Risk Factors for the Development of Atrial Fibrillation in HIV Infected Patients
Yaser Elnahar MD, Joseph Daoko MD, Anthony Al-Dehneh MD, Nishant Gupta MD, Vincent A. DeBari PhD, Fayez Shamoon, Constantinos A. Costeas MD
Department of Cardiology, Seton Hall University, St. Michael Medical Center, NJ
.
Patients with Human Immunodeficiency Virus (HIV) have an array of multi-organ involvement, including cardiovascular disease. CD4 count is one of the best parameters to monitor the severity of HIV disease. The arrythmogenic potential of HIV disease has not been well defined. The aim of the study is to establish whether an association between the severity of HIV and atrial fibrillation (AF) exists.
Out of a retrospective cohort of 780 HIV patients from January 2006 to December 2008, 40 patients were selected that developed AF during this period .The age and sex matched controls (n=40) were selected for comparison. The comparison between both groups was done using Fischer Exact Test. Bivariate and multivariate analysis was also performed to analyze the results.
The data shows that 47%(19/40) of the patients with HIV who developed AF had CD4 count lower than 250 as compared to 20%(8/40) in the control group (P value = 0.017)
The data supports the presence of a relationship between HIV and AF. Patients with lower CD4 counts are more susceptible to develop AF.
Correspondence to: Yaser Elnahar MD, Cardiology Fellow, Division of Cardiovascular Diseases, Seton Hall University, St. Michael Medical Center, 111 Central Avenue, Newark, NJ 07109.
Despite a declining rate of HIV (Human Immunodeficiency Virus) related death, proportions of HIV infected patients dying of other causes have increased.1 As an example, a death certificate study in New York City showed that the proportion of deaths among HIV-infected patients due to non–HIV-related causes increased from 19.8% to 26.3% between 1999 and 2006, reflecting mortality resulting from cardiovascular disease (CVD), substance abuse, and non–AIDS-defining cancers.2 Among individuals aged 55 years or older, CVD was the leading cause of death.
Atrial fibrillation is associated with a wide range of organ damage, ranging from stroke to heart failure. Furthermore, management of patients with this arrhythmia is a challenge. Once patients develop AF, it is incumbent upon the physician to balance rate control, anti coagulation and if needed rhythm control. Although there are many risk factors for developing this arrhythmia, there has not been any data to show that HIV is a risk factor.
Patients with HIV have an array of multi-organinvolvement, amongst which is cardiovascular disease. Cardiovascular manifestations include but are not limited to dilated cardiomyopathy, pericardial effusion, endocarditis, myocarditis, pulmonary hypertension and atherosclerotic heart disease. Literature review shows that there aren’t any published data on the arrythmogenic potential of HIV. This retrospective observational study was conducted to determine if there is an association between HIV and AF. Furthermore, we wanted to elucidate why certain HIV patients are more prone to developing AF.
This was a multi-center retrospective study at three urban teaching hospitals in the north eastern United States that are affiliates of the School of Health and Medical Sciences of Seton Hall University. The protocol was approved by the Institution Review Boards (IRB) of St Michael’s Medical Center (Newark, NJ), St Joseph’s Medical Center (Paterson, NJ) and Trinitas Regional Medical Center (Elizabeth NJ). Seton Hall University’s IRB delegates the approval process to the individual facilities at which the studies are performed.
This study examined a retrospective cohort of 780 HIV patients that developed AF after developing HIV during a period of 3 years from 2006-2008, inclusive. Within this cohort, we nested a case-control study consisting of subjects with AF (n=40) and subjects who did not demonstrate AF (n=40) during the above mentioned time period. The inclusion criteria for the study group were: HIV patients above the age of 18 and AF that developed after the diagnosis of HIV. All of the cases were persistent atrial fibrillation. In addition, all of the patients were treated with a rate and anticoagulation strategy; none of the patients were placed on rhythm control agents, such as amiodarone. This approach was primarily due to many of the interactions with most anti-retro viral medications. Furthermore, our approach was further attributed to the absence of strong data to support rhythm control, once rate control and anticoagulation has been addressed. Each patient was anticoagulated according the chads2 stroke risk. During the three year followup we did not have any thrombo-embolic events that were recorded at our institution. Whether these patients had a complication and were admitted to other institutions, we cannot confirm.
All medications were reviewed extensively in every patient included in the study; there were no patients that were taking any known arrhythmogenic agents. Patients that were on arrhythmogenic agents were excluded, even if they fit the other inclusion criteria.The CD4 count was measured at the time of diagnosis of atrial fibrillation. All of these patients follow up in our HIV clinics and access to their CD4 count at the time of diagnosis of AF was possible. Patients were excluded from the study if they had a history of AF prior to the development of HIV. Baseline characteristics included: age, sex, history of hypertension, diabetes mellitus, dyslipidemia, echocardiography findings of an EF <35% and left atrial enlargement (LAE) > 4cm.
Interval data (age) were tested for normality with the D’Agostino-Pearson omnibus normality test and were found to approximate a normal distribution. Thus, age was compared by an unpaired t-test. Categorical risk factors were cross tabulated with the two outcomes (presence/absence of AF) and tested for significance with Fisher’s exact test. Because of the retrospective nature of the study, clinical relevance was described by the odds ratio and 96% confidence intervals (CI). Characteristics for which univariate p values were ≤0.05 and lower 95% CI was ≥ 1.0, were considered as primary risk factors (RF). The level of significance for potential confounders was p ≤ 0.25. These were examined for interaction with the RF by logistic regression analysis and, if a significant interaction was observed, it was included in a model to adjust the RF for the confounder. For univariate analyses, we used Prisms® software (GraphPad Corp., San Diego). For bivariate logistic regression analysis we used a web-based routine.15
The two groups were compared with regard to baseline characteristics in Table 1. Based on the criteria described in the Methods, the only RF detected was CD4+ count < 250 cells/mm3, which yielded an OR = 3.62 (95% CI: 1.34 to 9.77). Several potential confounders (p ≤ 0.25) were identified: HTN, dyslipidemia and LAE. These were used to adjust the odds ratio and the resulting data are provided in Figure 1. It can be seen that adjustment for both LAE and HTN tend to demonstrate a dependence of CD4+ cell count on these parameters. However, when tested, the p – value for the interaction of both of these potential confounders with CD4+ cell count was not statistically significant (p = 0.14 for LAE and p = 0.31 for HTN).
Table 1. Baseline characteristics of the two groups. Gender is given as M/f and shown as mean +/- 1 SD; all other variables are those with the characteristics/those without.
|
HIV & AFIB |
P values |
| Gender |
27/13 |
26/14 |
1.00 |
| Age |
56.78 +/- 9.4 |
54.59 +/- 8.59 |
0.273 |
| HTN |
27/13 |
19/21 |
0.113 |
| DM |
11/29 |
10/30 |
1.00 |
| dyslipidemia |
13/27 |
300 |
5/35 |
0.059 |
| EF <35% |
10/30 |
5/35 |
0.252 |
| LAE |
13/27 |
5/35 |
0.059 |
| CD4 <250 |
19/21 |
8/32 |
0.017 |
Figure 1. The adjusted odds ratio for HTN, dyslipidemia and LAE.Model
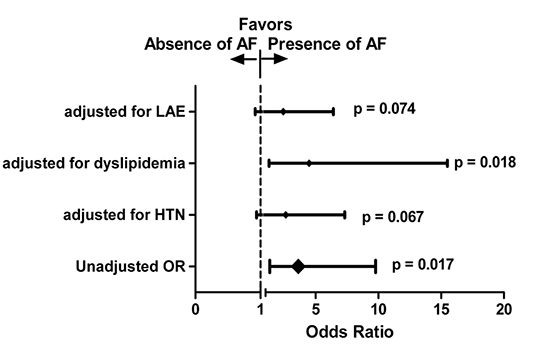
HIV infection is multisystemic and even with in an individual organ, it can manifest in multiple ways. The purpose of this study was to evaluate if patients with AIDS are more likely to have the arrythmogenic effects of HIV. The results support the hypothesis by the presence of a statistically significant difference in the prevalence of AF between the two groups. This association was still significant when we considered dyslipidemia as a confounding factor. However, the association between CD4 count and AF was not as significant when we factored in HTN and LAE as a confounding variable. This result can be explained by two factors: firstly, hypertension is on its own the most common risk factor for developing AF. Secondly, the number of patients in the study is small, thus limiting our power. Despite the fact that HTN and LAE favored CD4 not being an independent risk factor, a larger study might reverse that and is certainly warranted. Further, when the odds ratios were adjusted for both HTN and LAE, the P value approached a significant number (Figure 1) and thus demonstrates a dependence of CD4+ cell count on these parameters. As such, HIV infected patients with low CD4 counts have a greater risk of developing AF than those with better immune status. One possible explanation for this relationship may be through myocarditis. The aforementioned relationship may be a function of the severity of HIV infection. The incidence of AF was 5% (40 out of 780) which is not significantly more than the general population (1.2-6%) and AF was seen more in the patients with low CD4 counts. The true incidence of AF is likely higher; however, we had stringent exclusion criteria to shun any confounders. In addition to the listed exclusion criteria, all patients on any arrhythmogenic agents were excluded at the time of admission with AF. Further, patients that did not have a CD4 count at the time of diagnosis of AF were excluded. As such, we suspect that the incidence is higher than 5%. As an example there were patients that were excluded because we did not know which occurred first, the AF or HIV. This study was aimed to show that a low CD4 count is a risk for developing AF and warrants further investigations; the design was not to measure the incidence.
Atrial fibrillation (AF) is the most frequently diagnosed arrhythmia and affects 2.3 million people in the United States.3 Its prevalence increases with age, and as many as 9% of people older than 80 years are affected.4 Atrial fibrillation is characterized by a lack of coordinated atrial activity, and this loss of organized atrial contraction can lead to a myriad of clinical scenarios that include decompensated congestive heart failure (CHF), embolic cerebrovascular accident (CVA), ischemia, dizziness/weakness, and even asymptomatic patients with tachycardia.3 The pathogenesis of AF is now thought to involve an interaction between initiating triggers, often in the form of rapidly firing ectopic foci located inside one or more pulmonary veins, and an abnormal atrial tissue substrate capable of maintaining the arrhythmia.5 Although structural heart disease underlies many cases of AF, the pathogenesis of AF in apparently normal hearts is less well understood. Although there is considerable overlap, pulmonary vein triggers may play a dominant role in younger patients with relatively normal hearts and short paroxysms of AF, whereas an abnormal atrial tissue substrate may play a more important role in patients with structural heart disease and persistent or permanent AF.5
The known cardiovascular manifestations of HIV are: pericardial disease, myocardial disasese, infective endocarditis, cardiac tumors, vasculitis, coronary artery disease, hypertension, pulmonary hypertension and thrombosis and embolism.6 Amongst the aforementioned, hypertension is the most common cardiovascular manifestation. To date there has not been any data to suggest that HIV may invade the electric network of the heart.
HIV infects and gradually depletes CD4+ lymphocytes7 but may also affect other cell types, including monocytes/macrophages, endothelial cells, glial cells, intestinal epithelial cells, and possibly neurons. Studies have suggested that HIV may exhibit a cardiac tropism.8 HIV disease is an important cause of dilated cardiomyopathy,9 with a prevalence reported as 3.6% among cardiomyopathic patients. The prevalence is increasing as patients with HIV infection live longer.10 Patients with HIV-infection and dilated cardiomyopathy have a much worse prognosis than those with idiopathic dilated cardiomyopathy, hazard ratio of death 4.0.11,12 Recent studies on patients with idiopathic dilated cardiomyopathy found evidence of viral particles in endomyocardial biopsy specimens in up to two thirds of the patients.13 Among the viruses found that cause Myocarditis HIV has been a culprit. Myocarditis is the best studied cause of dilated cardiomyopathy in HIV disease.13 Review of the literature shows that myocarditis is arrythmogenic leading to atrial fibrillation (7-10%) and ventricular arrhythmia (39%).14 Review of all the literature did not yield any direct correlation between HIV and AF. In this study, we intended to evaluate whether HIV is a risk factor for developing AF. One possible explanation for this relationship may be through myocarditis. Although myocarditis is a well known cause of dilated cardiomyopathy, it is also a well known cause of AF. Furthermore, HIV is a well known cause of myocarditis, which once developed may cause AF. Unfortunately, we do not have any documented cases of myocarditis from these patients. However, this might be a plausible mechanism that could have preceded the onset of the atrial fibrillation. Many of our patients go to multiple hospitals within the same city and as such it is difficult to be certain of any recent admissions to other institutions for myocarditis.
HIV is a multisystemic disease, with many complications that arise as the CD4 count decreases and the immune system fails. The intent of this study was to show that there is a relationship between HIV and atrial fibrillation. This study shows that as the CD4 count decreases patients are at increased risk of developing Atrial fibrillation. One postulated mechanism is via myocarditis, which is relatively common in patients with a low CD4 count. Further studies are warranted to evaluate this subject.