Non-interventional Management of Symptomatic Pulmonary Vein Occlusion after Radiofrequency Ablation for Atrial Fibrillation
Logan Bittinger,MBBS, FRACPa, Anthony Tang,MDab
aDepartment of Cardiology, Royal Jubilee Hospital, Victoria, BC, Canada.
.bUniversity of British Columbia,BC, Canada
.
Pulmonary vein occlusion (PVO) after atrial fibrillation ablation is often highly symptomatic. In cases with a clear target, balloon angioplasty and stenting can be successful. In the absence of such a target, surgical lobectomy has been reported as a treatment option, but the natural history of physiological adaptation may outweigh the risks of invasive therapies and a non-invasive strategy is valid in these situations. We present a case of highly symptomatic PVO managed non-invasively, with complete symptom resolution and return to high-intensity exercise. Catheter intervention may not always be possible in the absence of a target vessel, and lobectomy may not be necessary to manage PVO.
Correspondence to: Dr Logan D Bittinger, Department of Cardiology, Royal Jubilee Hospital, 1952 Bay Street, Victoria, B.C., Canada V8R 1J8.
Pulmonary vein occlusion (PVO) after radiofrequency (RF) ablation for atrial fibrillation (AF) is rare and decreasing in frequency, but may be highly symptomatic. Management can be challenging and can include angioplasty or surgical lobectomy, but invasive strategies may not be necessary. We present a case of successful conservative management and discuss issues for management of PV occlusion.
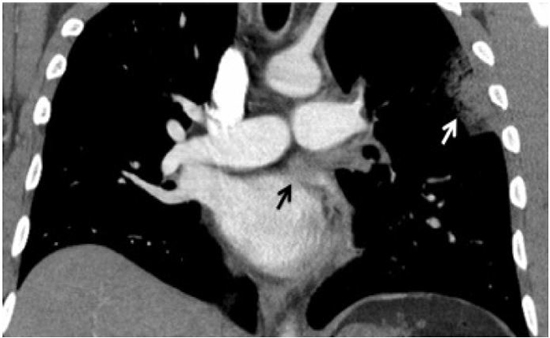
A 36-year-old man with drug refractory symptomatic paroxysmal atrial fibrillation (AF) presented for catheter ablation. His heart was structurally normal (chads2 score of 0). He underwent a pulmonary vein (PV) isolation procedure with wide antral lesions, using a 4mm irrigated tip radiofrequency catheter with power limited to 30W anteriorly and25W posteriorly. All PV were isolated. A recurrence of AF occurred at 6 weeks, and the patient returned for redo-ablation 3 months later. Computerised tomography (CT) revealed new stenosis in the left superior PV (LSPV) of 50% Image 1 with normal left inferior PV (LIPV). All veins had electrically reconnected and were re-isolated with wide antral lesions. The patient took warfarin for 6 months then changed to aspirin. Two weeks later, he reported severe dyspnea, cough, pleuritic pain and hemoptysis. Chest X-ray (CXR) showed left upper lobe consolidation with pleural effusion. CT angiography revealed occlusion of the LSPV, Image 2with venous congestion and edema/hemorrhage in the lingula.
Figure 1. CT scan of the chest with contrast: A significant stenosis in the left superior pulmonary vein is demonstrated (black arrow)
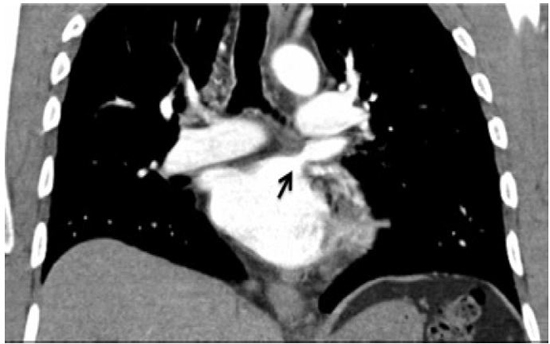
Figure 2. CT scan of the chest with contrast: Occlusion of left superior pulmonary vein (black arrow), with pulmonary infarct and alveolar hemorrhage (white arrow)
Ventilation/perfusion (V/Q) scan showed decreased perfusion in the left upper lobe, consistent with pulmonary infarction.
Imaging of the left atrium with angiography viatranseptal puncture could not demonstrate the ostium of the LSPV. Pulmonary wedge angiography revealed venous drainage only via the left inferior PV. A diagnosis was made of an ostial occlusion of the LSPV. No attempt at angioplasty was made due to concerns of the lack of a venous stump to target. Management options were presented to the patient and he expressed a desire for an initial trial of conservative management. Treatment included oxygen, analgesia, antibiotics, and mobilization, with no anticoagulation due to hemoptysis. The majority of symptoms resolved within a week, at which time the patient was discharged. A CXR, four months later revealed resolution of the pulmonary infarct with minimal scarring. Pulmonary function testing revealed normal lung function, including spirometry, lung volumes, diffusing capacity and flow-volume. Clinical review at 18 months found no residual symptoms, such as dyspnea, cough, or hemoptysis, with normal exercise tolerance, and the patient had returned to high-level aerobic exercise training. No further AF has been documented.
Pulmonary vein stenosis is an infrequent complication of AF ablation, with an estimated prevalence of 0.4% .1 Stenosis rates have reduced with adoption of wide antral circumferential ablation.2 PV occlusion is rarer, with rates decreasing from 1.4% in 2000 to 0.1% in 2004 with a change from ostial segmental ablation to antral ablation.3-5 Risk of stenosis correlates with ablation within PVs, RF power levels, operator experience, venous diameter and pre-existing stenosis.6 Previously stenosed veins carry higher risk of occlusion, and this progression may be asymptomatic.7 Up to 34% of occlusions occur after two or more ablations, with left veins occluding most frequently (LSPV 54%, LIPV 29%) as ablation within the vein is often required to avoid ablating within the atrial appendage.5
Thrombotic PV occlusion may correspond to warfarin cessation, the anticoagulation having maintained patency of a severely stenosed vein, but progression may occur despite anticoagulation.5,7 Some centers routinely screen for stenosis after RF ablation, though cost, availability of screening modalities, radiation exposure for CT and refusal by asymptomatic patients are limiting factors. CT frequently over-diagnoses PVO and occlusion should be confirmed by selective pulmonary artery wedge angiography, which may reveal a target for angioplasty.8,9
Severity of symptoms has been shown to correlate with percentage stenosis of total venous drainage of a lung, although PVO can be asymptomatic.5,10,11 Factors corresponding to symptoms include size of vein occluded, area of lung drained, presence of an ipsilateral PV stenosis, and the degree of collateral drainage. Severe symptoms, particularly dyspnea and pulmonary congestion, are indications for intervention as earlier intervention may result in better long-term segmental perfusion. If an ipsilateral vein is also stenosed, dilation of that stenosis relieves hemodynamic burden on the pulmonary vasculature and can improve symptoms.
In symptomatic cases of PVO (or subtotally occluded veins) with an identifiable target (eg venous stump), angioplasty ± stenting has been shown to reduce stenosis, improve pulmonary perfusion and resolve symptoms, particularly if performed early post occlusion.5,9 Intervention to adequately define anatomy should be offered to all patients with presumed occlusion on CT with subsequent dilation or stenting if possible. Angioplasty is frequently though not always successful and restenosis may occur despite stenting.
In cases without a target for angioplasty, management decisions are more challenging and include conservative therapy, surgical lobectomy or angioplasty of ipsilateral stenoses if present. Steliga et al. recently reported use of pulmonary lobectomy for symptoms secondary to lobar thrombosis after a PV occlusion. Nuclear perfusion scanning showed minimal blood flow to the affected lobe and a subsequent lobectomy resulted in resolution of symptoms of pain and cough.12 While lobectomy may potentially reduce symptoms and complications related to the infarcted lung, the risks of surgical morbidity are significant. In patients without the option of catheter intervention, conservative management may result in symptomatic improvement and obviate the need for lobectomy. A physiologically based argument can be made for conservative management:after occlusion, altered pulmonary hemodynamics result in redistribution of blood flow, opening of pulmonary vascular channels, maximization of collaterals, and neovascularisation.13,14 Di Biase et al. reported no or only mild residual symptoms in all of 18 patients after 14 months, regardless of PVO recannalisation, suggesting a natural history of symptom resolution with physiological adaptation, at least in mid-term follow-up. Longer follow-up out to ≥4 years suggests that patients with severe stenosis or total occlusion of a single vein without intervention may eventually develop symptoms, warranting continued follow-up in patients managed conservatively.15 Currently, data does not exist of follow-up of sufficient numbers of patients with conservatively managed PVO to draw definite conclusions of the natural history of these patients. The principles of conservative management are oxygenation, analgesia, managing hemoptysis, and antibiotic treatment to prevent infection, to allow time for collateralisation and alterations in pulmonary vasculature. This strategy was highly successful for our patient.
Management of PV occlusion can be challenging and controversial and an observation that symptomatic resolution may occur could support a conservative strategy. Every effort needs be made to confirm PVO, including pulmonary wedge angiography, to determine if a target for angioplasty is possible as this therapy can improve symptoms and perfusion. If a PVO lacks a target for angioplasty and an ipsilateral vein is stenosed, that vein should receive angioplasty, leaving the occlusion untouched. If there are no ipsilateral stenoses, the patient should be managed non-invasively with supportive therapies. Patients with known stenoses undergoing redo-ablation should have more intensive and routine re-imaging of PVs with consideration of intervention on a stenosed PV before ceasing warfarin.