The Relationship Between Atrial Fibrillation and Chronic Kidney Disease : Epidemiologic and Pathophysiologic Considerations for a Dual Epidemic
David D. McManus MD 1, 2, Jane S. Saczynski PhD 1, 2, Jeanine A. Ward MD PhD3, Khushleen Jaggi MD1, Peter Bourrell MD1, Chad Darling MD3, Robert J. Goldberg PhD2
1Department of Medicine, Division of Cardiology, Section of Electrophysiology, University of Massachusetts Medical Center, Worcester, MA, USA.2Department of Quantitative Health Sciences, University of Massachusetts Medical School.3Department of Emergency Medicine, University of Massachusetts Medical Center.
Atrial fibrillation (AF) presently affects over 2 million Americans, and the magnitude and population burden from AF continues to increase concomitant with the aging of the U.S. population. Chronic kidney disease (CKD) is present in 13% of individuals in the U.S., and the prevalence of CKD is also rapidly increasing. The increasing population burden of CKD and AF will profoundly affect the clinical and public health, since CKD and AF are both associated with lower quality of life, increased hospitalization rates, and a greater risk of heart failure, stroke, and total mortality. AF and CKD often co-exist, each condition predisposes to the other, and the co-occurrence of these disorders worsens prognosis relative to either disease alone. The shared epidemiology of CKD and AF may be explained by the strong pathophysiologic connections between these diseases. In order to promote a better understanding of CKD and AF, we have reviewed their shared epidemiology and pathophysiology and described the natural history of patients affected by both diseases.
Correspondence to:
David D. McManus, MD, Departments of Medicine and Quantitative Health Sciences , University of Massachusetts Medical Center, 55 Lake Avenue North, Worcester, MA 01655.
Atrial fibrillation (AF) is presently the most common sustained dysarrhythmia among Americans and the global burden of AF continues to grow.1-3 The prevalence of AF increases dramatically with advancing age and this arrhythmia is present in nearly 1 in every 10 individuals by the age of 80 years.3,4 The increasing population burden of AF has a profound impact on clinical and public health through its association with lower quality of life,5,6 increased hospitalization rates, and a greater risk of heart failure,7,8 stroke,9 and all-cause mortality.10-14
Chronic kidney disease (CKD) affects approximately 13% of the U.S. population and the national prevalence of end-stage kidney disease more than doubled between 1991 and 2004.15,16 Similar to the descriptive epidemiology of AF,17 the incidence of CKD increases with advancing age and progression to end-stage renal disease (ESRD) adversely affects the quality of life and longevity of millions worldwide.18 AF and CKD frequently coexist,19 each condition predisposes to the other,20 and the presence of concomitant AF and CKD worsens patient’s long-term prognosis relative to either disease alone.21,22
The shared epidemiology of CKD and AF may be explained by strong pathophysiologic connections between these two diseases (Table 1). Subclinical atrial remodeling, a process characterized by atrial structural and electrophysiological changes over time, is an important contributor to the initiation and maintenance of AF in many individuals.23,24 Even mild renal impairment may promote or accelerate atrial remodeling by up regulating neurohumoral (e.g., TGF-β) and pro-inflammatory (C-reactive protein) signaling cascades, promoting volume retention and imposing hemodynamic stress on the atria.25-27 CKD also results in chronic sympathetic nervous system activation, which may cause AF.28-29 On the other hand, and reflecting the inter-connections between these 2 diseases, AF can promote or accelerate the progression of CKD by decreasing cardiac output, through its association with heart failure and thromboembolic events, and by promoting the development of the cardiorenal syndrome (Table 1).30
In light of the growing number of patients affected by both CKD and AF, we have undertaken this work to review the mechanistic and epidemiological parallels between these two diseases, and the strong connections between AF, CKD, and several important cardiovascular risk factors. We have also drawn from the published literature on the natural history of AF in order to promote the evidence-based treatment of AF among patients with concomitant CKD.
Epidemiology of Atrial Fibrillation in Patients with Chronic Kidney Disease
Our group and others have shown that reduced kidney function and the presence of albuminuria are strongly associated with an increased risk for developing AF.18,31 Contemporary estimates suggest that individuals with ESRD and AF are 50% more likely to be hospitalized, and approximately two-thirds more likely to die, when compared to individuals in normal sinus rhythm.11 The relationship between kidney dysfunction and risk for AF is J-shaped, with the lowest risk of AF seen among patients with the least significant reduction in kidney function (Figure 1).18 However, the prevalence of AF is high even in patients with mild or moderate CKD, with contemporary estimates suggesting that nearly one in every five persons with Stage 1-2 CKD has AF.19,32
Figure 1. Cumulative incidence of Atrial Fibrillation by categories of estimated glomerular filtration rate in whites (top) and blacks (bottom)
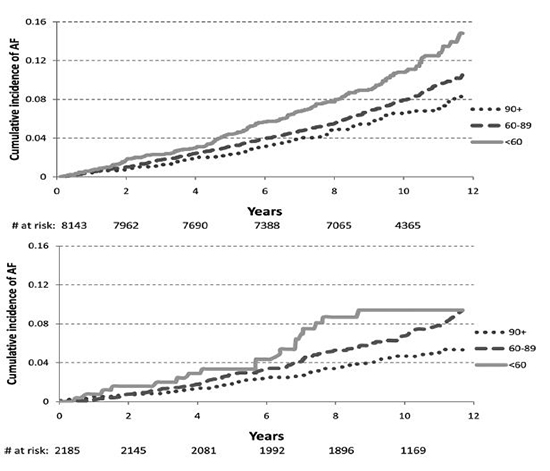
From the Atherosclerosis in Communities study. Reprinted with permission from Alonso, A. Circulation 2011
Epidemiology of Chronic Kidney Disease in Patients with Atrial Fibrillation
Although data are more limited with respect to the influence of AF on the incidence and progression of kidney dysfunction, AF is strongly and independently associated with incident CKD.20 In an international study involving over 235,000 men and women recruited at an ambulatory clinic visit, AF was associated with 1.8-fold higher odds of developing kidney dysfunction and 2.2-fold higher odds of developing proteinuria over a 6-year follow-up period.20 Similar to the current “epidemic” of AF, the recent national increase in new cases of CKD may be secondary to the aging of the population and an increase in the prevalence of several common important comorbidities, including diabetes, hypertension, and coronary heart disease.33-38
Atrial Fibrillation in Chronic Kidney Disease: Shared Predisposing Factors
AF, like CKD, disproportionately affects older individuals, with approximately one-half of all AF cases occurring in persons 75 years and older.4,39AF shares strong epidemiologic associations with other cardiovascular diseases and risk factors, such as coronary artery disease, hypertension, valvular heart disease, heart failure, obesity, diabetes mellitus, and obstructive sleep apnea.17,40 CKD also predominantly affects older Americans, and many of the chronic disease conditions and risk factors that predispose to AF are also associated with the development of CKD. Increasing rates of shared risk factors provide insights into the changing, and increasingly interwoven, epidemiology of AF and CKD.
Although the prevalence of CKD is higher among African Americans than among white individuals, the rate of progression from milder CKD to ESRD, and death rates from ESRD are higher among white as compared with non-white patients.41 Even though CKD is more commonly observed in women, women with CKD have a more favorable prognosis than men with CKD.42 It is notable that racial and sex differences in predisposition to AF also exist, with white individuals and men at higher risk for AF than African Americans and women, respectively.33 Although the epidemiology of AF in African Americans and women with CKD is poorly characterized, we hypothesize that an increased prevalence of AF among men and whites with CKD may cause a more precipitous decline in renal function and clinical status when compared with women and non-white individuals with CKD.32
Diabetes mellitus is associated with a markedly increased risk for AF in both American men and women.33 Diabetes also accounts for upwards of 2 in every 5 new cases of kidney failure in the U.S.35 Treatment of diabetes with insulin-sensitizing thiazolidinedione therapy has been shown to reduce recurrences of AF.44 In light of the association between increased adiposity, diabetes, AF and CKD, insulin resistance has been hypothesized to represent an important intermediate step in the progression to AF.45 To date, however, data are conflicted with respect to the role of insulin resistance in mediating the increased risk for AF among persons with pre-diabetes and diabetes.46
Microalbuminuria is common in diabetics and relates strongly to the risk of developing CKD.47-49 Acute or sustained hyperglycemia can cause microvascular damage in the kidney, leading to increased glomerular permeability and albuminuria.50-51 Albuminuria has been identified as an independent predictor of AF, irrespective of renal dysfunction.52 Since approximately one-half of patients with diabetes have at least some degree of albuminuria,47 a high prevalence of proteinuria may explain the significant burden of AF in diabetic populations.53 Hyperglycemic-induced renovascular disease promotes up-regulation of neurohumoral signaling cascades that also lead to development of pathological cardiac remodeling phenotypes associated with AF (Table 1).50 Further investigations are needed to investigate the mechanisms underlying the associations between diabetes mellitus, albuminuria, and AF.
Table 1. Common Risk Factors and Mechanisms between Atrial Fibrillation and Chronic Kidney Disease
| Common AF and CKD Risk Factors |
Age, hypertension, obesity, diabetes mellitus, valvular heart disease, acute coronary syndrome, heart failure |
| Common Intermediate Phenotypes |
LV hypertrophy, LA enlargement |
| Cardiac Remodeling Mechanisms in Chronic Kidney Disease |
|
| Neurohumoral signaling mechanisms |
Renin-angiotensin-aldosterone system, TGF-β1, sympathetic nervous system up-regulation |
| Inflammatory Mechanisms |
Increased systemic inflammation – increased circulating levels of CRP, TNF-α, fibrinogen, IL-6, increased oxidative stress |
| Stretch-related Mechanisms |
Abnormal calcium handling, shortened action potential duration, decreased refractoriness, dispersion of the refractory period |
| Fibrosis-related Mechanisms |
Disrupted cell-cell junctions, areas of delayed conduction, facilitation of re-entry |
MI = myocardial infarction, LV = left ventricular, LA = left atrial, TGF = tissue growth factor, CRP = C-reactive protein, TNF = tumor necrosis factor, IL = interleukin
Individuals with CKD are significantly more likely to have underlying coronary artery disdisease than men and women without impaired renal function,54 and prevalent CKD is an independent risk factor for subsequent adverse cardiovascular events.12 A history of prior coronary heart disease, and development of a myocardial infarction, are established risk factors for AF, conferring a 1.5 to 3-fold increased risk for AF.55 Cardiogenic shock and left ventricular dysfunction are more commonly observed following acute myocardial infarction in patients with, than in those without, CKD, providing a potential hemodynamic link between prevalent CKD, coronary artery disease, and AF.56
Hypertension is both a cause and consequence of CKD, with the majority of CKD patients affected by systemic hypertension.57 Moreover, with sodium retention, activation of the renin-angiotensin aldosterone system (RAAS), and sympathetic nervous system up-regulation, hypertension becomes increasingly refractory to treatment and more severe in advanced stages of CKD.58 This has important implications for both the development and management of AF, since the duration and intensity of exposure to elevated systolic and pulse pressures are critical contributors to pathological atrial structural remodeling over adulthood.23,59,60 Hypertension is associated with a markedly increased risk for AF and each 20-mmHg increase in pulse pressure may elevate the risk of AF by as much as one-quarter.61,33 Notably, the concomitant presence of hypertension and diabetes mellitus may also have a synergistic effect on the likelihood of developing AF, as diabetes has been associated with a 1.5-fold higher risk of AF among individuals with hypertension.62 It remains unclear, however, why the combination of diabetes and hypertension has such a profound effect on the risk for developing new-onset AF, but amplified activation of neurohumoral and catecholinergic signaling pathways common to CKD and AF is a likely explanation.
The ongoing obesity epidemic in developed countries has important public health implications in light of the known relations of increased adiposity with CKD and several common cardiovascular diseases, including AF.2,57,63-65 Poor kidney function is strongly associated with higher body mass, and each unit increment in body mass index increases the odds of CKD by approximately 5%.65 Obesity indirectly contributes to the development of CKD through its association with several underlying risk factors and predisposing conditions including diabetes, hypertension, and atherosclerosis.66 Obesity may also directly lead to the development of CKD; several epidemiologic investigations have shown that individuals with obesity are at a 3-fold higher risk for CKD, even after adjustment for blood pressure and presence of diabetes.63
Obese individuals are also at higher risk for AF, with each unit increase in body mass index conferring an approximate 4% increased risk for developing AF.67 Atrial structural remodeling appears to play an important role in mediating the relation between increased adiposity and risk for AF.67 The strong relations of the obesity-related disorder obstructive sleep apnea with CKD and AF also highlight the significant and complex associations between obesity, CKD, and AF. Sleep apnea is increasingly recognized as an important risk factor for AF and CKD,68 and the obstructive sleep apnea syndrome is also related to the underlying progression of both diseases.69 Mechanisms connecting obesity to the development of CKD and AF may be interlinked through crosstalk between fat, the kidney, the liver, and the heart through circulating signaling molecules such as fetuin A, adiponectin, resistin, and 5′-AMP activated protein kinase.70,71
Among patients with CKD, particularly those with end-stage renal disease, dystrophic vascular and valvular calcification is common and is likely secondary to chronic hyperphosphatemia.72,73 Dystrophic valvular calcification can lead to mitral annular calcification and aortic stenosis, two valvular disorders strongly linked to left ventricular hypertrophy, atrial enlargement, and AF.74 In fact, the presence of valvular heart disease increased AF risk by 3.4- and 1.8-fold in female and male Framingham Heart Study participants, respectively.17 Since even mild aortic stenosis and mitral regurgitation impose hemodynamic stress on the atrium and ventricle, valvular heart disease may accelerate atrial structural and electrical remodeling and is another comorbid illness that promotes the development of AF in patients with CKD (Figure 2).
Figure 2. Mechanisms Linking Atrial Fibrillation to Chronic Kidney Disease
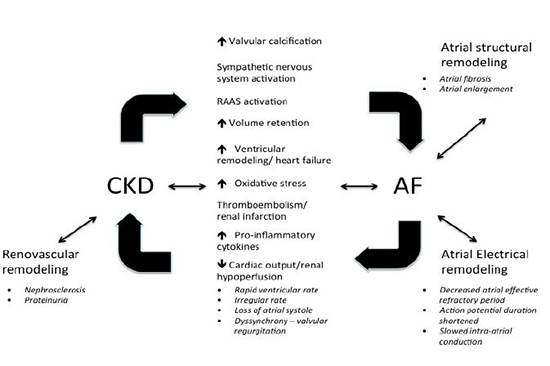
CKD = chronic kidney disease; AF = atrial fibrillation; RAAS = renin-angiotensin-aldosterone system
The majority of individuals with heart failure have significant renal impairment, and worsen ing renal function in persons hospitalized with heart failure is associated with an increased risk for adverse cardiac events.75-77 CKD is associated with a 1.4-fold and 2.4-fold higher risk for new-onset heart failure in men and women, respectively, compared to persons with normal renal function.78 Although common risk factors such as diabetes and hypertension contribute to the association between CKD and heart failure, the incidence rates of heart failure are higher among patients with CKD than would be expected on the basis of risk factors alone.79
AF and HF also frequently coexist, with 30% of all individuals with HF affected by AF.7,80 Individuals with AF are at significantly higher risk for developing HF in comparison to persons in normal sinus rhythm.7,8,30,81 Conversely, the diagnosis of HF is associated with a greater than 5-fold increased risk for development of AF, highlighting the strong connection between these two increasingly prevalent disorders.7 As we discuss in the next section, subclinical ventricular remodeling and clinical HF may represent a common pathway through which CKD promotes AF, and vice versa (Figure 2).
Pathophysiology of Atrial Fibrillation as it relates to Chronic Kidney Disease
Chronic kidney disease imposes hemodynamic, inflammatory, catecholinergic, neurohumoral, and metabolic stress on the heart (Figure 2).28,58,82,83 Subclinical phenotypes, such as left ventricular hypertrophy and atrial enlargement, are associated with CKD and develop in response to these stressors.84-86 Although ventricular and atrial remodeling may appear to be adaptive, enabling the heart to compensate for increases in preload and/or afterload imposed by CKD, these compensatory mechanisms may eventually become maladaptive and lead to a decline in cardiac function. Not surprisingly, increased echocardiographic left ventricular wall thickness and electrocardiographic left ventricular hypertrophy, two markers of ventricular remodeling commonly seen in patients with CKD, are also strongly associated with atrial enlargement and AF.59,85
Renin-Angiotensin Aldosterone System
Chronic kidney disease activates a number of stress-signaling pathways, including the RAAS. RAAS up-regulation results in impaired vascular growth, interstitial myocardial fibrosis, and myocyte apoptosis.87 Up-regulation of the RAAS may lead directly to AF through salt retention and atrial distension. Chronic RAAS activation also contributes to atrial remodeling independent of its hemodynamic effects in experimental studies of AF.88 Key components of the RAAS, including Angiotensin-converting enzyme, Angiotensin II (AII) and transforming growth factor-beta 1 (TGF-β1), are up-regulated in a systemic and tissue-specific manner in patients with CKD.87,89 The electrophysiologic and structural effects of chronic RAAS activation act in concert to promote the development of an atrial substrate that is vulnerable to AF (Figure 2).89-91 Although the benefits of RAAS inhibition with respect to AF prevention have not been systematically studied, data support the hypothesis that RAAS inhibition may reduce the incidence of AF in high-risk subgroups, such as those with CKD.89,92
Oxidative Stress and Inflammation
It is well known that CKD is associated with increased levels of circulating pro-inflammatory cytokines,26 perhaps because the accumulation of uremic toxins causes an endothelial inflammatory response.93 Exposure of endothelium to uremic toxins leads to changes in endovascular cellular phenotypes and production of many pro-inflammatory molecules,94-95 often leading to increased vascular stiffness, accelerated atherosclerosis, and the development of adverse cardiac remodeling phenotypes (Figure 2).86
Systemic inflammation and endothelial dysfunction play an important role in the pathogenesis of AF (Figure 2).25,96 The key role of inflammation in the development of AF is highlighted by the fact that histologic evidence of inflammation is seen in two-thirds of patients who go on to develop this arrhythmia.97 In particular, serum levels of interleukin-6, fibrinogen, and C-reactive protein, three important markers of inflammation that are elevated in patients with CKD,98 are also predictive of future episodes of AF.82,99 Over-expression of tumor necrosis factor-α (TNF-α), another inflammatory cytokine, is also associated with atrial fibrosis, ion channel dysfunction, and increased susceptibility to AF.100 Consistent with this working hypothesis, several studies have shown that anti-inflammatory agents, such as glucocorticoids, and medications with anti-oxidative properties, such as statins, may prevent AF.101 Further study into the anti-arrhythmic effects of several commonly used drugs with anti-inflammatory effects, particularly statins, in patients with CKD appears warranted.102-104
Sympathetic Nervous System Up-Regulation
CKD leads to chronic up-regulation of the sympathetic nervous system, as evidenced by the fact that kidney dysfunction is associated with lower heart rate recovery, an established marker of autonomic function, after exercise testing.105-108 Since enhanced automaticity in the left atrium and pulmonary veins is considered a major contributor to the pathogenesis of AF, kidney dysfunction may lead to AF through its association with sympathetic nervous system up-regulation.109 Enhanced autonomic tone during dialysis, along with acute changes in circulating volume and electrolyte concentrations, may explain why CKD patients on hemodialysis are at particularly high risk for AF (Figure 2).110
Chronic Kidney Disease and pathological atrial remodeling
Atrial remodeling – a process describing progressive atrial electrical and structural changes in response to chronic or repeated atrial injury– merits attention in understanding the pathogenesis of AF in the setting of CKD (Figure 2).90,91 Experimental models of AF show that atrial remodeling often precedes development of AF.90,91 Pathologic atrial remodeling is related to the presence and severity of CKD.86 Chronic intra-atrial or intra-ventricular pressure overload from CKD activates stress signaling pathways, most notably the RAAS, yielding impaired myocardial vascular growth, myocyte apoptosis and interstitial fibrosis as discussed above.23,31 Chronic RAAS activation, as evidenced by increased levels of circulating AII and TGF-β1, has been shown to contribute to atrial fibrosis and remodeling in AF experimental studies.90 Experimental data also show that the RAAS up-regulation also influences ion channel function and action potential duration, predisposing to intra-atrial reentry and AF. Independent of its effects on the RAAS, CKD may also contribute to pathologic atrial remodeling through its associations with sympathetic nervous system up-regulation, systemic inflammation, and valvular heart disease.8
Natural History of Atrial Fibrillation in Individuals with Chronic Kidney Disease
CKD is an independent predictor of adverse cardiac events among patients with AF.11 Long-term all-cause mortality rates, risk of non-fatal myocardial infarction and stroke, and hospitalizations for heart failure are markedly higher in individuals with AF and ESRD compared to those with ESRD alone.9-12 Markers of subclinical cardiac remodeling that accompany AF, including echocardiographic atrial enlargement, are also associated with an increased risk of dying and adverse cardiovascular events among patients with CKD.111 Although longitudinal data on the natural history of AF among patients with CKD not requiring renal replacement therapy are lacking, the presence of both CKD and AF identifies a subgroup of individuals at particularly high risk for thromboembolic events.13 After adjustment for known stroke risk factors, the presence of proteinuria increases the risk of thromboembolism by more than 50%, and there is a graded, increased risk of stroke associated with progressively lower estimated glomerular filtration rate.52 The association between CKD and stroke may be mediated by ongoing subclinical inflammation.112
Implications of Chronic Kidney Disease on the Treatment of Individuals with Atrial Fibrillation
Despite the fact that patients with both AF and CKD are at significantly higher risk of stroke than patients with either disease alone, renal insufficiency also increases the risk of bleeding with the use of antiplatelet and anticoagulant agents, and the optimal anticoagulation strategy for patients with AF and CKD remains unclear.9,52,113,114 The debate over which anticoagulant to use for patients with AF and CKD is unlikely to be resolved soon, however, since patients with CKD have been systematically excluded from most clinical trials of anticoagulants used in the setting of AF.
CKD increases the risk of complications from anti-arrhythmic drugs, such as digoxin, sotalol or dofetilide, which undergo renal elimination.115,116 CKD is also an independent predictor of arrhythmia recurrence after catheter ablation for AF.117 This association may be mediated by the association between CKD and atrial structural remodeling.117 CKD has also been identified as a major predictor of complications after catheter ablation in patients with underlying AF.117 In a cohort of more than 1,600 patients undergoing ablation for AF as well as several other arrhythmias, renal insufficiency was associated with a 2.5-fold higher rate of major peri- and post-operative complications.118 Additional prospective investigations of patients with CKD and AF are needed to determine how CKD affects treatment response and complications from catheter-based AF ablation.
In part due to the increasing prevalence of shared cardiovascular risk factors such as diabetes, obesity, and heart failure, and the aging of the population, the number of individuals in the U.S. and other developed countries affected by AF and CKD is growing rapidly. Independent of shared risk factors, patients with CKD are at increased risk for developing AF, and vice versa, suggesting strong mechanistic connections between these two conditions. Chronic activation of neurohumoral, pro-inflammatory, and catecholinergic signaling pathways in individuals with CKD promote cardiac structural and electrophysiologic changes over time, enhancing vulnerability to AF. Patients with both CKD and AF are at significantly increased risk for adverse cardiac events, including thromboembolism and death. In light of the fact that patients with CKD are often excluded from clinical studies involving patients with AF, the optimal strategy for managing patients with AF and concomitant CKD remains unclear. It is apparent, however, that patients with AF and CKD are at high risk for AF-related treatment failure and complications. Further longitudinal studies involving patients with CKD and AF are needed in order to determine how this important group of patients responds to contemporary AF treatments, including catheter ablation as well as novel anti-coagulants and anti-arrhythmic medications.