Computed Tomography Imaging in Atrial Fibrillation Ablation
Kavita Sharma, MD, Jeffrey A. Brinker MD, Charles A. Henrikson MD
Division of Cardiology, Department of Medicine, Johns Hopkins University School of Medicine, Baltimore, Maryland..
Atrial fibrillation (AF) is the most common supraventricular arrhythmia in adults and catheter ablation has increasingly become the therapy of choice for symptomatic, recurrent, drug refractory AF. The purpose of this review is to evaluate the utility of computed tomography (CT) imaging in guiding radiofrequency catheter ablation (RFCA), as well as highlight the additional radiographic and functional anatomic data provided by CT in comparison to alternative radiographic modalities.
Key Words : Atrial fibrillation, Wolff-Parkinson-White syndrome, Atrial muscle vulnerability, Accessory pathways, Abnormal atrial electrograms.
Correspondence to: Charles A. Henrikson, M.D, Johns Hopkins Medical Institutions, Division of Cardiology/Carnegie 592, Baltimore, MD 21205.
The most common sustained supraventricular arrhythmia in adults is atrial fibrillation (AF). The overall prevalence of AF is reported as 0.4%1 with prevalence increasing with age: 16-20% of those aged >85 years have AF.2 In most patients, AF is treated with the goal to reduce symptoms and decrease risk of stroke. Left atrial appendage (LAA) thrombi have been documented in approximately 10% of patients with AF in the absence of anticoagulation3,4 and lends to a risk of stroke several times higher than the general population.5
Catheter ablation has increasingly become the therapy of choice for symptomatic, recurrent, drug refractory atrial fibrillation (AF). Since the discovery of the pivotal role of the pulmonary veins (PV) in the genesis of atrial fibrillation by Haissaguerre et al. in 1998,6 electric isolation of the PVs has formed the cornerstone of current ablation techniques.7,8 This can be accomplished by surgical methods,9 or via catheter-based radio frequency ablation (RFCA).10 RFCA procedures typically involve the use of a variety of ablation catheters under fluoroscopic guidance to electrically isolate the PVs from the left atrium (LA), with or without the guidance of a three-dimensional (3D) electroanatomic mapping system.11,12
Despite advances in ablation techniques, RFCA continues to be a challenge in part due to the high
degree of variability in individual anatomy. It is known that anatomic imaging by fluoroscopy is limited by poor soft tissue resolution and exposure to ionizing radiation. Additionally, the catheter-based electroanatomic map often cannot replicate complex and highly variable PV and LA anatomy.13 The reported success rate of RFCA procedures at midterm follow up is 70-80% and major complication rate including PV stenosis, tamponade, and stroke is about 5%.14 The limited success rate and occurrence of major complications is in part due to the complexity and variability in PV and LA anatomy.
The purpose of this article is to provide a comprehensive review of the utility of computed tomography (CT) in image-guided RFCA, the additional radiographic and functional anatomic data provided by this imaging modality, and a comparison between CT and alternative radiologic modalities in RFCA.
CT for Left Atrium and Pulmonary Vein Mapping
Conventional pulmonary venous anatomy is defined as the presence of single right and left superior and inferior pulmonary veins that drain into the left atrium without any accessory veins (Figure 1). In a series of 201 patients who underwent evaluation of left atrium and pulmonary vein anatomy with thin-section (2.5mm) contrast enhanced CT scans, 71% of patients were found to have two ostia on the right side for upper and lower lobe veins, 28% had three to five ostia on the right side, due to one or two separate middle lobe vein ostia in 26% of patients. The majority of patients (86%) had two ostia on the left side for the upper and lower lobe veins; 14% were found to have a single ostium.15 In a smaller series of 34 patients, CT detected pulmonary venous variants in 6% of patients and identified other cardiac abnormalities, including suspicious pulmonary malignancy, mediastinal adenopathy, and coronary stenosis in 44% of patients.16
Figure 1A CT images of the left atrium. Axial image demonstrating the four pulmonary veins, appendage, and surrounding cardiac structures.
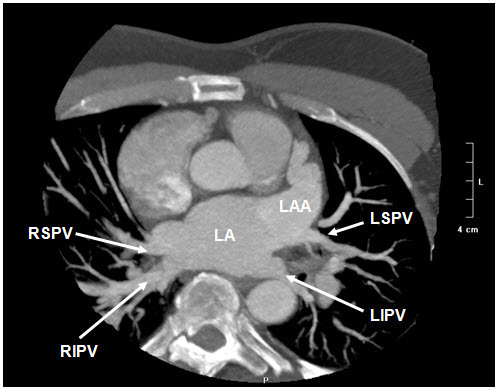
Figure 1B Coronal image demonstrating left atrial anatomy. LA=Left atrium, LAA=left atrial appendage, LSPV=left superior pulmonary vein, LIPV=left inferior pulmonary vein, RSPV=right superior pulmonary vein, RIPV=right inferior pulmonary vein.
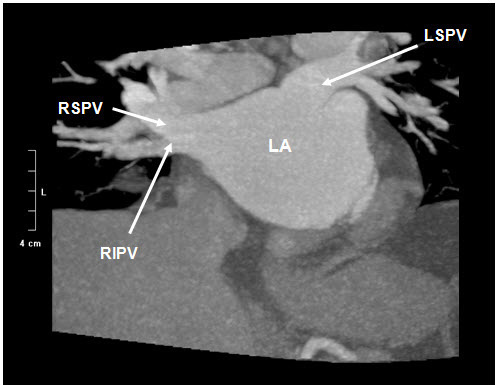
While there is considerable variation in the approach to RFCA for AF, at most centers the predominant approach to RFCA for AF focuses on PV isolation at the junction of their entry into the left atrium, or the ostium.17,18In addition to providing important anatomic landmark information, preoperative evaluation of the left atrium and the PVs has been shown to reduce fluoroscopic and procedural time.7,19,20,13 Preprocedural imaging for RFCA should include the following major features: (i) precise anatomy of the LA and PVs, (ii) precise measurement of each ostial diameter and the distance to the first branch, (iii) presence of accessory or supernumerary PVs, (iv) the LA dimension and the presence of LAA thrombus, and (v) major anomalies like common PV ostia, persistent left superior vena cava, anomalous pulmonary venous return, vein of Marshall, or hypoplasty or occlusion of PV.
CT imaging protocols vary by device manufacturer, model, detector number and institution-specific protocols with regards to radiation exposure. Multi-detector CT imaging at our institution is performed with the 320-detector row Aquilion One scanner (Toshiba Medical Systems, Japan). The cardiac CT protocol for all studies is a gated dynamic volume acquisition using slice thickness of 1.0mm, with 0.5mm reconstruction thickness. Image acquisition is gated to 40% of the R-R interval. For patients in atrial fibrillation, rate control is optimized to less than 80 bpm prior to the CT study. The tri-phase contrast protocol includes a total volume of 60mL (70mL if BMI > 29) of the noniodinated contrast material iopamidol (Isovue 370; Bracco Diagnostics, Princeton, NJ) administered at a rate of 4mL/sec in the following sequence: 20mL saline test injection, 40mL (100% contrast), 20mL (50% saline, 50% contrast), 65mL (30% saline, 70% contrast), followed by 30mL saline.
Figure 2A Computed tomography image of left atrium (LA) with left atrial appendage (LAA) with corresponding transesophageal echocardiogram images of LA and LAA. CT and TEE images demonstrating contrast filling defect compatible with a thrombus.
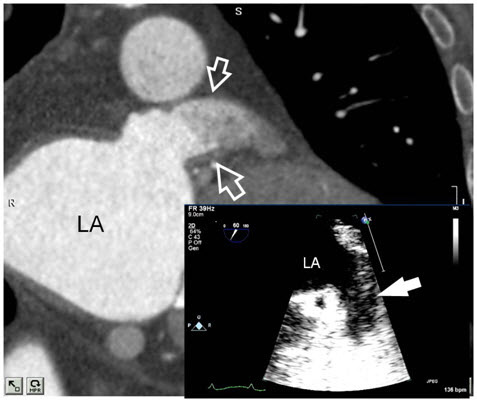
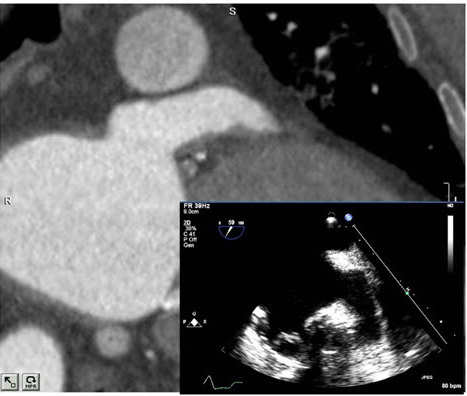
Figure 2B Right posterior oblique view highlighting the right veins.
CT versus Other Imaging Modalities for Pre-Procedural Assessment
There are a number of imaging modalities for pre- or intra-procedural assessment for AF ablation including fluoroscopy, transesophageal echocardiography (TEE),21 intracardiac echocardiography (ICE),22 magnetic resonance imaging (MRI),23 and CT angiography .24 Several studies have demonstrated the utility of cardiac multi-detector CT angiography to show the anatomy of the LA and PVs prior to RFCA. 24,25,26 In comparison to other imaging modalities, CT has been shown to be superior to fluoroscopy, TEE, and ICE to depict the numbers of PV ostia. 25,27,28 In addition, CT angiography showed equivalent diagnostic value to ICE in depicting supernumary PVs, without the user-dependent challenges with ICE.25 CT can also obtain detailed volumetric data with 0.5mm spatial resolution, allowing for better depiction of LA and PV structure compared to ICE and TEE.15 Perhaps most importantly, CT allows for images to be imported for procedure guidance into the real-time nagivation system.
CT for Left Atrial Thrombus Detection
The current practice for detection of LA and LAA thrombus usually consists of performing a TEE prior to RFCA. With the increasing use of CT prior to RFCA for anatomic guidance of RFCA, the efficacy of CT for detection of LA thrombus has been studied, although data is limited. LA thrombus is identified by CT as an intracavitary contrast filling defect, with attenuation characteristics similar to nonenhanced tissues and differing from surrounding normal pectinate muscle and fat by the reader (Figure 3).29,30 Anecdotally, CT can provide excellent imaging of LA thrombus, but its systematic use has not been consistently validated.
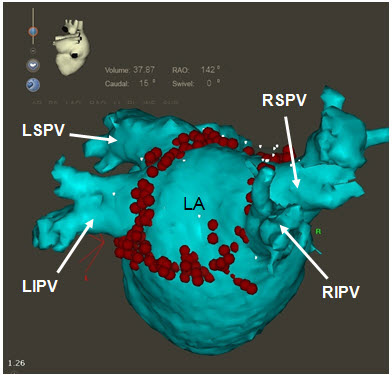
Figure 3A 3-D reconstructed image of the left atrium using the CartoMerge system. Ablation lesions are denoted by solid red dots. A. Left posterior oblique view highlighting the left pulmonary veins.
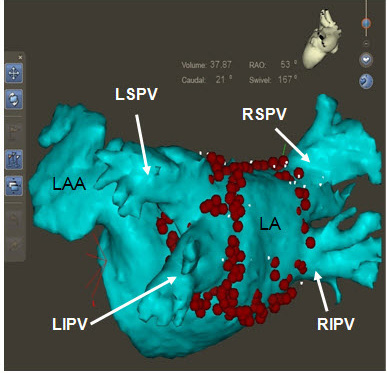
Figure 3B Right posterior oblique view highlighting the right veins.
Our group sought to determine the accuracy and interobserver variability of CT in the evaluation of LA thrombus prior to RFCA in 50 patients who underwent 64-slice multidetector CT scan and TEE prior to ablation 29 (Figure 2). We demonstrated that interobserver variability between the three CT readers was poor (highest kappa statistic 0.43, p=0.0001). Diagnostic accuracy was highly variable, with sensitivities ranging from 100% to 50% and specificities ranging from 85% to 44%. TEE reader agreement, in comparison, was 98%. Based on these findings, even amongst experienced observers, CT cannot reliably diagnose LA thrombus, as the interobserver variability is substantial compared to TEE. Potential factors affecting the accuracy of CT include image quality and the difficulty differentiating clot from pectinate muscle.
Jaber, et al. evaluated CT’s ability to detect LA thrombus, finding perfect CT accuracy (sensitivity and specificity of 100%) in detecting LA thrombus, however used an older generation CT scanner with 4-detector row and 500-ms gantry time.30 While the reason for this discrepancy is not entirely clear, this latter study did not provide interobserver variability, heart rate at the time of study acquisition, or x-ray tube current during scanning. Another study comparing 64-slice CT to TEE for detection of LA thrombus in 51 patients reported similar efficacy.31 Limitations of this study, however, included small population size, and lack of data on interobserver variability.
These studies suggest that the use of CT for the detection of LA thrombus is limited by high interobserver variability, modest diagnostic accuracy, and radiation exposure. At this time it does not appear that CT is an adequate substitute for the gold standard of TEE in the detection of LA thrombus prior to RFCA. Additional research directed at different image acquisition techniques, lowering heart rate, and the evaluation of newly evolving technological developments in CT should be pursued.
CT Image Integration-Guided Ablation versus Conventional Methods
Early case series evaluating image integration-guided RFCA (CartoMerge, Biosense Webster, Inc., Diamond Bar, CA, USA) found this method to be feasible, accurate, reproducible, and independent of breathing artifact.32,33,13,34 Initial experiences in our group with this method suggested that (i) CT and MR images can be successfully used for the anatomically correct extraction and reconstruction of the LA and PV anatomies; (ii) 3D MR/CT reconstructions can be accurately registered with a real-time mapping system using a combination of two registration strategies; (iii) 3D image integration allows for tailored RFCA to individual PV and LA anatomy during AF ablation (Figure 3).13
While limited, randomized control studies to evaluate the efficacy of image integration-guided RFCA suggest this method improves the success of RFCA. Martinek et al. evaluated clinical outcome data in 100 patients undergoing RFCA by either conventional method or image integration-guided method at 6 months . Overall success was reported to be 67.9% in the conventional group compared to 85.1% in the image-integrated RFCA group (p=0.018). Additionally, the group reported over 50% risk of PV stenosis in the conventional method group compared to 5.7% risk in the image integration-guided group. In a prospective, randomized trial of 290 patients, the atrial fibrillation-free survival rate was significantly higher in the image integration-guided RFCA group compared to the conventional method group (88% versus 69%, p=0.017) at a mean follow-up of 14 ± 12 months. While the majority of randomized control data evaluating image integration-guided RFCA demonstrates increased efficacy compared to conventional methods, Kistler et al. found that image integration-guided RFCA did not significantly improve clinical outcomes (acute outcomes or long-term outcomes at 6 months) in 80 patients who were randomized to conventional methods versus image integration-guided RFCA.
The majority of randomized controlled trials evaluating the efficacy of image integration-guided RFCA have been performed using CartoMerge technology (CartoMerge, Biosense Webster, Inc., Diamond Bar, CA, USA). Initial case series reports using alternative technologies, however, have also suggested feasibility and success for image integration-guided RFCA. Piorkowski et al. reported feasibility and successful placement of complex linear ablation line concepts guided by an integrated 3D image using the ‘Digital Image Fusion’ (DIF) algorithm provided by NavX-EnSite (Endocardial Solutions, Inc., St Paul, MN, USA) .
CT Imaging Prior to Balloon-based PVI
The limitations of RFCA including point-by-point creation of ablation lesions, complex navigation, and small but definite risk of complications (PV stenosis, stroke, atrial-esophageal fistula) has led great interest in developing balloon catheter-based ablation technology. Balloon catheters using various ablative energy sources including cryothermal energy (CRYO; CryoCath Technologies, Inc, Canada) and “hot” balloon ablation catheters which are elastic balloon ablation catheters composed of an antithrombotic, heat-resistant resin (Toray Industries, Inc., Houston, Texas), have and continue to be developed to allow for single or few energy applications to achieve PVI, rather than long, contiguous circumferential ablation lines formed in RFCA .
With the use of these techniques, CT is likely to be beneficial in providing individualized imaging of the LA chamber both for size and PV anatomy, as well as to help facilitate selection of the appropriate balloon catheter size and whether more than one balloon size may be required. As further technologies for pulmonary vein isolation are developed in the future, these are likely to be anatomically based, and imaging of the pulmonary veins and left atrium for procedure planning will continue to be a crucial part of the these procedures.
CT Evaluation of Post-Ablation Complications
There are a number of recognized major complications of RFCA, including stroke, tamponade, vascular injury, atrio-esophageal fistula, pulmonary vein stenosis/occlusion, hemothorax, heart block, acute lung injury, and mitral valve injury. The complication rates reported vary considerably; our group found an overall complication rate of 5% in a retrospective study of 641 patients post RFCA. 42 With regards to the cardiac and vascular complications, CT is a relatively low-risk, fast, and readily available imaging modality to assess for the majority of such complications.
A significant risk associated with balloon catheter-based technology is phrenic nerve palsy, with reported complication rates of 7.1% with the high-intensity focused ultrasound (HIFU) technique and 7.5% with the cyroballoon technique. In one study, CT imaging of the right pericardiophrenic artery could reliably locate the right phrenic nerve and, if located within 10mm of the right superior pulmonary vein, poses a higher risk of phrenic nerve injury using balloon ablation devices. In this regard, pre-procedural CT may provide critical information regarding phrenic nerve anatomy and location, thereby identifying patients at higher risk for phrenic nerve injury, although this is not yet routinely examined.
Pulmonary vein (PV) stenosis is a well known complication of PVI for atrial fibrillation management and while the majority of patients who develop PV stenosis are asymptomatic, it can be associated with significant morbidity. In a series of 608 patients undergoing PVI, Robbins et al reported the incidence of pulmonary vein stenosis following catheter ablation.46 All patients in the series underwent spiral CT at 1, 3, 6 and 12 months post-procedure. The study demonstrated that severe (≥70%) narrowing was detected in 3.4%, moderate (50% to 69%) narrowing in 4.4% and mild (<50%) narrowing in 7.7% of patients. From a total of 15 pulmonary veins considered to be totally occluded by sprial CT, 7 were confirmed by pulmonary vein angiography. Packer et al reported on the clinical presentation, diagnosis, and management of PV stenosis in 23 patients post PVI who underwent CT pre- and post-PVI to assess for PV anatomy and subsequent stenoses.18 The study found that CT was helpful in identifying the location and extent of stenoses, with limited yet significant correlation between the extent of stenoses seen on CT images and that seen at the time of angiography.
CT provides the additional advantage that it is operator-independent unlike other imaging modalities such as TEE and ICE. By delineating complex LA and PV anatomy, CT enables the RFCA operator to take a more individualized approach to RFCA for each patient. This preprocedural knowledge may allow for less use of radiation and overall decrease in procedure time during the RFCA. In addition, CT is safe for patients with implanted devices, a known limitation in MRI.
Regarding the limitations of CT, of primary concern is the radiation exposure associated with CT compared to other imaging modalities. Increasing data from large patient studies indicate that cumulative effective doses of radiation increase with advancing age, with CT imaging accounting for up to 38% of the total effective radiation dose . While the individual radiation dose from CT is far less than the radiation exposure incurred during a RFCA procedure itself, these findings have prompted a more individualized approach and critical evaluation of the necessity of CT imaging with respect to RFCA. Given the concerns for cumulative radiation exposure over a lifetime, one might consider MRI as the imaging study of choice prior to RFCA, especially in those with a known history of substantial lifetime radiation exposure and otherwise uncomplicated anatomy. Additionally, patients who are relatively young being considered for RFCA may be appropriate candidates for MRI rather than CT prior to RFCA to help minimize their lifetime cumulative radiation exposure. Children and young adults are inherently more radiosensitive and have more remaining years of life during which a radiation-induced cancer may develop.
After evaluation of the benefits and limitations, if CT is still felt to be the most appropriate imaging study for a particular patient, several studies have reported effective methods to reduce the radiation dose including: (i) lowering kilovoltage setting, (ii) automatic ECG-pulsed tube current modulation, (iii) dose modulation with mid-diastole, (iv) prospective gating, (v) non-ECG gating, and (vi) special filtering . Most importantly, there is always going to be a balance between radiation dose and image quality. RFCA operators should strive to reduce the radiation dose of the pre-procedure CT as much as possible while still obtaining an image quality adequate to help guide the procedure.
Computed tomography has many well-established uses in cardiovascular medicine. Review of both case series reports and randomized control studies demonstrates that CT is a suitable, if not preferred, modality for preprocedural assessment of LA and PV structure, size, and volume prior to RFCA, in addition to its use in the procedure to help guide ablation. CT has also been shown to be potentially beneficial prior to balloon catheter-based procedures, particularly by identifying patients at high risk for phrenic nerve injury complication. Regarding identification of LA thrombus, at this time CT does not appear sufficient to replace TEE prior to RFCA. Overall, as with any imaging technology, the benefits of CT must be weighed against limitations, particularly radiation exposure to the patient.