Assessment of DNA Damage After Ionizing Radiation Exposure in Patients Undergoing Cardiac Resynchronization Therapy Device Implantation or Atrial Fibrillation Ablation (The RADAR Study)
Mohit K. Turagam1, Venkat Vuddanda2, Donita Atkins3, Rakesh Venkata4, Bhavya Yarlagadda4, Himabindu Korra4, Jaya Pitchika4, Sudharani Bommana3, Dhanujaya R. Lakkireddy3
1Icahn School of Medicine at Mount Sinai, New York, NY.2Kansas City Heart Rhythm Institute and Research Foundation, Overland Park, KS.3Harvard Medical School, Boston, MA.4Division of Cardiovascular Diseases, Cardiovascular Research Institute, University of Kansas Hospital and Medical Center, Kansas City, KS.
There is limited data regarding effect of prolonged radiation exposure during electrophysiological (EP) procedures on direct DNA damage. Comet test has shown to assess DNA damage following radiation exposure.
We performed a single-center prospective observational study assessing direct DNA damage using the quantitative comet assay in patients undergoing cardiac resynchronization (CRT) and atrial fibrillation (AF) catheter ablation procedures. Venous comet assay was performed pre, immediately post procedure and at 3-month duration in twenty-two (N=22) patients who underwent catheter ablation for symptomatic AF and fourteen (N=14) patients who underwent CRT implantation.
The median [interquartile range (IQR)] fluoroscopy time, radiation dose and dose area product (DAP) were 34.3 (27.97 – 45.48) minutes, 853.07 (611.36 - 1334.76) mGy and 16,994.10 (9,023.65 – 58,845.00) UGym2 in the ablation group and 30.05 (18.75 - 37.33) minutes, 345.00 (165.09 - 924.79) mGy and 11,837.20 [7182.67 - 35567.75] UGym2 in the CRT group. When compared with pre-procedure, there was a statistically significant increase in median (IQR) DNA migration on comet assay in the ablation group immediately post procedure [+6.55 µm (0.78, 10.25, p=0.02)] that subsequently decreased at 3 months [-1.00 µm (-2.20, 0.78), p=0.03] but not in the CRT group.
There was a significant increase in DNA damage as detected by comet assay immediately post procedure that normalized at 3 months in patients undergoing AF ablation. Further large prospective studies are warranted to evaluate the impact of this prolonged radiation exposure and DNA damage on long-term follow up.
Key Words : Comet Assay, DNA Damage, Radiation Exposure, Cardiac Resynchronization Therapy, Catheter Ablation, Atrial Fibrillation.
Correspondence to: Dhanunjaya Lakkireddy,
The Kansas City Heart Rhythm Institute (KCHRI) @ HCA MidWest 12200, W 106th street, Overland Park Regional Medical Center
Overland Park, KS 66215
Electrophysiology (EP) procedures such as cardiac resynchronization therapy (CRT) and radiofrequency catheter ablation (RFA) have undergone significant expansion in the last decade and are widely performed for the management of congestive heart failure and atrial fibrillation (AF). Despite, the advent of electroanatomical mapping and 3-D technology both CRT and AF ablation are associated with prolonged procedural and fluoroscopy times, exposing patients and operators to substantial amounts of radiation[1,2]. The estimated effective radiation dose with AF ablation ranges from 15 - 100 milliSievert (mSv) and 2 – 95 mSv with CRT implantation which is equivalent to the radiation dose of 50 – 5000 chest radiographs[3]. Furthermore, cumulative radiation exposure was associated with increased risk of all-cause cancer and mortality in both patient and operators[4,5].
These radiation effects are due to direct and indirect DNA damage by formation of free radicals[6] and through various mechanisms including direct DNA breaks, breaks in cross bridges between DNA and proteins and denaturation of proteins, purines and pyrimidines bases[7]. It is estimated that each Gray unit (1 gray = 1000 mSv) of radiation exposure can cause approximately 20 DNA-DNA cross linkages, 40 double strand breaks, 150 DNA-protein cross links, 160-320 non-double stranded breaks clustered DNA damage, 1000 single strand breaks and >1000 DNA base damage per cell [1-1]. Total accumulated dose of radiation exposure has been associated with chromosomal aberrations due to defective DNA repair and increased risk of carcinogenesis and mutagenesis [7,10].
The blood comet assay is a single-cell gel electrophoresis technique that has emerged as a standard method for assessing direct DNA damage in circulation eukaryotic cells (lymphocytes and monocytes) in subjects exposed to radiation by various cytogenetic, biotechnological and epidemiological studies[11-16]. In this current era of aging population undergoing multiple EP procedures, there is limited data regarding the extent of direct DNA damage from prolonged radiation exposure. The purpose of this study was to estimate the extent of direct DNA damage in patients undergoing two commonly performed procedures (CRT implantation and AF ablation) that requires substantial amount of radiation by using the blood comet assay.
This is a single center, prospective sequential observational study to detect DNA damage occurring from prolonged radiation exposure in patients undergoing AF ablation and CRT implantation by using a blood comet assay. The study included 22 consecutive patients undergoing AF ablation and 14 consecutive patients undergoing CRT implantation. Patient who gave informed consent were enrolled in the study. The study was approved by the local Institutional Review Board. Complete medical history, medications and demographics were extracted from review of electronic medical records.
The study inclusion criteria were age ≥18 years and patients undergoing CRT implantation or AF ablation for standard indications. Exclusion criteria were subjects with any prior radiation therapy, history of cancer, active infection, prior chemotherapy, prior electrophysiological study or cardiac catheterization or CT scans in the last year. Patients with a history of occupational exposure to radiation were also excluded.
The primary end point was assessment of direct DNA damage estimated by distance of DNA migration on the alkaline comet assay immediately post-procedure and at 3-month duration compared with pre-procedure. The extent of DNA damage was assessed by distance of DNA migration (total length of the comet - diameter of the nucleus) on the comet assay using fluorescence microscopy. All patients were followed in clinic at 2 weeks and 3 months’ duration.
Atrial Fibrillation Ablation
All patients underwent standard catheter ablation procedure for symptomatic drug refractory AF (both paroxysmal and persistent). Briefly, the procedure was performed under general anesthesia using femoral vein access. The procedure required frequent repositioning of the camera between AP (anterior-posterior), LAO (left anterior oblique) and RAO (right anterior oblique) views to locate catheter placement and obtain transeptal access. Two transeptal punctures were made into the left atrium using intracardiac echocardiography, fluoroscopy and electromagnetic mapping system such as CARTO (Biosense Webster Inc.) or the NavX mapping system (St. Jude Medical). A standard Lasso/Pentaray (Biosense Webster Inc., Diamond Bar, California) or Spiral (St. Jude Medical, Minneapolis, Minnesota) catheters were used for mapping the left atrium. A 3.5 mm. open tip- irrigated ablation catheters (ThermoCool, Biosense Webster Inc; Tacticath, St.Jude Medical) was used for ablation. A 3-dimensional geometry of the left atrium was reconstructed using the mapping system. All patients underwent antral pulmonary vein isolation. Additional ablation of the cavotricuspid isthmus, roof line, posterior wall, mitral isthmus line and left atrial appendage ablation was at the discretion of the operator.
Cardiac Resynchronization Therapy
All patients underwent either a denovo CRT implantation or an upgrade for standard indications. The procedure technique was similar in all patients. Briefly, the procedure was performed with moderate sedation and local anesthesia. In patients undergoing denovo CRT – the cephalic vein cut down was performed in all patients for insertion of the right ventricular and right atrial lead while the axillary or subclavian was accessed with a micro puncture needle for implantation of the coronary sinus (CS) lead. A CS venography was performed just prior to cannulation with the CS lead which was inserted by over the wire technique. The procedure was performed under fluoroscopy. Majority of the procedure require AP (anterior-posterior) views but LAO (left anterior oblique) and RAO (right anterior oblique) were frequently used for CS cannulation based on operator preference.
A 10 milliliter (ml.) venous blood sample was collected 30 – 60 minutes prior to starting of the EP procedure. The subjects served as their own controls and another 10-ml. blood sample was collected immediately at the conclusion of the procedure and at 3 months follow up. The blood samples were processed in the epigenetics laboratory and blood comet assays were performed on all collected samples.
The technique of alkaline comet assay has been described in great detail previously[13,17]. This technique of assessing DNA damage to cells has been used previously in personnel who were exposed to occupational radiation[16,11]. Venous blood sample has been selected for use in our study since, the average procedure times for EP procedures with conventional modalities is about 142 ± 55.8 minutes and the fluoroscopic time is about 21.2 ± 4.8 minutes. This time would be sufficient to cause irradiation of all the circulating blood cells in the body. The procedure consists of preparation of the buffy coat from the peripheral blood sample. The blood sample is spun at 1500 rpm for 30 minutes and the mononuclear lymphocytes are aspirated from the junction of plasma and lymphocyte preparation media. A microscopic slide was then layered with normal melting point agar (NMPA) and allowed to cool down. In a separate centrifuge tube, the lymphocytes were mixed with NMPA. This mixture was then layered over the agar based glass. A third layer of NMPA was added over the lymphocyte agar layer. The cells were then lysed using a lysis solution. Once the cells lysed, the slide was then run through gel electrophoresis system for 30 minutes. Electrophoresis was performed at 25 V and 300 mA for 20 minutes following which the slides were neutralized with 0.4 M Tris (pH 7.5) and stained with 50 μL of ethidium bromide (20 μg/mL). Slide analysis was performed using a fluorescence microscopy (Olympus. 40x objective lens).
A comet analysis software [CometScore Pro (Tritek Corp, VA)] was used to estimate head diameter (nucleus), comet length and tail length. A total of 100 randomly captured comets from each individual slide were studied and the average of these 100 comets for comet tail length, tail movement, comet head diameter and length of comet were collected. Despite, several techniques of estimating DNA damage have been previously described including – tail length, head optical intensity, tail distribution moment etc, the extent of DNA damage in this study was assessed by distance of DNA migration (total length of the comet - diameter of the nucleus) on the comet assay using fluorescence microscopy. This technique is a widely accepted method of measurement[18].
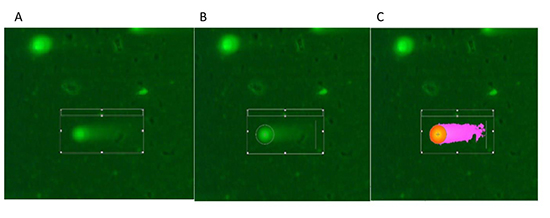
[Figure 1 (A,B,C)] demonstrates pre-procedural and post procedural comet assay and how the comet length analysis was performed. Before, after radiation exposure and at 3-month duration the comet parameters were compared. The entire procedure of preparing the comet assay slides and reading was performed at the Center for Epigenetics and Stem Cell Biology at The University of Kansas. To ensure uniformity, the available standardized protocol for blood comet assay was adapted.
Figure 1(A,B,C). Figure 1A, 1B, 1C: Fluorescence microscopy images and measurement of comet assay migration using a comet analysis software. A) The automated comet analysis software first randomly identifies a comet B) then estimates the comet head/nucleus and C) then separates the nucleus (red/orange) and the tail (pink) to calculate the comet length which is the difference between the whole comet length and the diameter of the nucleus
Categorical data were represented as counts (percentage) and continuous data as either mean (standard deviation) or median [interquartile range], if assumptions of normality were not met. We compared the baseline characteristics of patients who underwent AF ablation with CRT implantation. Proportions were compared using Chi-square test. Group means were compared using Welch two sample t test, or Fisher exact test as appropriate. For non-normally distributed data, hypothesis testing was performed using Kruskal-Wallis rank sum test. Standardized mean differences between the groups were reported for all the baseline co variates. We calculated the change in comet assay measurement immediately post procedure and at three months follow up from the baseline, for each patient and reported summary statistics as median [IQR] for each cohort (AF ablation & CRT implantation). Hypothesis testing was performed using Wilcoxon signed rank test for paired samples. We used “lme4” package in R to create a linear mixed effects model to investigate within subject variance (Supplementary [Table S1],[Figure S1]). We performed a pairwise correlation plot to examine the relationship between post procedure change in comet assay, age, BMI, procedure time, fluoroscopy time, radiation dose, and DAP to visualize and investigate significant correlations in the entire cohort. We performed linear regression to identify significant predictors of post procedure change in comet assay value. All statistical analyses were performed on mac OS Sierra (version 10.12.6) using R statistical computing program (version 3.3.2; Vienna, Austria).
Table S1. Linear mixed model fit by REML ['lmerMod'];
|
|
|
|
| Formula: Comet Assay ~ TIME + PROCEDURE + (1 | PTID); |
; Data: radarData_long
REML criterion at convergence: 871.6; |
Number of obs: 108, groups: SNO, 36 |
|
| Random effects: |
|
|
|
| Groups |
Variance |
Std.Dev. |
|
| PTID |
183.9 |
13.56 |
|
| Residual |
123.8 |
11.13 |
|
| Fixed effects: |
Estimate |
Std.Error |
t value |
| (Intercept) |
24.7642 |
2.5651 |
9.654 |
| TIME1 |
-0.6874 |
1.5141 |
-0.454 |
| TIME2 |
5.7587 |
1.5141 |
3.803 |
| PROCEDURE1 |
-8.1068 |
2.5651 |
-3.16 |
| Correlation of Fixed effects: |
(Intr) |
TIME1 |
TIME2 |
| TIME1 |
0.0000 |
|
|
| TIME2 |
0.0000 |
-0.5000 |
|
| PROCEDURE1 |
-0.2220 |
0.0000 |
0.0000 |
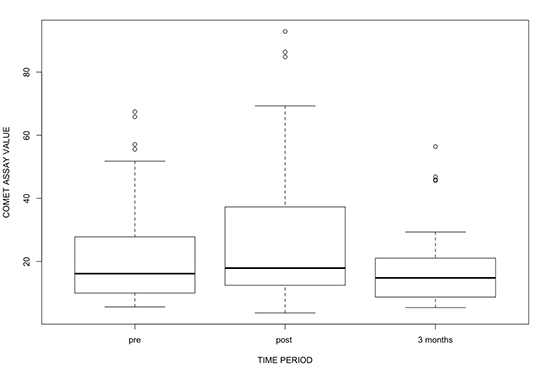
Figure S1. Box plots demonstrating distribution of comet assay values pre-procedure, post procedure and at 3 months follow up.
Baseline and Procedural Characteristics
AF ablation group included patients with a mean age of 65.09±9.15 years with a BMI of 31.7±9.4. CRT group included patients with a mean age of 67.8±11.8 years and a BMI of 31±3.9. Patients underwent denovo CRT in 66% and upgrade in 33% of patients. [Table 1] demonstrates the baseline characteristics of patients in both the groups. The median (IQR) fluoroscopy time in AF ablation and CRT group were 34.30 [27.97, 45.48] and 30.05 [18.75, 37.33] minutes respectively.
[Table 2] demonstrates the comet tail migration measurements before the EP procedure, after and at 3-month duration. Pre-procedural, immediately post – procedure and 3 - month median (IQR) DNA migration in the AF ablation group was 12.55 [8.85, 20.38] µm, 16.70 [12.22, 25.85] and 12.90 [7.25, 19.27] while in the CRT group was 20.70 [13.85, 54.62] µm, 29.66 [13.95, 67.08] and 19.15 [11.38, 39.58] respectively. In the AF ablation group, there was a significant increase (6.55 [0.78, 10.25] µm, p=0.02) in the median comet tail migration measurements pre-procedure vs. immediately post-procedure. Furthermore, the comet migration distance decreased but remained statistically significant (-1.00 [-2.20, 0.78] µm, p=0.03) at 3-month duration when compared with pre-procedure.
In the CRT group, there was a substantial increase (2.43 [- 1.90, 7.11] µm, p=0.17) in median (IQR) comet tail migration immediately post-procedure vs. preprocedural which was not statistically significant. The post procedural comet migration decreased (- 0.70 [- 4.39, 0.62] µm, p=0.17) at 3-months.
Table 1. Baseline demographics and procedural characteristics of patients in the study. SMD – Standard mean difference, AF – atrial fibrillation, CRT – cardiac resynchronization therapy, IQR – interquartile range.
| Baseline Characteristics |
AF Ablation (n = 22) |
CRT(n = 14) |
p value |
SMD |
| Age (Years) (mean [sd]) |
65.09 (9.15) |
67.86 (11.86) |
0.436 |
0.26 |
| Female (%) |
6 (27.2) |
4 (28.5) |
0.8 |
0.21 |
| Caucasian (%) |
20 (90.9) |
9 (64.3) |
0.125 |
0.67 |
| Body mass index (mean [sd]) |
31.74 (9.37) |
30.16 (3.98) |
0.556 |
0.22 |
| Hypertension (%) |
15 (68.2) |
10 (71.4) |
1 |
0.07 |
| Diabetes (%) |
6 (27.3) |
3 (21.4) |
1 |
0.14 |
| Coronary disease (%) |
7 (31.8) |
11 (78.6) |
0.017 |
1.07 |
| Myocardial infarction (%) |
3 (13.6) |
3 (21.4) |
0.878 |
0.21 |
| Coronary bypass surgery (%) |
3 (13.6) |
3 (21.4) |
0.878 |
0.21 |
| Chronic kidney disease (%) |
3 (13.6) |
3 (21.4) |
0.878 |
0.21 |
| Heart failure (%) |
10 (45.5) |
7 (50.0) |
1 |
0.09 |
| Cardiomyopathy (%) |
|
|
0.51 |
0.40 |
| Ischemic |
3 (13.6) |
4 (28.6) |
|
|
| Non-ischemic |
7 (31.8) |
3 (21.4) |
|
|
| Sleep apnea (%) |
8 (36.4) |
5 (35.7) |
1 |
0.01 |
| Chronic obstructive pulmonary disease (%) |
3 (13.6) |
5 (35.7) |
0.253 |
0.53 |
| Cerebrovascular accident (%) |
1 (4.5) |
0 (0.0) |
1 |
0.31 |
| History of Alcohol (%) |
12 (57.1) |
5 (35.7) |
0.369 |
0.44 |
| History of smoking (%) |
13 (59.1) |
6 (42.9) |
0.543 |
0.33 |
| Procedure time (minutes) (median [IQR]) |
152.00 [130.00, 200.00] |
148.50 [125.75, 191.50] |
0.849 |
0.17 |
| Fluoroscopy time (minutes) (median [IQR]) |
34.30 [27.97, 45.48] |
30.05 [18.75, 37.33] |
0.375 |
0.39 |
| Radiation dose(mGy) (median [IQR]) |
853.07 [611.36, 1334.76] |
345.00 [165.09, 924.79] |
0.095 |
0.92 |
| Dose – Area Product (UGym2) (median [IQR]) |
16994.10 [9023.65, 58845.00] |
11837.20 [7182.67, 35567.75] |
0.524 |
0.28 |
Table 2. Change in comet assay post – procedure and 3 months compared with pre – procedure. AF – atrial fibrillation, CRT – cardiac resynchronization therapy, IQR – interquartile range.
| Procedure |
Change in Comet Assay |
p value |
| AF ablation (n = 22) |
|
|
| (Immediate post procedure – pre-procedure) |
6.55 [0.78, 10.25] |
0.02304 |
| (3 months post procedure – pre-procedure) |
-1.00 [-2.20, 0.78] |
0.03482 |
| CRT implantation (n = 14) |
|
|
| (Immediate post procedure – pre-procedure) |
2.43 [- 1.90, 7.11] |
0.1726 |
| (3 months post procedure – pre-procedure) |
- 0.70 [- 4.39, 0.62] |
0.1726 |
We performed a pairwise correlation plot to examine the relationship between post procedure change in comet assay with other variables of interest such as age, BMI, procedure time, fluoroscopy time, radiation dose, and DAP and did not notice any significant correlation when examined in the entire cohort. Linear regression analysis using change in comet assay value as dependent variable also did not demonstrate any significant predictors such as age, BMI, smoking, alcoholism, procedure time, fluoroscopy time, radiation dose and DAP on the outcome.
In the present study, we investigated DNA damage in a group of 22 patients undergoing AF ablation and another 14 undergoing CRT implantations with no prior significant radiation exposure at a tertiary care teaching hospital. There was a significant increase in comet tail migration immediately post-procedure which normalized at 3-months duration in patients undergoing AF ablation but this change was not statistically significant in patients undergoing CRT implantation.
The comet assay is a well-established molecular technique in biomonitoring and estimation of DNA damage in patients exposed to radiation[11-13,19,20,16]. Singh et al. first demonstrated that comet assay when used under alkaline conditions, it was reported to be more sensitive for detection of alkali labile sites and assess both double- and single-stranded DNA breaks [21]. Comet assay is also one of the measuring techniques of European Standards Committee on Oxidative DNA Damage (ESCODD) and measures DNA breaks allowing fraction of DNA to migrate under electrophoresis towards the anode, forming a comet tail in which the percentage of DNA in the tail reflects the break frequency[16,22]. There are several advantages of assessment of radiation induced DNA damage using the comet assay – First, the test is highly sensitive in detection of radiation induced DNA damage with a reported detection limit of 5 cGy gamma rays in human lymphocytes. Second, the test estimates DNA damage at the cellular level (circulating lymphocyte/monocytes). Third, the test is easy to perform, cost – effective and not time consuming. The possibility that the test may have higher specificity in a subgroup of patients and type of cells remains an area of further investigation[23].
Some studies have reported elevated DNA damage among interventional cardiologists exposed to prolonged and cumulative radiation exposure, suggesting the need for adequate measures to protect and prevent health care personnel who work in areas of prolonged radiation exposure[24]. Another study reported significant DNA damage as measured by increase in comet tail length after a work day of occupational exposure among nuclear medicine and radiology workers[11]. Similarly, there is evidence regarding DNA damage reported in patients undergoing diagnostic and therapeutic radiation testing or interventions[25,26]. It is also estimated that a cumulative dose exposure of 100 mSv may additionally increase the risk of cancer of 1 in 100 patients, this dose can be reached in a patient undergoing 1-2 EP procedures[2].
However, currently there is no data regarding extent of direct DNA damage in patients undergoing EP procedures which require prolong fluoroscopy and radiation exposure. To the best of our knowledge, this is the first study investigating the effects of radiation exposure on direct DNA damage in patients undergoing EP procedures. Although, our results demonstrate that there was a substantial increase in post-procedural DNA migration in terms of absolute distance in patients undergoing both procedures which reversed at 3-month duration, these findings were only statistically significant in the AF ablation group. The limited power of 14 patients in the CRT group could be a potential explanation for the lack of statistical significance as we observed a similar trend in comet assay values as the AF ablation group. The normalization in comet assay migration at 3 months duration is likely due to process of DNA repair which includes cell cycle arrest, apoptosis etc. minimizing genomic instability [27]. Furthermore, all patients undergoing EP procedures showed large inter-individual variation in comet tail migration that was considered inherently unique to that particular patient. However, the comet tail measurement values pre-and post-procedure were within that particular range for each individual. The amount of radiation exposure observed in this series in somewhat higher, especially in an era of 3-D mapping and intracardiac echo. The reason being - (1) Some patients with persistent AF underwent extensive ablation as described, (2) Anatomical variations observed with coronary venous system and (3) majority of cases were performed by house staff including electrophysiology fellows.
A previous study showed a significant correlation between age and DNA damage as estimated by comet assay in workers with occupational exposure[20]. We performed a pairwise correlation plot to examine the relationship between post procedure change in comet assay and variables of interest such as age, BMI, smoking, alcoholism, procedure time, fluoroscopy time, radiation dose, and DAP and did not notice any significant correlation when examined in the entire cohort. In theory, we expect to notice increased damage form increased radiation dose but did not notice any strong correlation which can be attributed to the small sample size. This difficulty in establishing a correlation between radiation dose and DNA damage with comet assay has been reported previously, especially in cases with modest radiation exposure[28,20,16]. Previous studies have also demonstrated that smoking and alcoholism significantly increases DNA damage in individuals[29],[31]. We performed linear regression using change in comet assay value as dependent variable and did not notice any significant predictors of the outcome including age, gender, history of smoking or alcoholism. The small sample size is the likely explanation for the findings.
Our study has a few limitations that needs consideration. First, this is a prospective non-randomized study with limited sample size. Second, in spite of the prolonged fluoroscopy exposure, circulating blood cells may have a variable radiation exposure which can impact DNA migration on comet assay. Third, other confounding factors that could impact our results by potentially causing DNA damage and limit the ability for repair include medications, diagnostic testing, endogenous infections and poor nutritional status. Fourth, there is inter-individual variability in DNA migration with the comet assay which is unique for that individual. Fifth, these results do not apply for patients undergoing multiple EP procedures, where cumulative radiation exposure may impact DNA damage. Sixth, we did not assess other described methods such as comet head and tail intensity and tail movement for estimating DNA damage but overall, comet length is a widely-accepted method of estimating DNA damage. Finally, the differences in DNA damage in the AF ablation group cannot rule out the possibility of non – fluoroscopy related damage from the ablation itself, prior cardioversions and antiarrhythmic medications. However, currently there is a lack of data regarding the impact of local ablation on systemic DNA damage, especially in circulating eukaryotic cells as measured by the venous comet assay. Local cardiac comet assay measurements may shed further light into this issue. Despite, the limitations our pilot study provides valuable insights on the effect of prolonged radiation on direct DNA damage in patients undergoing CRT implantation and AF ablation and the continued need for radiation hygiene and protection. Further large prospective studies are warranted to evaluate the impact of this prolonged radiation exposure and ablation procedures on systemic DNA damage in both operators and patients exposed to multiple procedures on long term follow up.
It appears that most of the DNA damage in the immediate aftermath of the procedure is self-correcting at 3 months duration in this small group of patients and that there is a lack of permanent damage to the progenitor cells given the relatively short lifespan (few weeks to months) of any individual lymphocyte and monocyte. In the advent of significant DNA damage, some cells continue to persist and undergo mutagenesis resulting in cancer. Even though it is reassuring to see that the impact of fluoroscopy mediated radiation at 3 months it is still very important to minimize the radiation exposure. The ability of the human body to repair/replace cells diminishes with advancing age as it is governed by molecular, biochemical and genetic factors. Hence, these changes can be more profound in older patients undergoing AF ablation and CRT implantation being exposed to radiation exposure, especially those undergoing multiple procedures. Knowledge regarding the extent of DNA damage in patients undergoing electrophysiological procedures is important not only in helping patients make education informed decisions regarding repeat procedures but also the operator to be responsible regarding minimizing radiation exposure. Further studies are required to systematically evaluate the impact of radiation exposure on DNA damage in patients undergoing multiple electrophysiological procedures.
There was a significant increase in DNA damage as detected by comet assay immediately post procedure that resolved at 3 months in patients undergoing AF ablation. Further large prospective studies are warranted to evaluate the impact of this prolonged radiation exposure and DNA damage on long term follow up.