify patients with more advanced atrial disease and help to select optimal candidates for AF ablation.11,12,26 However, both technologies have limitations. Quantification of LA fibrosis lacks a validated protocol for LGE-CMR sequence acquisition. Segmentation and quantification tools tend to be self-customized and no external validation has been reported in the majority of institutions.12,16,25 Regarding LA sphericity, only a single-center study has been performed to date. A multicenter study is needed to confirm the reproducibility and applicability of the technique across centers, as well as to validate its predictive value. Further research must address these issues to ensure the future of this imaging modality in this context. Additionally, external validation is mandatory to ensure reproducibility and allow its implementation in the clinical setting.
The staging of LA disease by means of LA fibrosis or spherical deformation might also be applied to patients with cardiovascular risk factors to identify those patients at high risk of developing AF, for whom close follow-up and extended ECG monitoring might be cost-effective.
CMR As A Guide For Ablation
LGE-CMR has demonstrated its capacity to identify gaps after AF ablation and to guide redo procedures.16 However, the potential benefit over the conventional approach is yet to be elucidated. A randomized trial of CMR-guided vs conventional repeat AF ablation procedures is warranted.
Currently, PVI is the most common endpoint for AF ablation. However, in persistent and longstanding AF, PVI may be insufficient to reach comparable results as in paroxysmal AF and to prevent long-term recurrences. Recently, the randomized, controlled STAR AF II trial28 found no benefit of additional substrate modification (mitral and roof lines or ablation of complex fractionated activity) over PVI alone in patients with persistent AF. The authors suggest that more selective targets are needed to better characterize the specific arrhythmic substrate of a given patient. Along this line, Jadidi et al29 reported an inverse relationship between complex fractionated electrograms and the presence of fibrosis assessed by LGE-CMR. They found that CFAEs were most frequently located at the edge of fibrotic areas of the LA wall; hence, its role could be passive activation rather than an active mechanism of AF perpetuation. In fact, electrograms within the dense scar were less fractionated and had longer cycle length. In this regard, several studies have shown that a more extensive ablation based on debulking of the septal and posterior wall30 or isolation of low-voltage areas in the LA31 in addition to PVI might have a beneficial effect. However, no study to date has evaluated the effect of ablating fibrotic areas identified by CMR. Future research must address this intriguing question to elucidate if an CMR-guided ablation of arrhythmogenic substrate (fibrosis) offers some benefit over PVI alone in patients with advanced atrial disease.
Cardiac CMR can be a useful tool to select the best candidates for AF ablation, to guide the ablation procedure, and to evaluate LA remodeling after the ablation. The rapid growth of CMR in the setting of catheter ablation over the past few years has yielded promising results. Future research is needed to overcome its current limitations and allow its implementation in the standard work-up of patients referred for AF ablation.