Atrial Fibrillation (AF) is the most common sustained arrhythmia in clinical practice and is becoming an increasing problem for providers worldwide as its prevalence increases.1 This phenomena will result in a significant global burden of arrhythmia care and care of its’ consequences as between 0.4-1% of the general population and 10% of the population greater than the age of 80 years will develop AF.2 It is also likely that predictions of AF burden are low as common risk factors that predispose individuals to arrhythmia such as diabetes, HF, obesity, sleep apnea, hypertension, and coronary artery disease are also increasing.3-5 Moving forward we will anticipate that AF and its’ consequences and disease associations will have a great impact on multiple aspects of medical practice making preventive strategies to minimize risk of the arrhythmia and its’ complications paramount to improve quality of life, mortality, and limit medical resource utilization.
Although AF has been considered a risk factor for adverse cardiovascular events, controversy remains regarding the exact role of AF. Questions remain as to if AF is mainly a risk marker of other diseases that actually convey risk such as HF, impaired diastolic filling, and hypertension versus an actual risk factor. In order to understand the independent risk of AF, we previously examined its presence on outcomes across the CHADS2 score profile (congestive HF, hypertension, age ≥ 75 years, diabetes, stroke/transient ischemic attack). In a comparison of 343 AF patients versus 2,945 non-AF patients studied over 9.1±1.8 years, AF across the CHADS2 score profile incrementally and independently increased risk of major adverse cardiac events, death, and stroke.6 In this general population study, these data were supportive of AF as an independent risk factor of long-term adverse cardiovascular events. This conclusion was further supported by a subsequent study utilizing the INSTRINSIC RV database.7 In this study of 1530 patients, those with a history of AF had higher rates of HF hospitalization, death, and ICD shocks compared to those with no history of AF. Furthermore, new onset AF in those without a history of AF also increased these risks significnatly.7 These studies support AF as an independent risk factor of adverse outcomes. To the extent that AF independently impacts adverse cardiovascular outcomes, this review article will focus on these outcomes, in particular HF, stroke, and mortality, and discuss contemporary strategies for treatment.
Atrial Fibrillation and Heart Failure
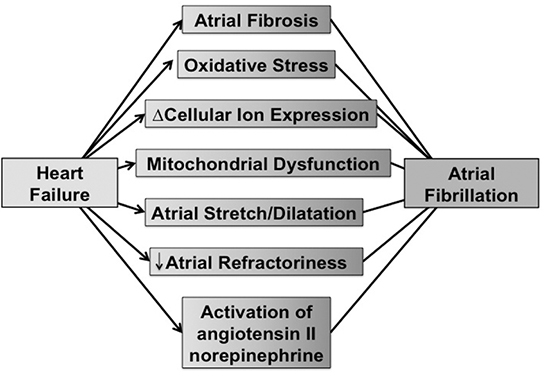
HF is a common clinical condition and shares many of the same baseline risk factors as AF. These diseases states can occur in independence, but are often seen together as a combined disease entity.8 In most cases of patients with both AF and HF, one condition predates the other allowing an understanding of which disease predisposed the patient to the other.8 However, the Framingham Heart Study demonstrated that one in five participants who developed both HF and AF were diagnosed with both conditions on the same day.3 Complicating the management of patients with AF and HF is that these conditions feed each other, adversely effecting therapeutic options and potentially increasing an endless circle of systemic complications, such as renal dysfunction, ICD shocks, stroke, dementia, impaired quality of life, and death (Figure 1).
Figure 1. The figure shows micro and macro drivers of atrial fibrillation in patients with preexisting HF.
There are clearly patients in which AF leads to rapid and at times very severe HF (Figure 2,Figure 3). Tachycardia-induced cardiomyopathy is one of the more straightforward examples of AF as an independent risk factor of cardiovascular risk. In a review of multiple animal studies and human clinical cases, Shinbane et al9 described anticipated outcomes with new onset AF with rapid ventricular rates. With rapid ventricular pacing, hemodynamic changes can develop as soon as 24 hours with progression to end stage HF in 3-5 weeks. However, with correction of the AF or rapid ventricular pacing, the myopathic process was reversible with pronounced recovery in 1 to 2 weeks.9 The reversibility and the temporal recovery of HF with restoration of sinus rhythm was also shown in a case series analysis by Grogan et al.10 In this study, patients that had AF, who were characterized as having idiopathic dilated cardiomyopathy, underwent a rhythm control strategy and showed an improvement of left ventricular ejection fraction from 25% to 52% (median follow up of 30 months). HF as a consequence of AF is relatively common, although the presentation and degree is variable. Statistics have shown that of the patients in the United States that have AF, 15-20% of them will subsequently be diagnosed with HF.1,3,8
Figure 2. The figure shows transechocardiographic images from a patient that presented with longstanding persistent atrial fibrillation prior to an anticipated ablation. Figure A shows the left atrial appendage with a large thrombus present. Figure B shows the pulse wave Doppler analysis of the appendage that is hypokinetic as verified by moderately reduced emptying velocities.
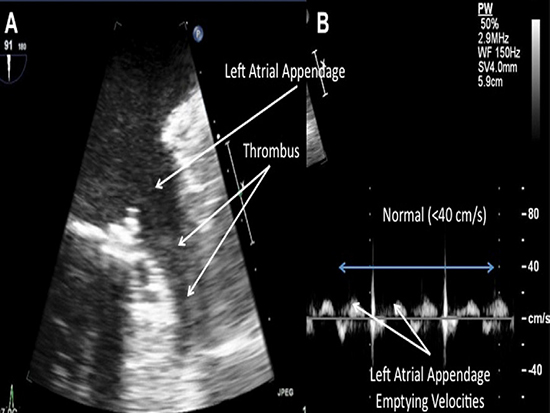
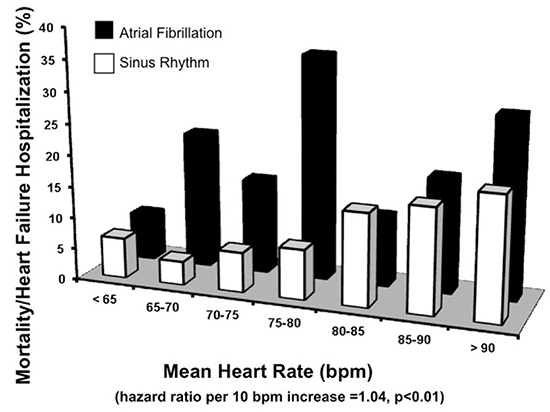
Figure 3. The figure shows the results of the INTRINSIV RV study regarding the impact of atrial fibrillation on risk of mortality and HF hospitalization. There was an increased risk of this endpoint across nearly all strata of heart rates.25
Just as HF is a consequence of AF, AF can be a consequence of ventricular dysfunction, either from systolic or diastolic HF. Men and women with HF are 4.5 and 5.9 times more likely, respectively, to develop AF than those without HF.11 In the Framingham Study, 17.1% of patients with a new diagnosis of HF subsequently developed AF.3 Severity of HF also influences risk; for example the prevalence of AF increases dramatically with worsening HF from 10% in NYHA class II to 50% in NYHA class IV HF.12 HF can result in altered cellular ion channel activities, changes in cellular ion expression, atrial stretch and enlargement, atrial fibrosisand increased sympathetic activation and as a result AF can be triggered and maintained (Figure 1).8,13-19 These micro and macro anatomic changes can negatively impact response to pharmacologic and nonpharmacologic treatments and increase risk for adverse outcomes such as stroke and mortality.8,20-21
The driving processes for AF associated with HF can be favorably impacted by optimizing use of medications such as angiotension receptor blockers and beta blockers.22-23 However, despite optimal contemporary pharmacologic management in patients with HF and an ICD, AF increases risk of worsening HF, ICD shocks, and mortality in patients enrolled in the INTRINSIC RV study.7 In this study of 1530 patients, 1356 (89%) had no history of AF at time of implant. Patients with a history of AF had higher rates of HF hospitalization [HR 2.14 (1.29-3.54), p<0.01], death [HR 2.22 (1.26-3.92), p<0.01], and any ICD shock [HR 1.75 (1.19-2.58), p<0.01] compared to those with no history of AF. Furthermore, new onset AF during the first three months of implant (45 of 1170, 4%) was associated with a significant increased risk of death [HR 2.86 (1.02-8.05), p=0.05] and a trend towards inappropriate ICD shock [HR 2.43 (0.87-6.75), p=0.09] and HF hospitalization [HR=1.17 (0.28-4.82), p=0.83].7
Choice and use of pharmacologic therapies in patients with the combined state of AF and HF remains important (Table 1). For example, use of carvedilol compared to metoprolol reduced the mortality impact of AF in patients with HF.22 There is active debate regarding mechanisms of benefit with beta blockade in AF patients with HF. One possibility is that the benefit was from rate control of rapid ventricular rates.24 However, when we performed a subanalysis of the INTRINSIC RV study,25 AF risk of mortality and HF hospitalization persisted across a broad heart rate strata compared to those without AF to suggest that rate alone does not account for the risks with AF and HF patients (Figure 2). In addition, ACE inhibitors, angiotensin receptor blockers (ARBs), and aldosterone blockers may favorably impact AF management and outcomes in patients with HF (Table 1).
Table 1. Medications for HF that impact AF incidence over study trial duration
| Medication
|
AF Incidence versus Placebo (%)
|
| Enalapril (SOLVD trial)75
|
5 / 24
|
| Trandolapril (TRACE trial)76
|
3 / 5
|
| Candesartan (CHARM)77
|
6 / 7
|
| Metoprolol (MERIT-HF)78
|
2 / 5
|
| Eplerenone (EMPHASIS-HF)80
|
3 / 5
|
Both pharmacologic and nonpharmacologic management of AF in HF are challenging with limited efficacies compared to their use in healthier states. Unfortunately use of these therapies has had only modest impact on the long-term outcomes of HF in the community over the past 2 decades, in particular mortality.26 Compounding these community data, the majority of antiarrhythmic drugs (AAD) increase mortality in HF through various mechanisms. The recently updated American Heart Association/American College of Cardiology guidelines list only amiodarone and dofetilide as first line options for rhythm control in HF, with the latter drug limited due to frequent concurrent useof diuretics and renal insufficiency in HF patients.27 Catheter ablation is considered second line in the event of medication failure or intolerance. Unfortunately, an aggressive pharmacologic rhythm control strategy with frequent cardioversions to maintain sinus rhythm long-term in HF did not favorably impact mortality, stroke, or worsening HF in the AF-CHF trial.28
Although rate control strategies fair favorably with AADs in the AF CHF trial, distinct and/or novel rhythm control strategies that are safe, tolerated, and durable may alter the neutral outcomes observed. For example, in the PABA-CHF trial, catheter ablation compared AV node ablation with biventricular pacing resulted in longer 6 minute walk times and higher on average ejection fractions at 6 months.29 The PABA-CHF trial largely studied the most aggressive minimally invasive rhythm and rate control strategies available, and showed improved outcomes in the rhythm control cohort in HF patients. Although catheter ablation was superior in the CABA-CHF trial, cardiac resynchronization still has mortality benefits in HF patients that develop AF. For example, cardiac resynchronization therapy in patients with HF that develop atrial tachyarrhythmias reduced mortality in the MADIT CRT trial.30
Although catheter ablation efficacy rates in HF patients are reduced compared to patients without HF, the outcomes are relatively favorable in comparison to those achieved with AADs. The catheter ablation data, similar to the PABA-CHF data, suggest that an aggressive rhythm control strategy, if successful, can positively influence HF outcomes. In a recent meta-analysis of 9 catheter ablation studies in AF patients with congestive HF, ejection fraction improved over long-term follow up on average by 11.1% (95% CI: 7.1-15.2, P < .001).31 The outcomes reported with catheter ablation coupledwith the largely poor outcomes reported with AADs Although catheter ablation efficacy rates in HF patients are reduced compared to patients without HF, the outcomes are relatively favorable in comparison to those achieved with AADs. The catheter ablation data, similar to the PABA-CHF data, suggest that an aggressive rhythm control strategy, if successful, can positively influence HF outcomes. In a recent meta-analysis of 9 catheter ablation studies in AF patients with congestive HF, ejection fraction improved over long-term follow up on average by 11.1% (95% CI: 7.1-15.2, P < .001).31 The outcomes reported with catheter ablation coupled with the largely poor outcomes reported with AADs prompt early consideration of catheter ablation in the management of AF in suitable patients with HF.
Atrial Fibrillation and Cerebral Vascular Disease
AF leads to an increased risk for ischemic stroke resulting from arterial thromboembolism and at least 15% of all ischemic strokes are associated with AF.32-34 Risk of cerebral thromboembolism in patients with AF increases with increasing CHADS2 risk score.35-37 Even in patients without AF, there is a linear increase in stroke across the CHADS2 risk score, but in comparison to patients with AF, the risks are consistently lower.6 Although generally applied to determine risk,the CHADS2 risk score is limited in its ability to make fine determinations of risk across diverse patients and to find patients with very low risk of stroke. The CHA2DS2-VASc scoring system which adds female gender, vascular disease, and age 65 to 74 years to the risk factors used in the CHADS2 score (Table 2) has been proposed to enhance risk stratification.37 However, both scores perform rather poorly with advancing age, particularly those >75 years.38-39 Likely due to ease of use the CHADS2 score remains the most commonly used scoring system in clinical practice.
Table 2. Atrial Fibrillation Risk Scores for stroke
| CHADS2 |
| Hypertension (1 point) |
| Age≥75 years (1 point) |
| Diabetes Mellitus (1 point) |
| Heart failure (1 point) |
| Ischemic stroke/TIA (2 points) |
| CHA2DS2-VASc |
| Hypertension (1 point) |
| Age ≥75 years (2 points) |
| Diabetes Mellitus (1 point) |
| Congestive Heart Failure or Ejection Fraction≤0.40 (1 point) |
| Stroke/TIA/Thromboembolism (2 points) |
| Vascular Disease (1 point) |
| Age 65-74 (1 point) |
| Female (1 point) |
Cardiovascular disease states that impact diastolic filling, such as HF, can result in morphologic changes that increase risk of stroke. For example, severe left atrial enlargement, decreased atrial transport, and left atrial appendage enlargement with reduced emptying flow velocities, increase blood stasis within the atrial appendage and risk of thrombosis (Figure 3). These morphologic and functional atrial changes can be easily studied by transesophageal echocardiogram and have been shown to significantly increase risk of stroke.40 Restoring sinus rhythm has been proposed as a means to minimize stroke risk. Unfortunately, in rate-versus-rhythm control trials such as AFFIRM trial,41 HOT-CAFÉ and CAFÉ II,42-43 PIAF,44 RACE,45 STAF,46 and AF-CHF,28 no significant stroke reduction was seen in the rhythm control cohorts. In the AFFIRM trial, there were actually more thromboembolism events in the rhythm control cohort which were felt to be due to inappropriate discontinuation of oral anticoagulation in the rhythm control arm. All of these trials had limitations that influence a pure rhythm versus rate control strategy analysis. These limitations include limited efficacy, tolerability, and potential toxicity of currently available AADs. However, the one take home message from AFFIRM in particular is that until we are certain of a significant improvements in rhythm control approaches, and their influence on stroke risk, we should treat the patient by baseline risks of stroke long-term.
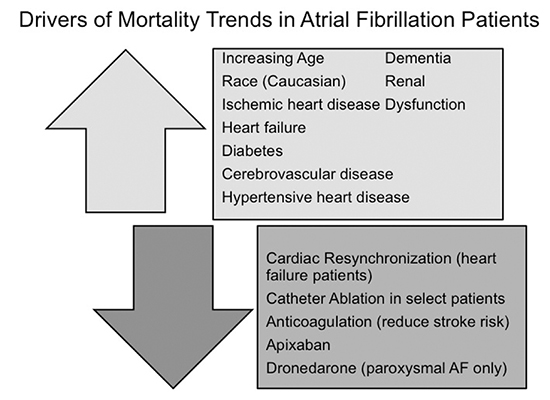
Figure 4. The figure displays potential positive and negative drivers of mortality in patients diagnosed with HF. The majority of positive drivers of mortality reflect the combined state of atrial fibrillation and other cardiovascular diseases. Therapies that may decrease mortality are limited.
Fortunately for patients, pharmacologic and nonpharmacologic strategies continued to evolve and improve. Newer therapies that are more effective, safe, and tolerable may allow for rhythm control strategies to reduce risk. For example, dronedarone decreased cardiovascular hospitalization and all cause mortality in the ATHENA trial.46 In a subanalysis of the ATHENA trial, dronedarone also decreased stroke risk compared to placebo despite similar anticoagulation strategies in both cohorts. Despite these optimistic findings for stroke, the safety of dronedarone in patients with AF subtypes other than paroxysmal and with common disease states with AF such as HF has not been favorable and as such limits its use broadly.47-48
Catheter ablation has been proven to be superior to drug therapy in suppressing AF and improving arrhythmia-based symptoms, quality of life and exercise capacity.49-52 Unlike pharmacologic therapies, which are limited long-term by ineffectiveness and toxicity, early analysis of long-term outcomes after catheter ablation for AF are favorable.53-54 Recently we evaluated all patients in a large system-wide study of Intermountain Healthcare who underwent catheter ablation for AF to understand the procedural impact on mortality, HF, stroke and dementia.55 A total 4,212 patients who underwent AF ablation were compared (1:4) to 16,848 age/gender matched controls with AF who did not receive ablation and 16,848 age/gender matched controls without AF. The AF ablation patients had a lower risk of stroke than AF patients without ablation. More importantly, the AF ablation patients had long-term stroke rates similar to the patients without any history AF. In patients that we had documented no recurrence of AF after ablation, stroke rates werelow at 1 year (1.0%), 3 years (1.7%), and long-term (1.7%).55 Just as important, long-term dementia rates were similar between those that underwent ablation and those with no history of AF-a finding suggestive of a favorable impact of sinus rhythm on the cerebral microvascular system. These findings in aggregate suggest that a durable rhythm control strategy may reduce stroke risk, although to what extent remains to be determined.
Although long-term continuation of anticoagulation based up on CHADS2 risk score has been recommended by the Heart Rhythm Society consensus statement56, observational studies have begun to emerge that suggest patients with moderate risk of stroke after catheter ablation may be safely managed with antiplatelet therapy alone. If these trials are correct, then they imply that catheter ablation that restores sinus rhythm reduces stroke risk. In one study of 327 patients [CHADS2score of 2 (45.4%) and 3 (23.2%)] in 68.8% of this cohort, oral anticoagulation was discontinued 6-12 months after catheter ablation if sinus rhythm was maintained and an antiplatelet agent started. No symptomatic ischemic cerebrovascular events were recorded during extended follow-up despite 91% of the patients having stopped oral anticoagulation and 89% their AADs.57 In another larger study of 3,355 patients of patients that underwent catheter ablation, 2,692 discontinued anticoagulation between 3-6 months. During a mean follow-up of 28±13 months no cerebral thromboembolic events occurred, despite discontinuation of oral anticoagulation in patients with a CHADS2 score ≥2. Just as important, in the group that remained on anticoagulation, 2% had a major hemorrhage illustrating the potential harm of these agents when used long-term.58 Treatment of patients with AF that had a stroke with long-term anticoagulation is equally important as this favorably impacts mortality.59
In addition to catheter ablation, surgical and nonsurgical approaches to remove or occlude the left atrial appendage have been advocated. In the setting of rheumatic heart disease, surgical removal of the appendage at the time of mitral valve replacement significantly reduced stroke (3% vs 17%).60 As such, removal of the appendage is considered standard of care for mitral valve surgeries to prevent postoperative thromboembolism. Unfortunately, when exclusion tools are used rather than excision, successful long-term closure is significantly reduced-a finding associated with risk of thromboembolism.60-61 Regarding catheter-based left atrial appendage occlusion devices, the most comprehensive data comes from the PROTECT AF trial using the Watchman device.62 In this trial, the primary endpoint of stroke, cardiovascular death, and systemic embolism was similar between patients receiving a Watchman device and those on long-term warfarin (3.0 vs 4.9 events per 100 patient years). The primary safety endpoint, a composite of major bleeding, pericardial effusion, procedure related stroke, and device embolization,was higher in the device group. The safety endpoint largely reflected procedural experience and has improved significantly with ongoing study.63
Anticoagulation strategies in all patients with AF, and in particular after catheter ablation, willcontinue to evolve with more available outcome data and as more patients are treated with anticoagulants that have a more favorable therapeutic index, less drug variability, and less risk of cranial hemorrhage compared to warfarin such a dabigatran,64 rivaroxaban,65 and apixaban.66 Of these, mortality was reduced with apixaban compared to warfarin group (3.52%/year vs. 3.94%/year; hazard ratio, 0.89; 95% CI, 0.80 to 0.99; P = 0.047) In aggregate new percutaneous left atrial appendage occlusion devices and these new novel anticoagulants are noninferior-superior to warfarin based primarily on a reduction in intracranial bleeding. Since cranial bleeding is the most significant endpoint influenced by these novel strategies moving forward assessment of this risk will be as important as risk of stroke in making a decision regarding which therapy to use. Similar to the CHADS2 score for stroke, risk stratification schemes such as HAS-BLED can assist in determining risk of cranial bleed.67
Atrial Fibrillation and Mortality
AF in general is associated with high long-term rates of both cardiovascular and total mortality.68 The impact on mortality is most pronounced in the elderly, particularly female with other cardiovascular disease states.32 Mortality often occurs early after incident diagnosis of AF (within the first 4 months).69 Unfortunately, there has been little impact to the observed mortality trends over the past two decades despite advances in both pharmacologic and nonpharmacologic therapies.69
At our center we have also found a substantial mortality associated with diagnosis of AF that increases steadily over 5 years of follow-up. In examining 16,848 patients diagnosed with AF from the Intermountain Healthcare database with at least 3 years of follow-up and that did not receive catheter ablation, we found that mortality rates were very high at 1 year (16.2%), 3 years (23.5%), and 5 years (27.9%).55Although these mortality rates were high, they are not unexpected given the adverse outcomes associated with AF coupled with the disease states that result in AF. Despite the frequent association with other cardiovascular diseases, the presence of AF in comparison to patients without AF independently increases mortality across the CHADS2 risk profile. AF also increasesmortality in patients optimally medically managed with HF and an ICD.3-7 AF impacts the body in general and across multiple organ systems resulting in higher rates of systemic inflammation, stroke, renal failure, HF, dementia, and reduces functional status and quality of life.6,8,11,55,70-71 As such it is not surprising that mortality rates in the community are high in patients with AF. Figure 4 highlights many of the negative and positive drivers of mortality in patients with AF.30,59,68,70, 72-73
Unfortunately, trials that have examined long-term mortality with rhythm versus rate control strategies such as AFFIRM and AF CHF showed no difference in mortality between the two strategies.28,41 In general, obtaining favorable long-term outcomes with AADs that may be proarrhythmic and cardiotoxic remains difficult. This challenge is no better illustrated than with the saga of dronedarone. In the ATHENA trial, dronedarone reduced the risk of death from cardiovascular causes by 29% and the risk of death from cardiac arrhythmia by 45%.74 However in patients with HF in the ANDROMEDA study, the data safety monitoring board terminated the study early after a median follow-up of 2 months, as 25 patients in the dronedarone group (8.1%) and 12 patients in the placebo group (3.8%) died (p=0.03). The cause of death in the dronedarone cohort was felt to be primarily related to worsening HF.48 In a subsequent study, dronedarone use in high risk permanent AF patients, increased all outcomes felt to be decreased in the ATHENA trial, specifically, cardiovascular mortality, stroke, and HF hospitalization.47
Similar to HF and stroke, catheter ablation may have a favorable impact on mortality. In our system wide analysis of Intermountain Healthcare patients that underwent AF ablation, compared to those with AF that did not, and patients with no history AF, mortality rates were low after ablation and similar to patients with no AF. For example, at 1 year, mortality after catheter ablation was 3.0% versus 4.7 in patients with no history of AF. 5-year mortality after catheter ablation was 7.6% versus 8.7% in patients with no history of AF. These mortality rates from the cohorts of patients that received a catheter ablation for symptomatic AF and patients with no history of AF aresignificantly lower than AF patients in general.55 Similar to the other outcomes discussed, these data suggest that if the rhythm control strategy is safe and durable, mortality as an outcome can be reduced. Nonetheless, our mortality data are observational and as such require confirmation in a prospective randomized study.
All outcomes with catheter ablation must be considered in the context of the majority of studies are for 1 year with few extending beyond that time frame. The favorable findings we discuss may change as we continue to follow these patients over longer periods. Fortunately the recently approved NHLBI sponsored trial Catheter Ablation Versus Antiarrhythmic Drug Therapy for Atrial Fibrillation (CABANA) will provide much needed long-term randomized outcomes after catheter ablation compared to contemporary AAD management approaches. The trial is powered to examine the possibility of a mortality benefit with catheter ablation. The trial also includes frequent ambulatory cardiac monitoring which will provide novel insight in a very large comparative study population regarding the role of AF burden on outcomes.