Smartwatch Alert Mimicking Implantable Cardiac Defibrillator Alarm During Sleep
Libo Wang, Anna Catino, Tirah Sheppard, Rashmee U.Shah, T. Jared Bunch, Benjamin A. Steinberg
Libo Wang, MD
Libo Wang, MD
Cardiovascular Medicine
30 N 1900 E, Room 4A100
Salt Lake City, Utah 84132
Consumers are now able to purchase mobile devices with built in arrhythmias alerts and other activities. Two devices (AliveCor KardiaBand and Apple Watch Series 4) are currently cleared by the FDA for single lead electrocardiographic detection of atrial fibrillation (AF) in those who do not have a history of diagnosed arrhythmia, based on feasibility studies shown over 99% specificity 1,2. Therefore, it is likely these devices will lead to increased healthcare utilization due to both true-positive and false-positive diagnostic alerts. Here we present the case of atypical healthcare presentation due to over-monitoring with consumer devices.
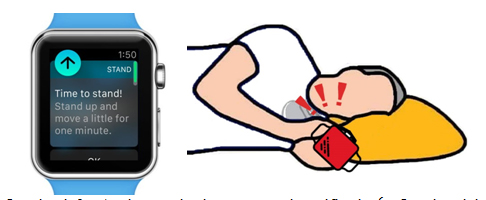
A 71-year-old male with a history of anxiety and chronic lymphocytic leukemia (CLL) on ibrutinib was diagnosed with new onset AF and nonischemic cardiomyopathy. Given the complexity of his medical management and increased risk of bleeding due to underlying CLL with thrombocytopenia, AF management was challenging and led to significant stress and anxiety of the patient. Maintenance of sinus rhythm was unsuccessful owing to recurrent AF triggers in the setting of ongoing ibrutinib therapy, as well as baseline bradycardia, and a conservative rate-only control strategy was pursued. Despite goal directed medical therapy for heart failure, stress cardiac MRI revealed a persistently reduced ejection fraction of 30-35%, without evidence of ischemia or infarction. The patient expressed escalating fears about dying from his newly diagnosed cardiac disease rather than his hematologic malignancy. He underwent placement of a dual chamber implantable cardioverter defibrillator (ICD) for primary prevention of sudden cardiac death and to support antiarrhythmic drug therapy. He subsequently underwent dofetilide loading and cardioversion for rhythm control of AF. Due to significant stress and anxiety around his worsening medical condition, he purchased an Apple Watch to, “monitor his heart rhythm”, despite the presence of continuous cardiac rhythm monitoring by ICD. One month after ICD placement, our patient awoke to recurrent vibratory sensations in the area of his ICD; an Abbott device capable of vibration alerts. He called the electrophysiology team the next morning, with great anxiety, to report his concern that this was a sign of ICD dysfunction or a low-battery alarm. Review of his remote ICD alerts did not reveal any sign of device malfunction or alarms; however, the vibratory alert feature was confirmed to be active. On further questioning, patient reports his recently-purchased Apple Watch (Apple, Inc., Cupertino, CA), has a variety of alert settings that were set to vibratory alert. On review of the watch alerts, he reported prior alerts at the time of vibratory sensation prompted by inactivity (Example demonstrated in [Figure 1]). In the left lateral decubitus position, these alerts were likely confused as originating from his ICD.
Figure 1. On the left: Apple watch time to stand notification6. On the right: diagram demonstrating patient’s smartwatch alert in proximity to patient’s ICD while sleeping in a left lateral decubitus position.
AF and heart failure are associated with significant anxiety and stress 3, 4. Post-traumatic stress disorder among patients with inappropriate ICD shocks has been well described 5. As consumer grade devices capable of rhythm detection become more accessible, usage of these devices will become more widespread with varying levels of user technological literacy and health literacy. While these devices may provide reassurance to patients, our case highlights that they can also lead to additional distress and reduced health-related quality of life. Individuals may present with alerts/alarms that may be true positives consistent with new diagnoses of arrhythmias, or false positives requiring clinician assessment and reassurance. Both may be associated with increased stress and anxiety. Despite promising performance of AF detection by the Apple Watch Study, it remains unclear if clinical encounters triggered by consumer-wearables lead to improved outcomes. As this case demonstrates, more healthcare is not necessarily better healthcare. Consumer arrhythmia monitoring devices will inevitably lead to increased healthcare encounters, with unclear benefit. This case also highlights one harm noted in the Apple Watch Study, in that some patients that wear an Apple Watch with frequent feedback experience higher levels of anxiety. As appropriate, targeted deployment and implementation of these technologies remains to be determined.
To our knowledge, this is the first case reported of a smartwatch alert erroneously mimicking as a pacemaker/ICD alert in a patient with an implanted device. We also recognize that increased vigilance due to the novel coronavirus infection may have contributed to our patient’s excess caution in his self-care. No direct clinical adverse event resulted. The alert resulted in undue stress and anxiety by the patient, and required additional healthcare resources during a period of extreme resource scarcity.
Consumer grade wearable / mobile devices are becoming more prevalent in the cardiology patient population. Our patient, despite having remote monitoring capabilities and a continuously-recording ICD, felt compelled to also self-monitor with a consumer-based, wearable device. This may have been prompted by baseline anxiety, legitimate concern of his cardiac and hematologic comorbidities, and perceived benefit in a FDA-cleared wearable device with arrhythmia detection capabilities. Wearable external devices such as a smartwatch may mimic alarms originating from an implanted pacemaker or defibrillator, resulting in unnecessary healthcare encounters and furthering patient anxiety. While consumer-based, wearable arrhythmia monitoring technology has the potential to improve care while reducing healthcare resource utilization, careful deployment and management will be required.