Left Atrial Volume and Post-Operative Atrial
Fibrillation after Aortic Valve Replacement
Yeruva Madhu Reddy, MD, Ruby Satpathy, MD, Xuedong Shen, MD, PhD, Mark Holmberg, MD, PhD, Claire Hunter, MD, Aryan Mooss, MD, Dennis Esterbrooks, MD
The Cardiac Center of Creighton University, Creighton University, Omaha, Nebraska, USA.
Post-operative atrial fibrillation (POAF) after valve surgery is associated with increased morbidity and mortality. Risk factors identified in the past to predict POAF are of moderate accuracy. We performed a retrospective analysis of 139 patients undergoing aortic valve replacement for aortic stenosis. Post-operative AF occurred in 44% of the patients. In multivariate analysis only left atrial volume (LAV) index was a predictor of POAF. A LAV index of >46 cc/m2 predicted POAF with a sensitivity and specificity of 92% and 77%. We propose that LAV index can be used preoperatively to identify patients at risk for POAF to target preventive interventions.
Post-operative atrial fibrillation (POAF) is common after valve surgery and is associated with increased morbidity and mortality. Many of the previously identified predictors of POAF are of moderate accuracy. Left atrial volume (LAV) index has been proposed in the past as a predictor of POAF in patients undergoing cardiac surgery. In patients with aortic stenosis (AS), increased LAV is a marker of severity of stenosis.
Left atrial volume index is a very good predictor of POAF in patients undergoing aortic valve replacement (AVR) for AS.
We performed a retrospective analysis of 139 consecutive patients with no previous atrial fibrillation (AF) undergoing AVR for AS in our center.
Post-operative AF occurred in 44% of patients. Patients with POAF had a longer hospital stay compared to patients without (12 vs 8 days; p < 0.001). In univariate analysis, age (p = 0.046), aortic valve area (p = 0.005) and LAV index (p < 0.001) were significant predictors of POAF. In multivariate analysis only LAV index (R2= 0.58; p < 0.001) predicted POAF. A LAV index > 46ml/m2 predicted POAF with a sensitivity and specificity of 92% and 77% respectively. Moreover, there was a significant increase in the incidence of POAF with increasing quartiles of LAV index, supporting causality.
Left atrial volume index is an excellent predictor of POAF in patients undergoing AVR for AS. It can be used for selecting patients who are at a high risk for developing POAF to target preventive interventions.
Correspondence to: Yeruva Madhu Reddy, MD, The Cardiac Center of Creighton University, 3006 Webster Street, Omaha, Nebraska 68131, USA.
Post-operative atrial fibrillation (POAF) is one of the most common arrythmias after cardiac surgery with an increased mortality, stroke and prolonged hospital stay.1-6 Patients undergoing combined valve and coronary artery bypass graft surgery with continuous rhythm monitoring have been reported to have the highest incidence of POAF (36-63%).1,2,5,6 Patients with symptomatic severe aortic stenosis (AS) have a poor prognosis with conservative treatment and aortic valve replacement (AVR) in these patients is also associated with higher incidence of post-operative complications including POAF.
There have been many studies in the literature identifying at least 17 pre-operative risk factors for POAF in various patient populations undergoing cardiac surgery.6 Most of these risk factors have only a moderate accuracy in predicting POAF independently and consistently.6 Scoring systems incorporating multiple risk factors have been developed but they may be cumbersome at times decreasing their applicability.4-7 Moreover, almost all of these studies included patients undergoing any type of cardiac surgery and extending their results to the higher risk group undergoing AVR may not be accurate.
Left atrial volume (LAV) index (LAV indexed to body surface area) is a validated marker of chronic left ventricular load and is closely related to the development of atrial fibrillation (AF) in patients with and without cardiovascular disease.8,9,10 In patients undergoing cardiac surgery LAV index has been shown to be strongest predictor of POAF.11 Though the study included patients undergoing AVR, their number was not reported. Increased LAV has been correlated with the severity of AS and it has been proposed to have a role in the development of AF.12,13,14 There are no studies reported to date that have evaluated the role of LAV index in predicting POAF in patients undergoing AVR for AS. Hence, we hypothesized that LAV index is an independent and a strong predictor of POAF in patients with severe AS undergoing AVR.
We performed a retrospective analysis of all consecutive patients who underwent AVR for severe AS in our hospital between January 2003 and December 2005. Patients with more than moderate mitral valve stenosis or regurgitation, history of atrial fibrillation or a previous valve replacement were excluded from the study. A total of 232 patients were underwent surgery during the above time period of which 93 patients were excluded based on the above criteria. The remaining 139 patients were included in the final analysis. The study was approved by our institutional review board.
The baseline characteristics including demographics, pertinent clinical history, medications, echocardiographic parameters were obtained. The LAV was calculated by the bi-plane area method as described by the 2005 American society of echocardiography guidelines for chamber quantification.15 In this method, the maximal left atrial (LA) area was planimetered just before the opening of the mitral valve in both two and four-chambered views after excluding the pulmonary veins and the LA appendage. Length of LA was measured by a line extending perpendicularly from the middle of the mitral valve annulus plane to the superior wall of LA in both views and the shorter of the lengths was used to calculate LAV. The LAV was calculated using the formula LAV = 0.85 x four-chambered area x two-chambered area ÷ LA length. The LAV index was calculated from the ratio of the LAV and body mass index.
Details of the type of surgery, perioperative cardiac medications and complications in the hospital were obtained from the hospital records. In our institution, all postoperative patients undergo continuous rhythm monitoring from surgery till discharge, which is confirmed by a board certified cardiologist everyday. We collected the occurrence and duration of AF episodes along with the length of hospital stay.
Data are presented as proportions (%) for categorical variables and as mean ± SD for continuous variables. For skewed variables, the median is reported. The association between patient variables (from previous medical history, medications and echocardiography parameters) and development of POAF was initially evaluated by univariate analyses. Comparisons were made between continuous variables by an ANOVA test and between categorical variables by a chi-square test. Fisher’s exact test was used wherever applicable. A Mann-Whitney U test was used for skewed variables. All variables from the univariate analyses with a p-value <0.2 were included in a multivariate regression analysis (backward selection method). The best cut-off of LAV index for predicting POAF was determined using a receiver-operating characteristics curve analysis (ROC curve). A hazards risk model was used to estimate the risk of developing POAF in the groups created by the above cut-off value of LAV index. Further, the group was divided into quartiles based on the LAV index and Cox regression survival analysis was performed to identify the POAF-free survival in each quartile. The statistical analysis was performed using SPSS software package, version 16.0 (SPSS inc., Chicago, Ill). A p-value of <0.05 was considered statistically significant.
A total of 232 patients underwent AVR for severe AS from January 2003 to December 2005 in our center. We excluded 93 patients from the analysis based on the exclusion criteria mentioned earlier. A total of 139 patients were included in the final analysis. Concomitant coronary artery bypass graft surgery was performed in 58 (42%) patients. The mean age of our study population was 72 ± 12.1 yrs and 55% (77/139) of them were men. Of the 139 patients, 61 patients (44%) developed POAF. The median time to onset for POAF was 3 days. Among patients who developed POAF, 69% (42/61) developed paroxysmal and the rest 31% (19/61) developed persistent AF. Among patients with paroxysmal AF, 31% (13/42) developed a single episode and 69% (29/42) developed multiple episodes of AF. Post-operative AF was associated with a significantly increased median length of hospital stay (12 days vs 8 days; p < 0.001). It was not associated with an increased stroke or death rate (8.2% vs 5.1%; p=0.35).
Table 1. Baseline clinical characteristics and medication use
|
POAF |
No POAF |
p-value |
| Male Sex (%) |
60.7 |
51.3 |
0.27 |
| Mean Age |
74.3 |
70.2 |
0.046 |
| Hypertension (%) |
73.8 |
75.6 |
0.80 |
| Diabetes Mellitus (%) |
26.2 |
34.6 |
0.29 |
| Smoking (%) |
24.6 |
25.6 |
0.89 |
| Hyperlipidemia (%) |
50.8 |
59.0 |
0.34 |
| Coronary Heart Disease (%) |
50.8 |
55.1 |
0.61 |
| Heart Failure (%) |
65.6 |
64.1 |
0.86 |
| Chronic obstructive lung disease (%) |
14.8 |
6.4 |
0.16 |
| Hypothyroidism (%) |
14.8 |
11.5 |
0.58 |
| Chronic Renal Insufficiency (%) |
11.5 |
12.8 |
0.81 |
| Beta Blockers (%) |
45.9 |
52.6 |
0.44 |
| Diuretics (%) |
49.2 |
42.3 |
0.42 |
| Amiodarone (%) |
4.9 |
1.3 |
0.32 |
| Statins (%) |
34.4 |
43.6 |
0.27 |
| ACEI/ ARB (%) |
56.6 |
43.4 |
0.16 |
| Thyroxine (%) |
18.0 |
12.8 |
0.39 |
POAF: Post-Operative Atrial Fibrillation; ACEI: Angiotensin Converting Enzyme inhibitors; ARB Angiotensin Receptor Blockers
Demographic and medication data is presented in table 1. Patients who developed POAF were older than those who did not (74 vs 70 years; p = 0.046). There were no significant differences between the medications used by both the groups. Baseline echocardiographic parameters, median length of hospital stay and concomitant coronary bypass surgery data are presented in table 2. Patients who developed POAF had a significantly smaller aortic valve area (0.75 vs 0.86 cm2; p = 0.005) and aortic valve area index (0.39 vs 0.45, p = 0.025) when compared to patients who did not develop POAF. Patients with POAF also had a significantly higher LAV (124 vs 74; p < 0.001) and LAV index (64 vs 39; p < 0.001) when compared to patients who did not develop POAF. Moreover, there was a significant negative correlation between aortic valve area and both LAV (r = -0.25; p = 0.003) and LAV index (r = -0.17; p = 0.04). There was a significant correlation between LA diameter and LAV (r = 0.18 p = 0.036) but not between LA diameter and LAV index (r = 0.06; p = 0.48).
Table 2. Baseline echocardiographic findings, hospital stay and concomitant coronary bypass surgery
|
POAF |
No POAF |
p-value |
| Left Ventricular Ejection Fraction |
51.9 |
55.8 |
0.07 |
| Aortic Valve Area in cm2 |
0.75 |
0.86 |
0.005 |
| Aortic Valve Area Index |
0.39 |
0.45 |
0.025 |
| Mean Aortic Valve Gradient in mmHg |
42 |
39 |
0.19 |
| Diastolic Dysfunction |
1.37 |
1.27 |
0.33 |
| Left Ventricular PW/SW dimension in cm |
1.35 |
1.34 |
0.80 |
| Left Atrial Diameter in cm |
4.53 |
4.34 |
0.07 |
| Left Ventricular Dimension in cm |
5.11 |
4.99 |
0.36 |
| Right Ventricular Systolic Pressure in mm Hg |
43 |
36 |
0.05 |
| Left Atrial Volume in cm3 |
124 |
74 |
<0.001 |
| Left Atrial Volume Index |
64 |
39 |
<0.001 |
| Hospital Stay (Median) in days |
12 |
8 |
<0.001 |
| Concomitant Coronay Artery Bypass (%) |
41 |
42 |
0.87 |
POAF: Post-Operative Atrial Fibrillation
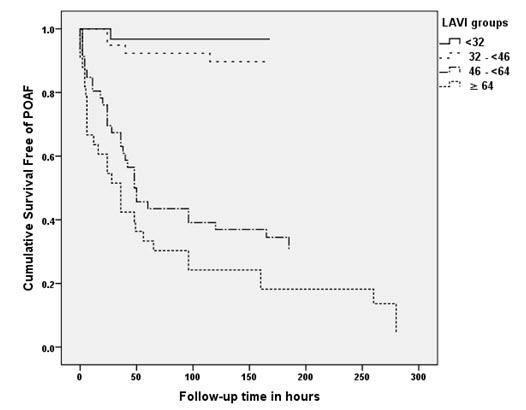
In univariate analysis age (p=0.046), aortic valve area (p=0.005) and LAV index (p<0.001) were significant predictors of POAF. After multivariate regression analysis only LAV index correlated significantly with the development of POAF with a R2 value of 0.54 (p<0.001). The receiver-operator characteristics curve analysis showed an optimum cutoff for LAV index as 46, with a sensitivity of 92%, specificity of 77%, positive predictive value of 76% and a negative predictive value of 92% for predicting POAF Fig 1. The area under the curve was 0.92 (95% CI, 0.87-0.97). Compared to patients with a LAV index < 46ml/m2, those with a LAV index > 46ml/m2 had an almost 10 fold increase in the risk of developing POAF (HR = 9.8; 4.2-22.9) as shown in figure 2. When patients were divided into 4 quartiles based on the LAV index (≤32; 32> ≤46; 46> ≤64 and >64) there was a significant trend for increased incidence in POAF among the quartiles (3.3%; 11.4%; 65.1% and 90%; p<0.001 for trend) as shown in figure 3.
Figure 1. Receiver operating characteristic curve for Left atrial volume index. Area under the curve = 0.92 (0.87-0.97); Standard Error = 0.025; p-value < 0.001
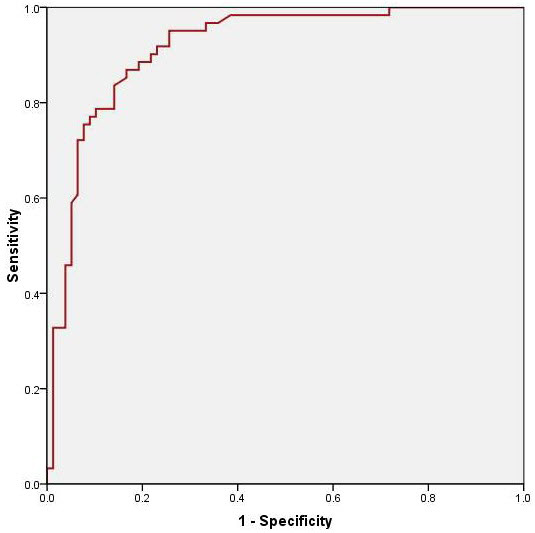
Figure 2. Incidence of POAF in 2 groups based on LAV index. (The cut-off of 46 was derived from the ROC curve in Figure 1. LAVI: Left Atrial Volume Index; POAF: Post-Operative Atrial Fibrillation
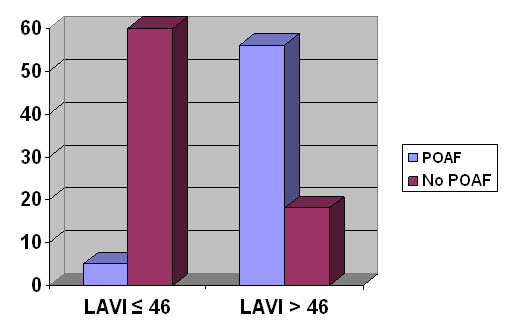
Figure 3. Cox-Regression survival analysis of the 4 quartiles based on the LAV index. Graded decrease in the survival free of POAF among quartiles of increasing LAV index
Post-operative AF is one of the most common complications after cardiac surgery and its incidence is high in patients undergoing valve surgery especially if combined with a concomitant coronary artery bypass surgery.1,2,5,6 It has been shown to be significantly associated with an increase in early and late mortality, stroke, myocardial infarction, persistent heart failure symptoms, respiratory failure, renal failure, shock, and cardiopulmonary arrest after cardiac surgery.2,5,6 Similar worse outcomes were reported with POAF in patients undergoing AVR.16-19 Post-operative AF is also associated with increased length of hospital stay resulting in increased hospital and healthcare costs.20,21 In our present study we did find a similar significant increase in the median length of hospital stay in patients who developed POAF compared with patients who did not (12 vs 8 days; p<0.001).
With the increased incidence of worse outcomes and increased healthcare costs with POAF, it is important to identify risk factors to predict its occurrence so that necessary proven preventive interventions can be implemented appropriately. Though many interventions have been proposed to prevent POAF, very few of them like beta blockers, bi-atrial pacing and intravenous amiodarone have consistently shown to decrease the incidence of POAF after cardiac surgery.6-22 Moreover, the use of intravenous amiodarone for unselected patient population undergoing cardiac surgery has not been shown to be cost-effective.23 Hence, there is an urgent need to identify patients undergoing cardiac surgery, especially AVR, who are at a higher risk of developing POAF, with a good accuracy.
There have been few studies in the past to identify risk factors to predict POAF in patients undergoing AVR. Orlowska-Baranowska et al. identified age, history of paroxysmal AF, presence of >300 paroxysmal supraventricular beats in 24 hours rhythm monitoring before surgery and presence of supraventricular tachycardia as independent predictors of POAF.24 Ducheschi et al. identified age, left atrial enlargement, history of paroxysmal AF and post operative electrolyte imbalance as independent predictors of POAF.25 Recently, Banach et al. identified low body mass index, impaired left ventricular ejection faction, increased left ventricular end-systolic thickness and end-diastolic diameter as independent preoperative predictors of POAF in patients undergoing AVR.19 Most of these risk factors had moderate accuracy in predicting POAF and some of them were postoperative variables making them difficult to serve the purpose of identifying patients preoperatively to implement preventive measures.
Many studies have reported LAV as a predictor of developing AF in a wide variety of patient populations, both with and without known cardiovascular disease.8,9,10 Aortic stenosis by itself and in combination with hypertension, increases the left ventricular afterload which in turn increases LA wall tension resulting in an increase in LA size. In addition, LAV is a better measure of LA size than LA diameter, especially when it is enlarged, owing to its asymmetric enlargement.26 Increased LAV has been shown to be positively correlated with the severity of AS.12 In our present study we did find a good negative correlation between aortic valve area and both LAV and LAV index confirming the above finding.
Left atrial enlargement, which represents atrial anatomic remodeling, might be associated with electrical remodeling. Atrial myocardial stretch activates extracellular signal-related kinase cascade, leading to atrial fibrosis, which has been proposed to increase the dispersion of the atrial effective refractory period leading to atrial arrhythmia including AF13,14 Osranek et al. reported that an increased LAV index independently predicted POAF after cardiac surgery.11 Since increased LAV is a marker of the severity of AS and is an excellent predictor of POAF, we planned to study its predictive power exclusively in patients undergoing AVR for severe AS.
In our present study, age, aortic valve area and LAV index were predictors of POAF in univariate analyses. In multivariate analysis, LAV index was the only independent predictor consistent with the pathophysiological basis for the development of POAF. Though LA diameter correlated with LAV, it did not predict POAF. This supports the proposition that LAV and LAV index are better measures of LA enlargement than LA diameter. Most importantly, the graded increase in the risk of developing POAF with increasing LAV index quartiles, in our study, supports causality as proposed earlier.11 Also, it is only the severe LA enlargement group ( LAV index of >40 as per the recent American Society of Echocardiography guideline statement)15 who has a significant increased risk of POAF.
In conclusion, in a single center, retrospective analysis of patients undergoing AVR for severe AS we identified a LAV index as the single most important predictor of POAF. A LAV index cut-off of 46 cc/m2 predicted POAF with a sensitivity of 92% and a specificity of 77%. In clinical practice a similar cut-off can be used in patients undergoing AVR to identify patients at high risk for POAF. Proven peri-operative interventions, aimed at decreasing POAF, if applied to this high-risk group may turnout to be very cost-effective as compared to applying to the entire group.
Important limitation of our study is its retrospective nature. The LAV index of our patient population is higher compared to the previous studies, which may be a part of the pathophysiology of severe AS, though an overall sicker population cannot be excluded. This may limit the extension of the findings to other populations. The size of the population studied is also small, which may be one of the reasons why we did not identify some of the previously identified predictors of POAF. Finally, this is a single center experience and it needs to be validated in other patient populations undergoing AVR for AS.