Endurance Sport Practice and Atrial Fibrillation
Naiara Calvo MD, Lluis Mont MD, PhD
Thorax Institute, Hospital Clínic, University of Barcelona, Institut d’Investigacio Biomèdica August Pi i Sunyer (IDIBAPS).
Correspondence to: Dr. Lluis Mont, Thorax Institute (ICT) – Cardiology Department, Hospital Clinic, University of Barcelona, Villarroel 170, Barcelona 08036, Catalonia - Spain.
Atrial fibrillation (AF) is the most common cardiac rhythm disorder in clinical practice, with an estimated prevalence of 0.4% to 1% in the general population,1 increasing with age to 8% in those older than 80 years.2 The recognized risk factors for developing AF include age, hypertension, structural heart disease, diabetes mellitus, and hyperthyroidism.3 However, the etiology remains unclear in a significant number of patients younger than age 60 in whom no cardiovascular disease or any other known causal factor is present. This condition is termed lone AF4 and may be responsible for as many as 30% of patients with paroxysmal AF seeking medical attention.5,6
Although regular physical activity clearly reduces cardiovascular morbidity risk,7,8 in recent years long-term endurance sport practice has been recognized as a risk factor for AF9-16table 1. However, the underlying mechanism explaining this association is unclear.
Table 1. Studies demonstrating an increased risk in AF in individuals practicing endurance sports. Reproduced with permission of Mont et al.19
| Studies |
Type of study |
% Men |
Age |
Type of sports |
Cases/controls |
| Kaarjalainen et al |
Longitudinal case/control |
100% |
47±5 runners49±5 controls |
Orienteering |
262/373 |
| Mont et al |
Retrospective/compared to general
population |
100% |
44 ±13 sports
49 ±11 non-sports |
Endurance sports >3h/week |
70 lone AF |
| Elosua et al |
Retrospective case/control |
100% |
41±13 AF patients
44±11 controls |
Endurance sports Current practice and >1500 cumulated hours of practice |
51/109 |
| Heidbuchel et at |
Case/control in patients undergoing flutter ablation |
83% |
53±9 sports
60±10 controls |
Cycling, running or swimming >3hours/
week |
31/106 |
| Molina et al |
Longitudinal case/control |
100% |
39±9 runners
50±13 sedentary |
Marathon running |
252/305 |
| Baldesberger et al |
Longitudinal case/control |
100% |
67±7 cyclist
66±6 golfers |
Cycling |
134/62 |
| Mont et al GIRAFA study |
Prospective case/control |
69% |
48±11 |
Endurance sports |
107/107 |
The aim of this review is to analyze the evidence of the association between AF and endurance sport practice, the pathophysiological mechanisms responsible for this association, the clinical characteristics of this arrhythmia, and the available therapeutic options.
Atrial Fibrillation and Endurance Sport
In 1998, Karjalainen et al.9 evaluated the presence of AF in 228 veteran male orienteers (cross-country runners) and compared this with AF prevalence in a matched control group. Lone AF was diagnosed in 12 orienteers (5.3%) vs. 2 control subjects (0.9%). Our group has analyzed the proportion of patients with lone AF who engage in frequent and long-term sports activity.10 In a cohort of 1160 consecutive patients seen at the outpatient arrhythmia clinic, 70 individuals had lone AF and all were <65 years of age. Of these 70 patients, 32 reported engaging in endurance sport practice, defined as at least 3 hours a week. Surprisingly, 63% of all men with lone AF in this study actively participated in sports, significantly higher than the 15% of males in the general population. A case-control study of the same population of LAF patients and two age-matched controls from the general population for each case found the risk of developing LAF was more than five times greater [OR 5.06(1.35–19)] in those currently practicing an endurance sport.12 This association was observed at more than 1500 lifetime hours of sport practice, suggesting the existence of a threshold point. A further study of lone AF incidence among marathon runners, compared to sedentary men, demonstrated a higher incidence of AF in runners (annual incidence: 0.43/100 for runners, 0.11/100 for sedentary men).15
Heidbuchel et al.16 assessed the influence of sports activity on the risk of AF after the ablation of atrial flutter. Of 137 patients undergoing ablation of the right atria isthmus because of atrial flutter, 31 (23%) regularly participated in endurance sports. A history of competitive sports practice was associated with a higher post-ablation risk of developing AF [multivariate HR 1.81 (1.10–2.98, P 0.02)]; ongoing practice of an endurance sport (19 patients) after the ablation also increased the risk of AF [multivariate HR 1.68 (0.92–3.06), P 0.08].
Baldesberger et al.13 recruited 62 Swiss cyclists who had participated at least once in the Tour de Suisse professional bicycle race in 1955–1975 and compared the incidence of arrhythmias with a control group of 62 male golfers. The controls, matched for age, weight, hypertension, and cardiac medication, had never engaged in high endurance training. Paroxysmal or persistent AF or atrial flutter was reported more often in the cyclists (P 0.028).
In contrast with these previous studies, Pellicia et al.17 reported that LAF was uncommon among competitive athletes and the incidence was similar to that observed in the general population. However, the study was performed in young athletes at the height of their activity, while studies supporting the association have been performed in middle-aged individuals, after many years of sport practice. Additionally, the largest and most recent epidemiological study.18 supports that this association is not a matter of selection: after adjustment for multiple potentially confounding lifestyle factors and health conditions, vigorous exercise activity was associated with an increased risk of developing AF.
Pathophysiology of AF in Endurance Athletes
The pathophysiologic mechanisms responsible for increased AF risk in individuals who practice an endurance sport remain unclear.19 Research has focussed on autonomic nerves and the possibility of exercise-related structural changes in the atria.
Influence of The Autonomic Nervous System
In the past decade, the role of the cardiac autonomic nervous system (ANS) in the initiation and maintenance of AF has been actively investigated. Several basic and clinical studies have indicated that initiation of paroxysmal AF requires the activation of both the sympathetic and parasympathetic components of the cardiac ANS.20-23Coumel24 studied the influence of autonomic innervations in the appearance of AF and atrial flutter, finding that “vagal AF”: [1] predominantly affects males between 30 and 50 years of age [2] , usually occurs at night and rarely occurs between breakfast and lunch when the sympathetic tone is high [3], rarely occurs during exercise or emotional stress [4], is frequently triggered during relaxation after stress and [5] is often preceded by bradycardia lasting from seconds to hours. However, he did not establish a relationship between these episodes of AF and sport practice. Experimental animal models have demonstrated that atrial fibrillation can be induced by acetylcholine25 and that increasing vagal tone shortens the atrial refractory period, which, combined with atrial stimulation, induces AF.26 In the GIRAFA study, 70% of consecutive patients with lone AF had vagal AF. Therefore, the increased vagal tone induced by endurance sport practice could facilitate the appearance of AF.
Exercise and Structural Changes in The Atria (Fibrosis, Inflammation and Dilatation)
Whether long-term endurance sport practice induces structural changes in the atrium (e.g., enlargement, fibrosis) that may create a favourable substrate for the disease- similar to hypertension or structural heart disease as a consequence of chronic volume and pressure overload- is a matter of debate. A small study by Frustaci et al.27 analyzed the structural changes in the atria of patients with lone AF. Inflammatory lymphonomonuclear infiltrates, compatible with myocarditis, were found in 66% of the patients; a non-inflammatory cardiomyopathic process in 17%; and patchy fibrosis in the remaining 17%.
Recent experimental data from our group show that endurance exercise may induce fibrosis in a rat model of chronic exercise, particularly at the atrial and right ventricular level.28 Therefore, it seems that endurance exercise may produce pathological changes in the heart, at least in an experimental model. Lindsay and Dunn29 showed an increase in humoral markers of fibrosis in veteran athletes compared to sedentary subjects, suggesting that long-term sport practice may provoke fibrosis as part of the hypertrophic process in veteran athletes.
Much attention has been devoted in the past few years to the role of inflammation in AF. The contribution of the inflammatory cascade to AF onset is suggested by the high incidence of AF in post-operative cardiac patients, who are in a state of intense inflammation.30,31,32 Other studies have suggested that inflammation leads to “atrial myocarditis” with subsequent electrical and structural atrial changes, resulting in AF initiation and persistence.33,34 Left atrial dysfunction has been described in patients with increased CRP but without AF, suggesting that inflammation affects left atrial function.35 Many studies have related an increase in CRP and interleukins in both paroxysmal and persistent AF.34-36 Elevated CRP and IL-6 might also contribute to generation and perpetuation of AF, as evidenced by marked inflammatory infiltrates, myocyte necrosis, and fibrosis found in atrial biopsies of patients with lone AF.27
Additionally, previous studies have found that excessive training may lead to tissue injury, which activates circulating monocytes, in turn producing large quantities of IL-1β and/or IL-6 and/or TNF-α and systemic inflammation. Their findings show a relationship between these inflammatory biomarkers and AF.37,38 However, there are no studies in the literature that confirm an association between AF, inflammation and exercise.
Although the underlying mechanism for structural changes is not clear, recent echocardiographic data suggest that structural remodelling is often present in the atrium of elite athletes without AF. Pelliccia et al.17 described the remodelling induced by exercise in elite sport athletes. Their study shows that those involved in regular endurance practice have a larger atrium than the sedentary controls. Furthermore, a significant proportion (20%) showed enlarged atria according to established normal values.
GIRAFA study data14 showing that patients with LAF had a larger atrium than the controls suggest that subtle structural changes at the atrial level may account for the appearance of AF. The study further showed that patients with a first episode of AF had the same atrial size compared to those suffering recurrences, which might mean that structural changes were present before onset of AF. On the other hand, patients with AF had larger LV mass, even after normalizing for body surface area. This further supports the idea that exercise had some repercussions in the ventricles, although the diastolic function index did not differ from sedentary controls. Although diastolic dysfunction has been proposed as the mechanism for atrial enlargement, it seems that volume and pressure overload act directly in the atrium, even before acting at the ventricular level.
Clinical Characteristics of Sport-related Atrial Fibrillation
The typical clinical profile of sport-related AF is a man in his forties or fifties who has been involved in regular endurance sport practice since his youth and is still active in middle age. The AF is usually paroxysmal with crisis, initially very occasional and self limited, then progressively increasing in duration. Characteristically, AF episodes occur at night or after meals. As many as 70% of patients may suffer predominantly from vagal AF.14 Since the AF crisis rarely occurs during exercise, the patient is reluctant to accept a relationship between the arrhythmia and sport practice. The crises typically become more frequent and prolonged over the years and AF becomes persistent. Progression to permanent AF has been described by Hoogsteen et al.11 in 17% of individuals in an observational series. In the GIRAFA study, 43% presented persistent AF.14 The AF crisis frequently coexists with common atrial flutter in many patients.
Treatment for AF in Athletes
Data’s on the reversibility of arrhythmia upon sport cessation are scarce. Furlanello et al.39 described a good response to sport abstinence in top-level athletes with AF. Our observations suggest that limiting physical activity seems to significantly reduce the number of crises, particularly in those with recent onset and minimally dilated atrium. However, these patients are very dependent on physical activity and it is difficult for them to follow this advice to reduce the risk associated with endurance sport practice. Biffi et al.40 also showed a very significant decrease in ventricular ectopy in athletes upon sport cessation.
After all of the potential contributing factors have been eliminated (i.e., medical conditions such as hyperthyroidism, pericarditis, Wolff-Parkinson-White syndrome, hypertrophic cardiomyopathy or long QT syndrome; substances such as cocaine, caffeine, anabolic steroids and sympathomimetics in cold medicines), the recommendation of the Study Group on Sports Cardiology of the European Association for Cardiovascular Prevention and Rehabilitation41 is that athletes in an early stage of paroxysmal AF discontinue training for two months to stabilize sinus rhythm. The degree of improvement during this resting period determines if athletes can resume their training.
In athletes without other cardiac disorders, the recommendation for sports participation also largely depends on the ventricular rate during AF. If there is a history of high ventricular rate or hemodynamic instability during AF, the athletes should be instructed to stop exercising if palpitation or related symptoms occur. Such patients may need medications that slow the ventricular rate, ideally at doses that do not cause sinus bradycardia at rest or chronotropic incompetence during exercise. When the heart rate during recurrent paroxysms of AF (or during permanent AF) is acceptable at an athlete’s maximum physical performance and there are no signs of hemodynamic impairment, sports activity can be resumed.
Task Force 7 of the 36th Bethesda Conference42 recommended that athletes with asymptomatic AF in the absence of structural heart disease be permitted to participate in any competitive sport, provided they maintain a ventricular rate that increases and slows appropriately and is comparable to that of a normal sinus response in relation to the level of activity, while receiving no therapy or therapy with AV nodal-blocking drugs. Asymptomatic athletes who have AF episodes lasting 5 to 15 s with no increase in duration during exercise can participate in all sports.
The possible long-term role of medications (ACE inhibitors, angiotensin inhibitors, Calcium-antagonists or beta-blockers) in preventing cardiac hypertrophy remains to be elucidated, although angiotensin inhibitors do seem to play a role in improving the results of cardioversion or AF ablation.43,44 Particular caution should be exercised with the monotherapeutic use of class 1 antiarrhythmic drugs in AF patients. These drugs may prevent AF recurrences, but also can convert AF into slow atrial flutter, which may conduct one-to-one to the ventricles during situations of high sympathetic tone.45,46 Impregnation of the ventricles with the class 1 drug will lead to broad QRS complexes (resembling VT) and profound negative inotropic effects that can result in cardiogenic shock and even sudden death. Class 1 drugs can be initiated for the prevention of AF episodes after adequate ventricular rate control during exercise has been assured.
When therapy with class 1 drugs is indicated in athletes, a combination with diltiazem or prophylactic ablation of the flutter circuit must be considered. The “hybrid” therapy of class 1 drugs and the ablation of flutter may obviate the need for maintenance therapy with bradycardic agents.47,48
In some athletes with paroxysmal AF, class 1 drugs can be used for acute reconversion therapy, known as the “pill-in-the pocket” approach.49 It is prudent to instruct these patients to refrain from sports as long as the arrhythmia persists and until at least one half-life of the antiarrhythmic drug has passed.
On the other hand, circumferential pulmonary vein ablation (CPVA) is established as an effective and safe treatment of AF, with success rates ranging from 30% to 85% in the general population.50 A study by Furlanello et al.51 described a highly successful ablation series, with 90% success after a mean of two ablation procedures in 20 athletes, without major complications.
A recent study by our group of CPVA effectiveness after a first procedure52 showed no difference between a population of endurance athletes with lone AF and other patients with AF figure 1; endurance sport practice was not a predictor of the results of the ablation procedure. Our study confirmed LA diameter and long-standing AF as the most powerful predictors of AF recurrence after CPVA; in addition, persistent AF showed a trend toward a higher probability of recurrence (p=0.054).
Figure 1. Kaplan-Meier curves for long-term freedom from recurrent arrhythmias after a single ablation procedure in lone AF athletes group (dashed line) and control group (solid line). Reproduced with permission from Calvo et al.52
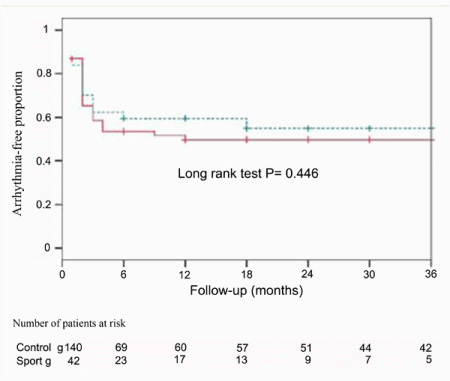
In patients with predominant atrial flutter, ablation of the flutter is frequently associated with a higher incidence of AF recurrences, as pointed out by Heidbuchel et al.16 However, although ablation seems to be quite effective, endurance sport cessation associated with drug therapy seems to us a more suitable approach as an initial therapy, particularly in non-professional veteran athletes.
In competitive and leisure-time athletes with unsuccessful rhythm control or under rate control therapy, anticoagulation may be necessary depending on the presence of classic risk factors for thromboembolic events.53 Anticoagulation therapy excludes these individuals from sports with a risk of bodily collision or trauma.41
Regular exercise has proven to be a healthy habit that decreases risk factors and atherosclerosis. However, long-term endurance sport practice has been demonstrated to increase the risk for recurrent AF, showing a relationship between accumulated hours of practice and AF risk. The underlying mechanisms remain to be elucidated, although structural atrial changes (dilatation and fibrosis) are probably present. Therefore, further studies are needed to clarify whether a threshold limit for the intensity and duration of physical activity may prevent AF, without limiting the cardiovascular benefits of exercise.