Dofetilide Initiation and Implications of Deviation From the Standard Protocol – A Real World Experience
Tawseef Dar1, Ghulam Murtaza 2, Bharath Yarlagadda3, Bader Madoukh2, Lesley Bravin4, Venkat Vuddanda5, Valay Parikh4, Madhu Reddy4, Dhanunjaya Lakkireddy2
1Department of Nuclear Cardiology, Massachusetts General Hospital, Boston, MA.2Kansas City Heart Rhythm Institute and Research Foundation, Overland Park, KS.3Division of Cardiology, University of New Mexico, Albuquerque, NM.4Division of Cardiovascular Diseases, University of Kansas Hospital and Medical Center, Kansas City, KS.5Department of General Internal Medicine, Brigham and Women's Hospital, Harvard Medical School, Boston, MA.
Manufacturer/federal drug administration (FDA) recommends inpatient initiation of dofetilide with the manufacturer providing an initiation algorithm. The outcomes of algorithm deviation have not been reported outside of clinical trials.
We sought to perform a chart review of all the patients admitted for inpatient initiation of dofetilide to report on the incidence of protocol deviations and their implications.
We performed a retrospective review of all patients over a 15-month periodwho were initiated on dofetilide for the very first time or reinitiated on dofetilide after a break of three months or more at our institution. We assessed data about patients who were given dofetilide without adherence to the protocol (i.e. protocol deviation).
A total of 189 patients were included in the study with a median age of 66 ± 9 years. Mean baseline QTc interval was 436 ± 32 msec, and 61% (116/189) were in atrial fibrillation (AF) at the time of dofetilide initiation. In 9% (17/189) of patients, the drug was discontinued due to intolerance or inefficacy. Therapy in 49% (93/189) of patients was noted to deviate from manufacturer recommended protocol with deviations more than once in some patients during the same hospitalization. Baseline QTc exceeding 440 msec(>500msec in conduction abnormalities) was the most frequent deviation (25%; 47/189).Ventricular tachyarrhythmia occurred in 4% (7/189) of patients, did not differ between patients, and occurred with and without protocol deviations (5% vs 2%; p = 0.27).
In our retrospective study, there were frequent deviations from the manufacturer-recommended algorithm guidelines for dofetilideinitation, primarily due to prolonged baseline QTc interval. The impact of these protocol deviations on drug discontinuation was uncertain; however, significant adverse events were significantly higher in the deviation group compared to the group that fully adhered to the protocol. Further multicenter studies are warranted to clarify our findings.
Key Words : Dofetilide, Atrial fibrillation, Protocol, Deviation.
DhanunjayaLakkireddy, MD, FACC, FHRS
Executive Medical Director
The Kansas City Heart Rhythm Institute (KCHRI) @ HCA MidWest
Professor of Medicine, University of Missouri, Columbia, MO
12200, W 106th street, Oveccrland Park Regional Medical Center
Overland Park, KS 66215
Atrial fibrillation(AF) is the most prevalent cardiac arrhythmia. Treatment involves medications to control either the rate while in AFor to pursue a rhythm control strategy to maintain sinus rhythm. Although clinical trials have not revealed a mortality benefit of rhythm control strategy versus rate control strategy [1-3], some patients are extremely symptomatic while in AF and a rhythm control strategy is necessary for these patients. Rhythm control strategy is the current standard of care when rate control is ineffective orpatient is symptomatic despite controlled ventricular response [4]. AF leads to negative remodeling of the atria[5] and sinus rhythm restoration, aside from symptomatic improvement, has been show to reverse the process of remodeling and therefore break the vicious circle[6]. In addition, AF, with rates poorly controlled despite use of maximal doses of AV nodal blocking agents, leads to tachycardia-mediated cardiomyopathy and ultimately congestive heart failure (CHF), making rhythm control strategy more suitable, especially in young patients[7]. One study of antiarrhythmic drug (AAD) therapy showed that patients who received dofetilide had a significantly higher probability of remaining in sinus rhythm up to 1 year compared to those who received placebo, even though there was no benefit in all-cause mortality.[8] Sinus rhythm can be restored via pharmacologic or electrical cardioversion and can be maintained with use of AAD.AAD continue to remain the first line maintenance therapy and are often utilized to facilitate conversion from AF to sinus rhythm[9].
Amiodarone is the most potent antiarrhythmic agent but is associated with significantside effects most of which are non-cardiac. Dofetilide, a class III antiarrhythmic agent, blocks rapid component of delayed rectifier potassium current(IKr) inhibitor channels and is associated with minimal non-cardiac side effects. It increases action potential duration due to delayed repolarization and prolongs QT interval as a result of prolongation of effective and functional refractory period of the His-Purkinje system and the ventricles. Dofetilide is moderately effective in converting AF to sinus rhythm butexhibits 58% efficacy in maintaining sinus rhythm at the end of one year[10]. The package insert pertaining to dofetilide specifies that in patients with supraventricular arrhythmias, the incidence of TdP at dosing >250mcg BID is 0%, at 250mcg BID is 0.3%, at >250-500mcg BID is 0.9%, and at >500mcg is 10.5% . As a result, the FDA, at the time of initial approval of dofetilide in 1999, mandated that dofetilide could only be started in patients after administering the initial 5 doses over three days in the inpatient setting with continuous EKG montitoring.
Furthermore, in July 2011, the FDA released a detailed protocol for initiation of dofetilide suggesting the starting dose and appropriate dosage changes depending on the changes in QTc post initiation and creatinine clearance. The adherence to this protocol and the consequences of deviation are unknown. Outside of the clinical trials involving dofetilide, there is very little data from the real-world experience regarding the frequency of protocol deviations and their outcomes.
Hence, we performed an observational study including patients who were admitted to our hospital for dofetilide initiation with an emphasis on patients whose therapy deviated from the manufacturer recommended protocol.
Upon getting approval from the Institutional Review Board, we performed a retrospective review of all patients, over a 15-month period, who were initiated on dofetilidefor the very first time or reinitiated on dofetilideafter a break of three months or moreat our institution.Patients were included if there was documentation of an EKG with QTc at baseline and two hours after each dose. Patients with long QT syndrome or prior history of cardiopulmonary resuscitation due to any cause were excluded from the study.Every attempt was made to discontinue all prior antiarrhythmics for a period amounting to five half-lives of respective drugs.
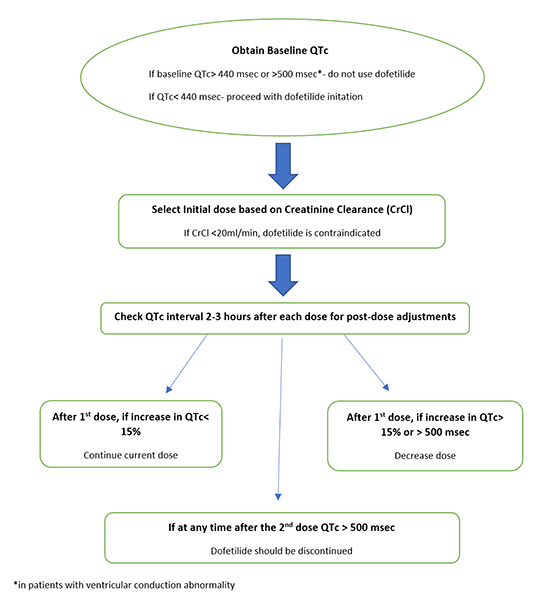
Patients were dosed as per University of Kansas Hospital Pharmacy protocol for inpatient initiation of dofetilide and the protocol was executed via an electronic order set which is in line with the FDA/manufacturer’s recommendations. Doses were mainly based on the initial QTc (or QT interval if heart rate was>60 beats/minute) and creatinine clearance as shown in Figure 1.
Protocol implementation was overseen by the pharmacy team. Every attempt was made to encourage compliance with the protocol. Any violation of FDA recommended dosing algorithm at any stage was considered a deviation from protocol (Figure 1).Efforts were made to detect those patients who received dofetilide not in accordance with the protocol.
Figure 1. Dofetilide dosing Algorithm (FDA approved/Manufacturer recommended).
Data collection and Study outcomes
From the chart review, we collected baseline demographic information, medical history, baseline laboratory blood test results, including creatinine clearance, electrolytes, EKG information pre-and post-dofetilide initiation, direct current cardioversion, and pharmaceutical interventions.
The primary outcome of the study was to evaluate the incidence of deviations from FDA-recommended protocol for inpatient dofetilide initiation in clinical practice. Secondary outcomes included incidence of intolerance/inefficacy leading to drug discontinuation, need for electrical cardioversion, and incidence of ventricular tachyarrhythmia specifically in patients in whom there was a protocol deviation.
Drug Intolerance: Symptomatic drug adverse effects including symptomatic proarrhythmias; symptomatic adverse effects eg: headache, dizziness, nausea, back pain, and abdominal pain; laboratory abnormality including QT prolongation but without symptoms leading to physician directed drug discontinuation.
Serious adverse event (SAE): Any ventricular tachyarrhythmia read as Torsades de pointes, ventricular fibrillation, or sustained ventricular tachycardia was classified as a serious adverse event.
Continuous
variables are expressed as mean ±standard deviation (SD) if variables are normally distributed, and median (interquartile range) when deviations from normality were present. Categorical variables are expressed as counts and percentages. Categorical variables were compared between the groups using chi-squared test or fisher's exact test. Continuous variables were compared using independent sample t test. A two tailed p value less than 0.05 was considered statistically significant. Binary logistic regression model was used to determine factors related to serious adverse events. Statistical analysis was performed using IBM SPSS Statistics version 23.0 (IBM, Armonk, New York).
Baseline characteristics (AF vs Sinus rhythm)
Baseline characteristics of the 189 patients included in the study are shown in Table 1. Mean age was 66 ± 9 years (range 34 – 86 years), and 63 % (n=120/189) were males. Hypertension, coronary artery disease,and cardiomyopathy were seen in 50 % (n=95/189), 29 % (54/189) and19% (35/1189) of patients, respectively. Severe cardiomyopathy (left ventricular ejection fraction >35%) was seen in 14% (27/189) of patients. The majority of patients (164/189, 87%) had a creatinine clearance ≥ 60 mL/min, calculated using the Cockgroft Gault formula. At the time of dofetilide initiation, 61 % (116/189) were in AF or AFL. Mean baseline QTc interval was 436 ± 32 msec (range 349-530 msec). With regard to the distribution of patients among various QTc intervals, 66% (125/189) of patients had a QTc ≤ 440 msec, 32% (61/189) had a QTc of 441-500 msec, and 2% had a QTc of more than 500 msecat baseline. There was a 19% (35/189) prevalence of a baseline ventricular conduction abnormality.
Table 1. Demographics and other clinical variables of the overall study population
| Variable |
Dofetilide Initiation (Total N=189)
n (%)
|
| Demographics:
Female/Male,
Median age in years ± SD,
Obese (ABW > 40% IBW)
|
69 (37%) /120 (63%),
66 ± 9 (34-86),
77 (41%)
|
| Medical History:
Hypertension,
Coronary artery disease,
Ischemic cardiomyopathy,
CHF or EF > 40%
|
95 (50 %),
54 (29%),
35 (19%),
27 (14%)
|
| Creatinine Clearance,
≥ 60 mL/min
|
164 (87%)
|
| Serum Electrolytes:
Median potassium (range) in mEq/L,
Median magnesium (range) in mEq/L,
|
4.3 (3.3-5.1),
1.9 (1.3-2.5)
|
| Electrocardiogram:
Mean baseline QTc (range) in msec ±SD (range),
In AF or AFl upon initiation |
436 ± 32 (349-530),
116 (61%) |
AF= atrial fibrillation; AFl= atrial flutter; ABW= adjusted body weight; IBW= ideal bodyweight
Chemical vs electrical cardioversion
Of the 116 patients who presented in AF or AFL, 65% (75/116) required direct current cardioversion while being treated with dofetilide. Overall, 50% (94/189) of patients required electrical cardioversion at some point while in the hospital secondary to AF persistence. At the time of discharge, 11% (20/189) of patients were not in sinus rhythm despite attempts at electrical or chemical cardioversion. In addition, 10% (18/189) were brought back for a repeat attempt at electrical cardioversion 1-2 weeks later, of whom 6% (11/189) had successful electrical cardioversion, with drug discontinuation occurring in 4% (7/189) of patients.
Therapy in 49% (93/189) of patients was noted to deviate from manufacturer-recommended protocol with some of the patients deviating more than once, in different ways, during the same hospitalization.The reasons for deviations are listed in Table 2. Baseline QTc exceeding 440 msec(>500msec in patients with conduction abnormalities) was the most frequent deviation (25%;47/189), followed by QTc interval > 500 msec any time after the second dose without subsequent discontinuation of dofetilide therapy (18.5%; 35/189) (Figure 2).
Figure 2. Bar chart diagram showing different types of deviations.
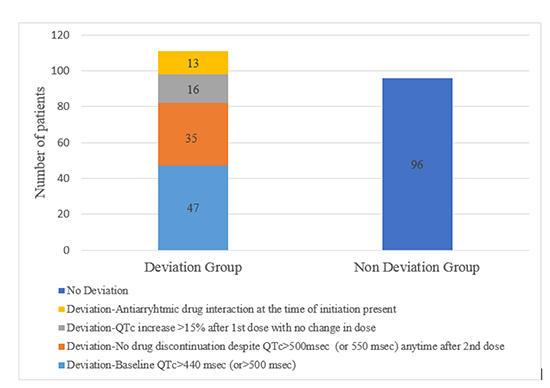
Table 2. Protocol deviations encountered during Dofetilide loading
| Types of deviations from manufacturerrecommended guidelines |
No. of patients n(%) |
| 1. Dofetilide initiation with an elevated baseline QTc > 440 msec (or > 500 msec in conduction abnormalities) |
47 (25) |
| 2. QTc interval > 500 msecanytime after the 2nd dose without subsequent discontinuation of dofetilide therapy |
35(18.5) |
| 3. Increase of QTc interval > 15% above baseline post 1st dose without subsequent dofetilide dose reduction |
16 (8.5) |
| 4. Antiarrhythmic drug interaction at time of initiation (patients who were previously on amiodarone or dronedarone and had shorter washout periods than recommended) |
13 (7) |
Dofetilide dosing and QTc prolongation
QTc prolongation (>500msec) was present in 18.5% of patients after getting the second dose, but only two of those patients had persistently prolonged QTc at the completion of loading dose in whom the drug was discontinued. The prolongation of QTc interval >15% occurred in 8.5% of patients after the first dosing but without significant issues during the subsequent dosing and at time of discharge. None of these patients needed drug discontinuation. Even though 6.5% of patients did not have thorough drug washout from other AADs, there was no significant impact on QTc and/or subsequent clinical outcomes. Post chemical or electrical cardioversion, the QTc intervals at the time of discharge were within the normal limits.
Drug discontinuation and Adverse events (Deviation vs non deviation group)
Overall, dofetilide therapy was discontinued in 9% (18/189) of patients secondary to inefficiency or drug intolerance. The rate of drug discontinuation for “deviation group” was 13% (12/93) vs 6% (6/96) for “non-deviation” group (p= 0.14). A total of 11 patients (torsades de pointes in five following prolonged QTc, non-sustained ventricular tachycardia in two, and QT prolongation without torsades de pointes in four) had drug intolerance leading to drug discontinuation. The overall incidence of SAEs was 2.6% (5/189). The rate of SAE was 4% (4/93) in “deviation group” vs 1% (1/96) in “non-deviation group” (p value = 0.20). One patient in the deviation group had a baseline QTc of 530msec and developed ventricular fibrillation while being monitored in the hospital. Interestingly, out of 5 patients with SAEs, 4 had QTc>500 milliseconds at one point after the second dose, but only 1 had continued the drug despite such QTc prolongation. There were only two patients who had a baseline QTc >440 msec (including one with baseline QTc >500). Comparison of baseline characteristics of the two groups is shown in Table 3.
Of note, in patients with severe cardiomyopathy (ejection fraction >35%), deviation was encountered in more than half of the patients (55%, 15/27) with only 1 (3.7%, 1/27) SAE reported.
Table 3. Demographics and clinical characteristics of deviation and non-deviation groups
| Characteristics |
Deviation Group (n=93) |
No Deviation Group (n=96) |
P value |
| Demographics |
|
|
|
| Mean Age |
66 ± 8.3 |
65±9.4 |
0.12 |
| Gender-Male,
Female
|
60 (65%),
33 (35%)
|
60 (62.5%),
36 (37.5%)
|
0.68 |
| Comorbidities |
|
|
|
| LVEF >40% |
15 (16%) |
12 (12.5%) |
0.42 |
| Heart rate >60 at initiation |
20 (16.8%) |
3 (4.3%) |
0.01 |
| Open heart surgery in last 2 months |
1 (0.8%) |
0 (0%) |
0.447 |
| MI or unstable angina in the past |
5 (5%) |
0 |
0.086 |
| AF upon initiation |
67 (72 %) |
49(51%) |
0.054 |
| Creatinine clearance |
82 ±17 |
80±20 |
0.93 |
| Serious adverse event |
4 (4%) |
1 (1 %) |
0.20 |
| Drug discontinuation rate |
12 (13%) |
6 (6%) |
0.14 |
| QTc interval at discharge in msec |
435 ± 26 |
428± 19 |
0.66 |
n (%) or mean ± SD; LVEF= left ventricular ejection fraction; MI= myocardial infarction
AAD continue to remain the first-line maintenance therapy after cardioversion to sinus rhythm. However, most of the available antiarrhythmic drugs are associated with serious cardiac and/or non-cardiac side effects like arrhythmias, QT prolongation, and liver or lung toxicities. Dofetilide is one of these antiarrhythmic drugs indicated for maintenance of sinus rhythm (Class 1A recommendation) based on reports from several trials like SAFIRE-D (Symptomatic Atrial Fibrillation Investigative Research on Dofetilide) trial[10] and DIAMOND (Danish Investigations on Arrhythmia and Mortality on Dofetilide) study[12]. However, due to risk of dose-dependent QT prolongation and serious arrhythmias, including torsades de pointes, the FDA has mandated that dofetilide initiation take place in the inpatient setting under continuous monitoring following the set protocol per manufacturer (Figure 2). Despite its favorable side effect profile compared to other antiarrhythmic drugs,dofetilide is still being used as a second or third line drug by most cardiologists [13]. The reason for such practice could be prerequisites like inpatient initiation as well as strict protocol guidelines per manufacturer. Of note, dofetilide is available in only a few countries outside of the U.S., and the restrictions on its use are different from restrictions in the U.S.
We completed a retrospective review of patients who underwent dofetilide initiation at our center and looked at various parameters during initiation to see how strictly cardiologists were following the manufacturer protocol, and to discern the implications of protocol deviations. We found that almost half of the patients (49%, 93/189) had a deviation from the protocol, and some patientshad recurrent deviation during their initiation process.
The most common deviation from the protocol was elevated baseline QTc (>440 msec or >500 msec if ventricular conduction abnormality was present) followed by continuation of the drug despitea measured QTc >500 msec (or>550 msec if ventricular conduction abnormality present) any time after the second dose. The overall incidence of serious arrhythmias was 2.6%, which is the same rate reported in previous studies (SAFIR-D). Similarly, the SAE rate in patients with severe cardiomyopathy is the same as reported in DIAMOND study[12].The mean baseline QTc in most of these patients was 436 msec. There were individual variations in the QTc cut-off values with which each of the prescribers were comfortable. Oftentimes, an accurate QTc interval may be difficult to calculate due to the presenting rhythm of AF and AT, despite correction algorithms based on the rhythm. Unless the QTc is grossly abnormal, borderline QTc intervals above 440 msec may still clinically do well.
The inherent limitations in accurate QTc interval measurement when patients are in AF or AFL may lead to overestimation of QTc. The 440msec cut off may not be valid entirely based on real world experience. Unless the QTc is grossly abnormal, a lot of patients can have dofetilide initiation and subsequent cardioversion to be able to accurately measure QTc. Some of the standard protocol numbers used are mostly arbitrary and supported by historical data. These guidelines should be updated periodically based on real world data. Prospective drug registries can help answer a lot of these questions.
Our study was conducted in a single center and was retrospective in nature. The reasons cited by the physicians for drug initiation despite protocol deviation were not reported. Of all the enrolled patients, 39% of patients who had a protocol deviation were in normal sinus rhythm; accurate QTc measurement in these cases would not have been difficult. A larger multi-center registry for dofetilide loading and maintenance would be helpful in better understanding the real-life implications and for better defining the loading parameters and variability in dosing among patients with varying baseline characteristics. Lastly, despite protocols in place to facilitate adherence, some providers did not adhere to the protocol due to personal preference and experience. Methods to emphasize better protocol adherence and documentation are needed.
Our single center experience withdofetilide initiation showed that deviations from protocols are routinely encountered. However, the implications of most of these deviations on drug discontinuation need further elucidation, and the group that had protocol deviations had significantly more serious adverse events compared to the group that had complete protocol adherence. The protocol for dofetilide initiation may need some revision pending further robust data. Larger multicenter studies are needed to better delineate our findings.