The Impact of Atrial Fibrillation on In-Hospital Outcomes in Patients With Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing Coronary Revascularization with Percutaneous Ventricular Assist Device Support
Gupta Sonu1*, Desai Rupak2*, Hanna Bishoy1, Deshmukh Abhishek3, Kumar Gautam4, Sachdeva Rajesh125, E Berman Adam5
1Division of Cardiology, Morehouse School of Medicine, Atlanta, GA, USA..2Division of Cardiology, Atlanta VA Medical Center, Decatur, GA, USA..3Division of Cardiology, Mayo Clinic, Rochester, MN, USA..4Division of Cardiology, Emory University School of Medicine, Atlanta, GA, USA..5Division of Cardiology, Medical College of Georgia, Augusta University, Augusta, GA, USA..*Authors share equal contribution to the manuscript..
Atrial fibrillation (AF) is common in acute myocardial infarction complicated by cardiogenic shock (AMI-CS) requiring percutaneous ventricular assist device (pVAD-Impella®) support during percutaneous coronary interventions (PCI). We evaluated the effects of a coexistent diagnosis of AF on clinical outcomes in patients with AMI-CS undergoing PCI with pVAD support.
The National Inpatient Sample (2008-2014) was queried to identify patients with AMICS requiring PCI with pVAD support and had a concomitant diagnosis of AF. Propensity-matched cohorts (AF+ vs AF-) were compared for in-hospital outcomes.
A total of 840 patients with AMICS requiring PCI with pVAD support (420 AF+ vs 420 AF-) were identified in the matched cohort. Patients with AF were older (mean 69.7±12.0 vs 67.9±11.3 yrs, p=0.030). All-cause in-hospital mortality rates between the two groups were similar (40.5% vs 36.7%, p=0.245); however, higher postprocedural respiratory complications (9.5% vs 4.8%, p=0.007) were seen in AF+ group. In-hospital cardiac arrests were more frequent in the AF- group (32.0% vs 19.2%, p<0.001). We examined the length of stay (LOS), transfer to other facilities, and hospital charges as metrics of health care resource consumption and found that the AF+ cohort experienced fewer routine discharges (13.1% vs 30.2%), more frequent transfers to other facilities including skilled nursing facilities or intermediate care facilities (27.3% vs 17.8%; p<0.001), more frequently required the use of home health care (14.3% vs 7.1%; p<0.001). The mean LOS (11.9±10.1 vs 9.11±6.8, p<0.001) and hospital charges ($308,478 vs $277,982, p=0.008) were higher in the AF+ group.
In patients suffering AMICS requiring PCI and pVAD support, a coexistent diagnosis of AF was not associated with an increase in all-cause in-hospital mortality as compared to patients without AF. However, healthcare resource consumption as assessed by various metrics was consistently greater in the AF+ group.
Key Words : acute myocardial infarction, cardiogenic shock, atrial fibrillation, arrhythmia, percutaneous ventricular assist device (pVAD);, Impella, outcomes.
Correspondence to: Adam E Berman, MD, MSc, MPH
Division of Cardiology, Medical College of Georgia, Augusta University,
1120 15th Street, BBR6518A, Augusta, GA 30912, USA
Atrial fibrillation (AF) is the most prevalent supraventricular arrhythmia observed in the general population [1]. Advanced age is the most common risk factor, with a lifetime risk of developing AF >10% after the age of 55 years [2]. Prior studies have demonstrated that the presence of AF portends a worse outcome in patients with AMI and may serve as an independent predictor of mortality [3]-[6].
AF is a common pre-existing comorbidity in patients with AMI, but it may also develop de novo owing to the acute hemodynamic insult to the atria in the setting of AMICS, where acute ventricular pump dysfunction leads to an acute increase in intracardiac pressures and mechanical distention of the atria. These hemodynamic and structural changes to the atria result in electrophysiologic derangements that may lead to AF, the development of which can result in further hemodynamic compromise.
Hemodynamic support devices have often been utilized to bridge recovery in patients with acute myocardial infarction complicated by cardiogenic shock (AMICS) undergoing percutaneous coronary intervention (PCI). Owing to its favorable outcomes and relatively noncomplex procedural deployment, pVAD devices, particularly the Impella device (Abiomed, Danvers, MA), are increasingly utilized. Based on studies [7]-[10] suggesting the possibility of improved survival outcomes with the use of pVAD for protected PCI, the Impella ® has found increasing use in clinical practice in the setting of AMICS [11]. Considering the previously demonstrated association of worse clinical outcomes in AMI patients with AF, we performed a propensity-matched analysis investigating the impact of AF on in-hospital outcomes in patients with AMICS requiring pVAD support for revascularization using a large national inpatient database.
We conducted a retrospective observational study utilizing the US National Inpatient Sample (NIS) database from 2008 through 2014. The Agency for Healthcare Research and Quality (AHRQ) as a part of the Healthcare Cost and Utilization Project (HCUP) keeps a record of NIS, which nearly depicts data from an estimated 35 million weighted discharges annually [12]. The NIS embodies a stratified 20% section of hospital records from community hospitals across 44 states, representative of over 95% of the US population. The NIS database includes up to 25 discharge diagnoses, along with on admission comorbidities, up to 15 inpatient records of procedures, and documented complications during a hospital stay. Sampling design and discharge weights (DISCWT) provided in the database were incorporated to appraise national estimates. The information on the self-weight design of the database is detailed online [12]. The International Classification of Diseases, Ninth Revision Clinical Modification (ICD-9 CM) codes were utilized to identify the diagnoses of interest, comorbidities and primary endpoints. Informed consent and institutional review board approval for this study were not required owing to the de-identified data source.
Adult hospitalizations with a principal diagnosis of AMI complicated by CS were identified from January 2008 through December 2014. Of the AMICS encounters queried, those in which the patient required PCI with pVAD support were further identified using ICD-9 CM codes. Inpatient encounters in which other mechanical hemodynamic support devices were utilized [e.g. LVAD, intra-aortic balloon pump (IABP), extracorporeal membrane oxygenation (ECMO)] or those with missing data were excluded. Eligible hospitalizations were then further stratified into two study cohorts, AF+ vs. AF-, using the ICD-9 CM code 427.31. The study population was identified using an algorithm as shown in [Figure1].
Figure 1. Study population selection algorithm
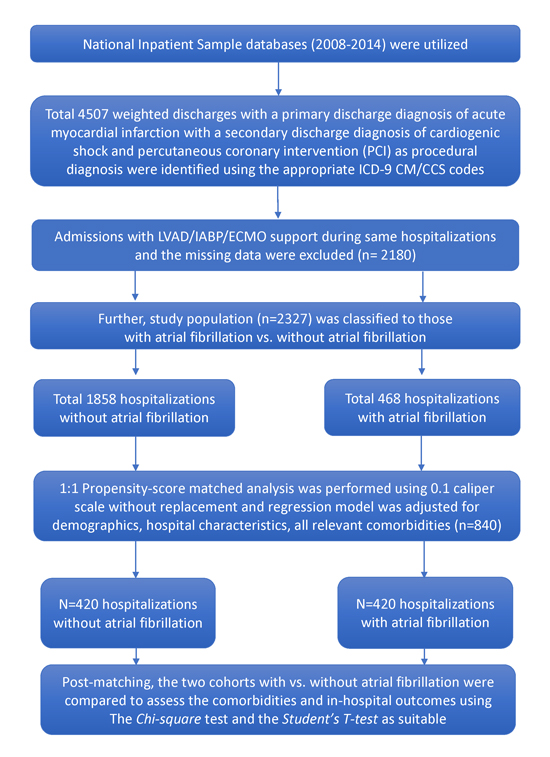
Baseline demographics, hospital characteristics, medical comorbidities, and complication rates were compared between the AF+ and AF- groups. Patient-level sociodemographic and hospital-level attributes were directly drawn out as provided, whereas the AHRQ comorbidity indicators were employed to identify on admission comorbid disorders using apposite ICD-9 CM codes as detailed earlier [13], [14]. We queried the AMICS cases for in-hospital complications using the validated diagnostic codes present among the secondary diagnoses fields (Supplementary Table 1 ).
Supplementary Table 1. ICD-9 CM and CCS Codes Used to Identify Comorbidities and Procedural Complications
| Comorbidities/Complications |
ICD-9 CM/CCS |
Codes (Diagnostic/Procedural) |
| Acute Myocardial Infarction |
CCS |
100 |
| Atrial fibrillation |
ICD-9 CM |
427.31 |
| Cardiogenic shock |
ICD-9 CM |
785.51 |
| Multivessel percutaneous coronary intervention |
ICD-9 CM |
00.41 00.42 00.43 |
| Hemodialysis |
ICD-9 CM |
39.95 V45.1 |
| Status pacemaker |
ICD-9 CM |
V45.01 |
| Smoking |
ICD-9 CM |
305.1, V15.82 |
| Dyslipidemia |
CCS |
53 |
| Implantable Cardioverter Defibrillator status |
ICD-9 CM |
V45.02 |
| Ventricular tachyarrhythmia |
ICD-9 CM |
427.1, 427.41, 427.42 |
| Prior transient ischemic attack/stroke |
ICD-9 CM |
V12.54 |
| Percutaneous ventricular assist device (Impella®) |
ICD-9 CM |
37.68 |
| Intraaortic balloon pump |
ICD-9 CM |
37.61 |
| Extracorporeal membrane oxygenation |
ICD-9 CM |
39.65 |
| left ventricular assist device |
ICD-9 CM |
37.66 |
| Hemorrhage or hematoma complicating a procedure |
ICD-9 CM |
998.1, 998.11, 998.12 998.13 |
| Perioperative autologous transfusions of whole blood or blood components |
ICD-9 CM |
V58.2, 99.00 |
| In-hospital cardiac arrest |
ICD-9 CM |
427.5 |
| Postoperative myocardial infarction |
CCS |
100 |
| Postoperative stroke |
ICD-9 CM/CCS |
997.02, 109 |
| Post procedural respiratory complications |
ICD-9 CM |
997.3 997.31 997.32 99739 |
| Acute kidney injury + dialysis |
ICD-9 CM |
584.9+39.95 |
| Postoperative infection |
ICD-9 CM |
998.5, 998.51, 998.59, 999.3, 038.0, 995.91, 995.92 |
| Septic shock |
ICD-9 CM |
785.52 |
ICD-9 CM = The International Classification of Diseases, Ninth Revision, Clinical Modification CCS = Clinical Classifications Software, https://www.hcup-us.ahrq.gov/toolssoftware/ccs/ccs.jsp
The primary outcomes were all-cause in-hospital mortality and the development of complications, including cardiac arrest, post-procedural hemorrhage, respiratory complications, acute kidney injury (AKI) requiring dialysis, infection and septic shock. The secondary outcome was the economic burden of disease, as measured by the length of stay (LOS, in days), discharge disposition and the total cost of the inpatient encounter (in US dollars).
Categorical variables were represented as numbers and proportions and assessed with the Chi-Square test. Continuous variables were reported as the mean and standard deviation and were matched using Student’s t-test. Discharge weights with strata and cluster designs provided by the HCUP were accounted for using complex modules to appraise nationwide assessments. To control the existent selection bias in the unmatched cohort, a propensity score-matched analysis of the AF+ vs. AF- AMICS patients’ cohort was accomplished. We performed a multivariable logistic regression model after adjusting for demographics, hospital-level elements and all relevant cardiovascular comorbidities present on admission to calculate a propensity score for each discharge record. The subsequent individually matched propensity score was used for the nearest neighbor matching of both groups (AF+ vs. AF-) with a caliper width tolerance of 0.01. Later, we compared and tabulated AMICS encounters requiring PVAD and the PCI outcomes between both AF and non-AF groups in the propensity score-matched cohort. Absolute standardized differences between pre-matched and post-matched cohorts were compared to assess the residual imbalances (Supplementary []). A greater than five percent post-matching difference between the two groups (AF and non-AF) was considered as statistical significance. IBM SPSS version 24.0 (IBM Corp., Armonk, NY) was applied to conduct the statistical analyses.
Supplementary Figure 1. Absolute standardized differences before propensity-score matching vs. after propensity-score matching

A total of 2326 encounters with a principal diagnosis of AMI complicated by CS requiring PCI with pVAD support were identified during the study period. 20.1% of these encounters also featured a concomitant diagnosis of AF. The study cohort was comprised of 468 AF+ vs 1,858 AF- patients. AMICS patients with AF who required pVAD support were significantly older (70.7± 11.8 vs 63.7±12.5, p <0.001) and more often white (72.2% vs 70 %, p <0.045) males. Compared to the AF- cohort, the unmatched AF+ cohort consisted of older (age >65yrs; 69% vs 47.2%, p <0.001), Hispanic (11.7 % vs 9.9%, p <0.045), non-electively admitted (93.6% vs 93.3%, p =0.806), Medicare (61.6% vs 49.7%, p <0.001) enrollees with higher income quartiles (23.3% vs 16.9%, p <0.001) and were admitted to small as well as large urban teaching and non-teaching hospitals [Table 1]. There was a higher proportion of males in both cohorts (71.6 % vs 78.6 %), though the proportion of females was higher in the AF+ group (28.4% vs 21.4%) (p=0.020). After propensity matching, a cohort of 420 AF and non-AF patients were comparable regarding race, admission day, type of admission, median household income, hospital bed size, location and region with a standardized difference of <10% between both groups.
Table 1. Study Population Undergoing Coronary Revascularization with PVAD (Impella®) Support Following Acute Myocardial Infarction Complicated by Cardiogenic Shock With vs. Without Atrial Fibrillation
| Variables |
Before Matching (weighted N=2327) |
After Matching (weighted N=840) |
|
| No AF (n=1,858) |
AF (n=468) |
P |
No AF (n=420) |
| Age (yrs) at hospitalization |
|
|
|
| Mean age (Mean±SD) |
63.7 ± 12.5 |
70.7 ± 11.8 |
<0.001 |
67. 9 ± 11.3 |
| 18-44 |
7.0% |
3.1% |
<0.001 |
* |
| 45-64 |
45.9% |
27.9% |
<0.001 |
34.7% |
| ≥65 |
47.2% |
69.0% |
<0.001 |
62.9% |
| Sex |
|
|
0.888 |
|
| Male> |
71.1% |
71.5% |
|
78.6% |
| Female |
28.9% |
28.5% |
|
21.4% |
| Race |
|
|
<0.045 |
|
| White |
70.0% |
72.2% |
|
73.8% |
| American African |
9.7% |
6.4% |
|
6.0% |
| Hispanic |
9.9% |
11.7% |
|
9.4% |
| Asian and Pacific Islander |
3.5% |
* |
|
* |
| Native American |
* |
* |
|
* |
| Others |
6.4% |
7.5% |
|
8.4% |
| Admission day |
|
|
0.022 |
|
| Weekend |
78.1% |
82.9% |
|
82.3% |
| Weekday |
21.9% |
17.1% |
|
17.7% |
| Type of Admission |
|
|
0.806 |
|
| Non-elective |
93.3% |
93.6% |
|
95.1% |
| Elective |
6.7% |
6.4% |
|
4.9% |
| Primary Payer |
|
|
<0.001 |
|
| Medicare |
49.7% |
61.6% |
|
59.2% |
| Medicaid |
8.4% |
4.3% |
|
8.5% |
| Private including HMO |
30.7% |
30.9% |
|
26.3% |
| Self-pay/no charge/other |
11.1% |
3.2% |
|
32.3% |
| Hospital characteristics |
|
|
|
|
| Median household income quartile as per patients’ zip codex |
<0.001 |
|
|
| 0-25th |
31.5% |
23.5% |
|
20.2% |
| 26-50th |
31.4% |
24.6% |
|
21.4% |
| 51-75th |
20.3% |
28.6% |
|
28.6% |
| 76-100th |
16.9% |
23.3% |
|
29.7% |
| Hospital bed size¥ |
|
|
0.004 |
|
| Small |
5.0% |
8.5% |
|
10.6% |
| Medium |
22.3% |
17.9% |
|
14.3% |
| Large |
72.7% |
73.6% |
|
75.1% |
| Hospital location/teaching status |
|
|
0.010 |
|
| Rural |
4.5% |
* |
|
* |
| Urban non-teaching |
30.4% |
26.5% |
|
27.1% |
| Urban teaching |
65.1% |
71.4% |
|
70.5% |
| Hospital region |
|
|
0.006 |
|
| Northeast |
15.2% |
14.9% |
|
16.5% |
| Midwest |
14.2% |
18.0% |
|
15.3% |
| South |
49.6% |
41.3% |
|
46.5% |
| West |
21.0% |
25.7% |
|
21.7% |
P<0.05 (bold) indicates statistical significance.
After propensity-matching, a remarkably higher number of comorbidities in the AMICS AF+ group disappeared, but the proportion of patients with hypertension (61.9% vs 54.9%, p=0.042) and fluid electrolyte disorder (55.75 vs 47.8%, p = 0.025) remained higher in patients with AF. The proportion of patients with coagulopathy was significantly lower in the AF+ group after propensity matching (20.1% vs 28.6%, p = 0.004). Cardiovascular comorbidities were similar in both groups following propensity matching [Table 2].
Table 2. Comorbidities in Study Population Undergoing Coronary Revascularization with PVAD (Impella®) Support Following Acute Myocardial Infarction Complicated by Cardiogenic Shock With vs. Without Atrial Fibrillation
| Comorbidities (%) |
Before Matching (weighted N=2327) |
After Matching (weighted N=840) |
|
No AF (n=1858) |
AF (n=468) |
P |
No AF (n=420) |
AF (n=420) |
P |
| Alcohol abuse |
2.4% |
3.2% |
0.342 |
* |
3.5% |
0.307 |
| Deficiency anemias |
15.7% |
33.7% |
<0.001 |
29.6% |
28.2% |
0.663 |
| Congestive heart failure |
3.2% |
4.3% |
0.241 |
* |
4.8% |
0.062 |
| Chronic pulmonary disease |
17.2% |
15.9% |
0.484 |
13.1% |
17.7% |
0.067 |
| Coagulopathy |
23.4% |
22.2% |
0.574 |
28.6% |
20.0% |
0.004 |
| Diabetes, uncomplicated |
30.4% |
29.9% |
0.823 |
32.2% |
28.6% |
0.270 |
| Diabetes with chronic complications |
5.6% |
8.5% |
0.018 |
5.9% |
7.2% |
0.480 |
| Dyslipidemia |
43.5% |
47.9% |
0.085 |
50.2% |
46.5% |
0.284 |
| Drug abuse |
3.2% |
3.2% |
0.973 |
6.1% |
3.5% |
0.079 |
| Hypertension |
58.6% |
63.8% |
0.040 |
54.9% |
61.9% |
0.042 |
| Fluid and electrolyte disorders |
53.5% |
56.1% |
0.307 |
47.8% |
55.7% |
0.025 |
| Smoking |
35.9% |
32.0% |
0.119 |
34.5% |
33.4% |
0.734 |
| Obesity |
15.3% |
16.0% |
0.016 |
15.7% |
14.4% |
0.572 |
| Obstructive sleep apnea |
3.8% |
4.3% |
0.612 |
7.2% |
4.8% |
0.145 |
| Peripheral vascular disorders |
15.3% |
16.0% |
0.724 |
16.6% |
14.3% |
0.348 |
| Renal failure |
23.3% |
34.2% |
<0.001 |
24.8% |
30.0% |
0.095 |
| Hemodialysis status |
8.8% |
15.0% |
<0.001 |
14.1% |
10.8% |
0.142 |
| Previous MI/PCI/CABG |
17.8% |
19.1% |
0.527 |
20.4% |
16.5% |
0.135 |
| Multivessel PCI |
27.5% |
36.2% |
<0.001 |
28.3% |
31.0% |
0.393 |
| Prior TIA/Stroke |
3.0% |
4.3% |
0.151 |
4.8% |
3.6% |
0.392 |
| Pacemaker status |
1.1% |
3.2% |
0.001 |
* |
* |
* |
| ICD status |
2.2% |
3.2% |
0.181 |
* |
3.6% |
0.307 |
| Ventricular tachyarrhythmias |
42.3% |
39.5% |
0.262 |
40.3% |
40.6% |
0.921 |
P<0.05 (bold) indicates statistical significance, AF= Atrial Fibrillation, PVAD=Percutaneous Ventricular Assist Device, MI=Myocardial Infarction, PCI=Percutaneous Coronary Intervention, CABG=Coronary Artery Bypass Grafting, TIA=Transient Ischemic Attack, ICD=Implantable Cardioverter-defibrillator.
No difference seen in all-cause in-hospital mortality in AF+ vs AF- (40.5% vs 36.7%, p=0.245) patients [Table 3]. Additionally, no dissimilarities were noted between the groups in the rates of post-procedural hemorrhages, infection, AKI requiring dialysis, and septic shock in AF in the matched cohort. Interestingly, in-hospital cardiac arrest was remarkably higher in the AF- group (32% vs 19.2%, p<0.001). Postoperative respiratory complications were seen more often in AF+ patients (9.5% vs 4.8%, p = 0.007). Indices of healthcare/economic burden were universally higher in the AF+ group, as assessed by less number of routine discharges (13.1% vs 30.2%), the higher number of transfers to other facilities (27.3% vs 17.8%), greater utilization of home health care (14.3 % vs 7.1%) (p<0.001), a greater mean length of stay (11.9 ± 10.1 vs 9.11 ± 6.8 days, p <0.001) and greater hospital charges ($308,478 vs. 277,982, p=0.008) as compared to the AF- cohort.
Table 3. Outcomes and Complications of Coronary Revascularization with PVAD (Impella®) Support Following Acute Myocardial Infarction Complicated by Cardiogenic Shock With vs. Without Atrial Fibrillation (After Propensity-score Matching)
| Complications |
1:1 Propensity-Matched Cohorts |
P |
| All-cause in-hospital mortality |
154 (36.7%) |
170 (40.5%) |
0.245 |
| Hemorrhage or hematoma complicating a procedure |
30 (7.1%) |
20 (4.8%) |
0.147 |
| In-hospital cardiac arrest |
245 (32.0%) |
181 (19.2%) |
<0.001 |
| Perioperative stroke |
<11* |
40 (9.5%) |
<0.001 |
| Postoperative respiratory complications |
20 (4.8%) |
40 (9.5%) |
0.007 |
| Postoperative AKI requiring dialysis |
24 (5.8%) |
20 (4.7%) |
0.536 |
| Postoperative infection |
65 (15.5%) |
75 (17.9%) |
0.347 |
| Septic shock |
25 (6.0%) |
35 (8.4%) |
0.177 |
| Disposition |
|
|
<0.001 |
| Routine |
133 (30.2%) |
75 (13.1%) |
|
| Transfer to short term hospital |
54 (8.3%) |
45 (4.7%) |
|
| Other transfers including SNF, ICF & others |
164 (17.8%) |
173 (27.3%) |
|
| Home health care |
44 (7.1%) |
91 (14.3%) |
|
| Mean Length of stay Mean (±SD) |
9.1 (± 6.8) days |
11.9 (± 10.1) days |
<0.001 |
| Mean hospital charges |
$277,982 |
$308,478 |
0.008 |
P-values <0.05 (bold) indicates clinical significance, AF= Atrial Fibrillation, pVAD=Percutaneous Ventricular Assist Device, AF=Atrial Fibrillation, AKI=Acute Kidney Injury, SNF=Skilled Nursing Facility, ICF=Intermediate Care Facility
To the best of our knowledge, this is the first study to analyze the impact of a concomitant AF on the outcomes of AMICS patients undergoing PCI with pVAD support as assessed with a large national inpatient database. Importantly, there was no difference in all-cause in-hospital mortality in the AF+ and AF- cohorts. While a cardiac arrest was significantly more common in AF- patients, AF+ patients were more likely to experience postoperative respiratory complications. Finally, the economic burden of disease was significantly higher when AF was present, as demonstrated by an increased prevalence of non-routine disposition, increased utilization of home health care services, greater LOS, and increased hospital charges.
AF has traditionally been associated with higher morbidity and mortality in the general population, which is generally attributed to the higher prevalence of comorbid conditions and advanced age in these patients [15], [16]. In keeping with prior studies, AF+ patients tended to be older and male in our analysis [17]. Prior studies have suggested that patients with AMI who were hemodynamically stable and developed AF had greater morbidity and mortality [3], [4] which persisted even after adjustment for baseline characteristics. In the same vein, Sakata et al showed patients with new-onset AF in the setting of AMI had higher mortality [18], and Crenshaw et al also demonstrated an independent effect of AF on 30-day mortality in post-MI patients [19]. The presence of AF in the setting of acute ventricular dysfunction may further exacerbate these patients’ hemodynamic compromise, accounting for the worse outcome of these patients. While the literature remains contentious regarding AF being an independent predictor of AMI-related mortality, it is notable that the presence of AF was not associated with an increase in all-cause in-hospital mortality in our study population [2], [20], [21].
Clinical outcomes in patients with AMICS may be influenced by multiple factors, including a deranged systemic inflammatory response, and treatment success is not merely dependent on the restoration of cardiac output [22]. In our study, we found no statistical difference in in-hospital all-cause mortality between AF+ and AF- patients with AMICS undergoing PCI with pVAD support, in keeping with a recent sub-study of the IABP-SHOCK trial that investigated the impact of AF on AMICS [23]. In the sub-study, the presence of AF was not associated with early or late mortality in patients with AMICS. Furthermore, there was no increase in mortality in patients who presented with AF or developed AF in-hospital as compared to patients without AF. Also noted were similar occurrences of non-fatal events and functional outcomes in both groups. Interestingly, these investigators also observed that AF loses its prognostic significance with increasing severity of sickness [23]. Hickey et al studied the impact of AF on patients with end-stage heart failure requiring LVAD support and found that the presence of AF was not associated with increased mortality [24]. Similarly, Stulak et al observed that AF did not portend higher mortality in LVAD use in heart failure [25]. Our findings are in keeping with these studies.
Surprisingly, the rate of cardiac arrest was higher in AF- patients in our study cohort. Several hypotheses may be advanced to explain this observation. It is possible that this observed difference can be due to the higher use of anti-arrhythmic and anticoagulant medications in AF+ patients, thereby affording a protective effect on the AF+ group. However, owing to the retrospective nature of this analysis and a lack of information regarding medication use, we cannot further explore this possibility.
From a health economics perspective, the presence of AF resulted in significantly greater indices of economic burden and health care resource consumption, as demonstrated by a greater length of stay, higher total hospitalization costs, and a higher acuity of discharge disposition in the AF+ group. One possible explanation for this finding is the higher incidence of respiratory complications noted in the AF+ group, which may have led to increased use of long periods on mechanical ventilation.
The present study should be interpreted within the context of numerous limitations. The NIS is a retrospective database, so we cannot reliably comprehend the time duration and type of AF occurrence with a clear differentiation between a new-onset AF and a pre-existing AF. Information regarding medication use, laboratory studies, and other clinical variables are unavailable, a limitation inherent to NIS data. Propensity matched analysis was done to minimize selection bias. Despite these measures, it is impossible to control for all potential confounders and residual imbalances between the groups. Long-term outcomes cannot be evaluated using the NIS databases due to a lack of follow-up data. As there are no separate ICD-9 codes available to differentiate between Impella® and Tandem heart®, the results of our analysis cannot be individualized towards any single PVAD device. However, since 2013 most of the growth in PVAD use has occurred with the use of Impella® especially in AMI [26] and the use of Tandem heart is in semi-urgent situations as this involved Tran septal puncture. Despite these limitations, a large sample size and comprehensive propensity-score matching enabled us to perform this unique analysis.
In conclusion, the presence of a concomitant diagnosis of AF in the setting of AMI complicated by cardiogenic shock in patients undergoing PCI with the use of pVAD is not associated with an increase in all-cause mortality. Importantly, the presence of AF was associated with a greater economic burden of disease, as demonstrated by significantly higher hospitalization costs, greater LOS, and a greater proportion of non-routine discharges, possibly owing to the higher rate of respiratory complications observed in this group.
- Kannel WB, Wolf PA, Benjamin EJ, Levy D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol 1998;82:2n-9n.
- Sugiura T, Iwasaka T, Ogawa A, Shiroyama Y, Tsuji H, Onoyama H, Inada M. Atrial fibrillation in acute myocardial infarction. Am J Cardiol 1985;56:27-29.
- Schmitt J, Duray G, Gersh BJ, Hohnloser SH. Atrial fibrillation in acute myocardial infarction: a systematic review of the incidence, clinical features and prognostic implications. Eur Heart J 2009;30:1038-104510.1093/eurheartj/ehn579.
- Jabre P, Roger VL, Murad MH, Chamberlain AM, Prokop L, Adnet F, Jouven X. Mortality associated with atrial fibrillation in patients with myocardial infarction: a systematic review and meta-analysis. Circulation 2011;123:1587-159310.1161/circulationaha.110.986661.
- Bang CN, Gislason GH, Greve AM, Bang CA, Lilja A, Torp-Pedersen C, Andersen PK, Kober L, Devereux RB, Wachtell K. New-onset atrial fibrillation is associated with cardiovascular events leading to death in a first time myocardial infarction population of 89,703 patients with long-term follow-up: a nationwide study. J Am Heart Assoc 2014;3:e00038210.1161/jaha.113.000382.
- Sutton NR, Seth M, Ruwende C, Gurm HS. Outcomes of Patients With Atrial Fibrillation Undergoing Percutaneous Coronary Intervention. J Am Coll Cardiol 2016;68:895-90410.1016/j.jacc.2016.05.085.
- Flaherty MP, Khan AR, O'Neill WW. Early Initiation of Impella in Acute Myocardial Infarction Complicated by Cardiogenic Shock Improves Survival: A Meta-Analysis. JACC Cardiovasc Interv 2017;10:1805-180610.1016/j.jcin.2017.06.027.
- Batsides G, Massaro J, Cheung A, Soltesz E, Ramzy D, Anderson MB. Outcomes of Impella 5.0 in Cardiogenic Shock: A Systematic Review and Meta-analysis. Innovations (Phila) 2018;13:254-26010.1097/imi.0000000000000535.
- O'Neill WW, Schreiber T, Wohns DH, Rihal C, Naidu SS, Civitello AB, Dixon SR, Massaro JM, Maini B, Ohman EM. The current use of Impella 2.5 in acute myocardial infarction complicated by cardiogenic shock: results from the USpella Registry. J Interv Cardiol 2014;27:1-1110.1111/joic.12080.
- Griffith BP, Anderson MB, Samuels LE, Pae WE, Jr., Naka Y, Frazier OH. The RECOVER I: a multicenter prospective study of Impella 5.0/LD for postcardiotomy circulatory support. J Thorac Cardiovasc Surg 2013;145:548-55410.1016/j.jtcvs.2012.01.067.
- Lemaire A, Anderson MB, Lee LY, Scholz P, Prendergast T, Goodman A, Lozano AM, Spotnitz A, Batsides G. The Impella device for acute mechanical circulatory support in patients in cardiogenic shock. Ann Thorac Surg 2014;97:133-13810.1016/j.athoracsur.2013.07.053.
- Databases H. HCUP Databases. Healthcare Cost and Utilization Project (HCUP). August 2018. Agency for Healthcare Research and Quality, Rockville, MD. www.hcup-us.ahrq.gov/nisoverview.jsp. Accessed March 2019.
- Desai R, Patel U, Sharma S, Amin P, Bhuva R, Patel MS, Sharma N, Shah M, Patel S, Savani S, Batra N, Kumar G. Recreational Marijuana Use and Acute Myocardial Infarction: Insights from Nationwide Inpatient Sample in the United States. Cureus 2017;9:e181610.7759/cureus.1816.
- Desai R, Singh S, Fong HK, Goyal H, Gupta S, Zalavadia D, Doshi R, Savani S, Pancholy S, Sachdeva R, Kumar G. Racial and sex disparities in resource utilization and outcomes of multi-vessel percutaneous coronary interventions (a 5-year nationwide evaluation in the United States). Cardiovascular Diagnosis and Therapy 2018;9:18-29.
- Go AS, Hylek EM, Phillips KA, Chang Y, Henault LE, Selby JV, Singer DE. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. Jama 2001;285:2370-2375.
- Benjamin EJ, Wolf PA, D'Agostino RB, Silbershatz H, Kannel WB, Levy D. Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation 1998;98:946-952.
- Rathore SS, Berger AK, Weinfurt KP, Schulman KA, Oetgen WJ, Gersh BJ, Solomon AJ. Acute myocardial infarction complicated by atrial fibrillation in the elderly: prevalence and outcomes. Circulation 2000;101:969-974.
- Sakata K, Kurihara H, Iwamori K, Maki A, Yoshino H, Yanagisawa A, Ishikawa K. Clinical and prognostic significance of atrial fibrillation in acute myocardial infarction. Am J Cardiol 1997;80:1522-1527.
- Crenshaw BS, Ward SR, Granger CB, Stebbins AL, Topol EJ, Califf RM. Atrial fibrillation in the setting of acute myocardial infarction: the GUSTO-I experience. Global Utilization of Streptokinase and TPA for Occluded Coronary Arteries. J Am Coll Cardiol 1997;30:406-413.
- Behar S, Zahavi Z, Goldbourt U, Reicher-Reiss H. Long-term prognosis of patients with paroxysmal atrial fibrillation complicating acute myocardial infarction. SPRINT Study Group. Eur Heart J 1992;13:45-50.
- Lehto M, Snapinn S, Dickstein K, Swedberg K, Nieminen MS. Prognostic risk of atrial fibrillation in acute myocardial infarction complicated by left ventricular dysfunction: the OPTIMAAL experience. Eur Heart J 2005;26:350-35610.1093/eurheartj/ehi064.
- Thiele H, Zeymer U, Neumann FJ, Ferenc M, Olbrich HG, Hausleiter J, de Waha A, Richardt G, Hennersdorf M, Empen K, Fuernau G, Desch S, Eitel I, Hambrecht R, Lauer B, Bohm M, Ebelt H, Schneider S, Werdan K, Schuler G. Intra-aortic balloon counterpulsation in acute myocardial infarction complicated by cardiogenic shock (IABP-SHOCK II): final 12 month results of a randomised, open-label trial. Lancet 2013;382:1638-164510.1016/s0140-6736(13)61783-3.
- de Waha S, Schoene K, Fuernau G, Desch S, Eitel I, Poss J, Meyer-Saraei R, Eitel C, Tilz R, Schuler G, Werdan K, Schneider S, Ouarrak T, Zeymer U, Thiele H. Prognostic impact of atrial fibrillation in cardiogenic shock complicating acute myocardial infarction: a substudy of the IABP-SHOCK II trial. Clin Res Cardiol 2018;107:233-24010.1007/s00392-017-1175-1.
- Hickey KT, Garan H, Mancini DM, Colombo PC, Naka Y, Sciacca RR, Abrams MP, Solove M, Zeoli N, Flannery M, Garan AR, Biviano AB. Atrial Fibrillation in Patients With Left Ventricular Assist Devices: Incidence, Predictors, and Clinical Outcomes. JACC Clin Electrophysiol 2016;2:793-79810.1016/j.jacep.2016.03.009.
- Stulak JM, Deo S, Schirger J, Aaronson KD, Park SJ, Joyce LD, Daly RC, Pagani FD. Preoperative atrial fibrillation increases risk of thromboembolic events after left ventricular assist device implantation. Ann Thorac Surg 2013;96:2161-216710.1016/j.athoracsur.2013.07.004.
- Abiomed website; Aug 2. 2013 Abiomed Surpasses 15,000 Impella Patients in the United States: New Impella CP Reaches 1,000th Global Patient Milestone. press releasehttp://investors.abiomed.com/releasedetail.cfm?ReleaseID=782468 [Accessed December 30, 2018].