Usefulness of 3D Mapping in Catheter Ablation of Residual AF Driver Showing Fibrillatory Activation in Isolated SVC
Yuta Sakaguchi1, Daisuke Izumi1, Yasuhiro Ikami1, Kenichi Iijima1, Tohru Minamino1, Takayuki Inomata1
1Department of Cardiovascular Medicine, Niigata University Graduate School of Medical and Dental Sciences, Niigata, JAPAN.
Ectopic potentials in the SVC can act not only as a trigger but also as a driver of AF. These potentials may be refractory to conventional pulmonary vein isolation for AF. In the present case, the atrium resumed a sinus rhythm spontaneously on the completion of SVC isolation and AF could no longer be induced. However, there was residual fibrillatory activity in the SVC. These findings suggest that residual activity in the SVC worked as a driver of AF. SVC reconnection is commonly reported, with a prevalence of 70%. Thus, we decided to eliminate residual fibrillatory activity in the SVC to prevent recurrence of AF after the third catheter ablation session. In the presence of a fibrillatory pattern, conventional activation mapping techniques cannot map residual SVC activity; however, ICL mapping using the CARTO system can identify the target site of ablation in the isolated SVC.
Key Words : .
Kenichi Iijima, M.D., Ph.D. Department of Cardiovascular Medicine Niigata University Graduate School of Medical and Dental Sciences 1-757 Asahimachi Chuou-ku, Niigata 951-8510, JAPAN
Atrial fibrillation (AF) is a commonly observed arrhythmia that often causes clinical complications such as heart failure and embolic stroke. Initiation and perpetuation of AF can be linked to interaction between triggers and the arrhythmogenic substrate. The triggers can be premature atrial beats or a rapidly firing focus that act to initiate AF, and the arrhythmogenic substrate perpetuates AF through its electrophysiological, mechanical, and anatomical characteristics 1. AF triggers have been shown to originate mainly from the pulmonary veins (PVs) 2, and PV isolation has been performed worldwide as a standard therapy 3. In addition, numerous studies have suggested that premature beats from non-PV foci such as the superior vena cava (SVC), coronary sinus, Marshall ligament, crista terminalis, and left atrial posterior free wall could also be triggers of AF 4. Concomitant ablation of these non-PV foci with PV isolation has been reported to reduce the recurrence rate of AF 5.
Residual potentials, which are often based on automaticity or triggered activity, are frequently observed in isolated PVs 6. Of the two types of residual potentials, non-fibrillatory discrete potentials and fibrillatory potentials, the latter are considered to be more closely associated with recurrence of AF after PV isolation 6. Residual potentials also can be observed in the isolated SVC following circumferential SVC isolation 7-8. Several recent studies have observed that potential in the SVC acts not only as an AF trigger but also as a driver 7-8. In patients who have residual “fibrillatory” potential in the SVC, ablation of the residual potential in addition to the SVC isolation may improve the maintenance of sinus rhythm 7. However, the strategy is unclear for identifying the target site for ablation of residual fibrillatory activation in the isolated SVC.
Nademanee et al. were the first to propose that targeting sites with complex fractionated atrial electrograms (CFAE) could be an effective primary strategy for AF ablation 9. One of the clinically implementable advantages of CFAE-guided ablation is its ability to identify the ablation target, even in the case of fibrillatory activity such as sustained AF, thus overcoming the disadvantage of conventional activation mapping based on a constant cycle length and activation pattern. Dedicated CFAE software implemented in the CARTO system enables visualization of CFAE for localization of candidate ablation sites in AF ablation 10. However, the CFAE module has not been verified for application to residual fibrillatory activity in the isolated SVC.
We report a case of paroxysmal AF in which residual fibrillatory activation in the isolated SVC was deemed the driver of AF, and the CFAE module in CARTO accurately identified the target ablation site in the SVC.
A 44-year-old female with a 3-year history of palpitations due to paroxysmal AF was admitted to our hospital for a third session of catheter ablation. General examinations including echocardiography and cardiac computed tomography detected no structural heart disease. Left atrial diameter was 31 mm. In the first session, she underwent PV isolation (PVI) with a cryoballoon catheter (Arctic Front Advance, Medtronic, Minneapolis, MN) and inferior vena cava–tricuspid annulus isthmus line ablation using an irrigated tip catheter (Flexability, Abbott, Minneapolis, MN). However, her symptoms and AF recurred even with beta-blockers, and she underwent a second session. We confirmed recurrence of electrical connection between the left atrium and the left PVs that were isolated in the first session, and then performed re-PVI with a radiofrequency catheter (Thermocool ST SF, Biosense Webster, Inc, Diamond Bar, CA) as well as additional empiric SVC isolation. In these two sessions we confirmed that there were no non-PV foci even after intravenous infusion of 10 μg of isoproterenol; however, the patient suffered recurrent palpitations and AF at 6 months after the second session, and a third session was scheduled.
In the third session, we confirmed that there was no PV reconnection under guidance of the CARTO cardiac mapping system (Biosense Webster, Inc, Diamond Bar, CA). SVC reconnection was confirmed by AF initiated by premature atrial complexes from the SVC after the intravenous infusion of 10 μg of isoproterenol. Accordingly, we attempted to re-isolate the SVC using an irrigation catheter. Prior to the ablation, we mapped the phrenic nerve area with pacing from the ablation catheter at the output of 10V and the duration of 2ms, and the targeted area with high ICL in the SVC had sufficient distance from phrenic nerve area. During re-isolation of the SVC, the patient’s cardiac rhythm restored sinus rhythm spontaneously upon completion of reisolation of the SVC, regardless of the residual fibrillatory activation in the SVC [Figure 1]. We thought that the residual fibrillatory activation could be presumed as the driver of the patient’s AF because her AF was not maintained after SVC re-isolation and her AF would recur easily when SVC reconnection occurred in the future. Thus, we decided to eliminate the residual fibrillatory activation in the SVC. After identifying the ablation target site, we performed CFAE mapping by revisiting the interval confidence level (ICL) using the CFAE module of CARTO [Figure 2] because conventional activation mapping cannot be applied to fibrillatory activation. The ICL map revealed high ICL in the anterior aspect of the SVC. Radiofrequency application with output of 25 W to this site successfully terminated the residual fibrillatory activation in the SVC [Figure 3]. No other SVC potential was observed after the ablation, and fibrillatory activation was no longer inducible in either the entire atrium or the SVC. At one year after the last ablation session, the patient has experienced none of further palpitations, episodes of AF, and symptom suggesting SVC stenosis due to the ablation.
Figure 1. Fibrillatory activation in the superior vena cava (SVC).
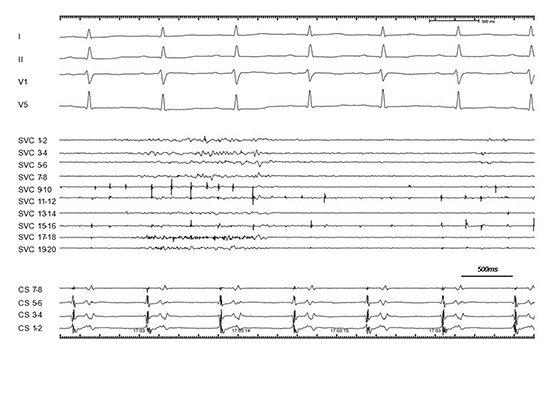
The atrium resumed sinus rhythm spontaneously after re-isolation of the SVC; however, fibrillatory activation was still observed in the isolated SVC.
We present a case of paroxysmal AF in which residual fibrillatory activation in the isolated SVC could be deemed the driver of AF, and its precise location in the SVC was successfully identified using the CFAE module of the CARTO system.
Figure 2. Interval confidence level (ICL) map in the superior vena cava (SVC) with the CARTO system
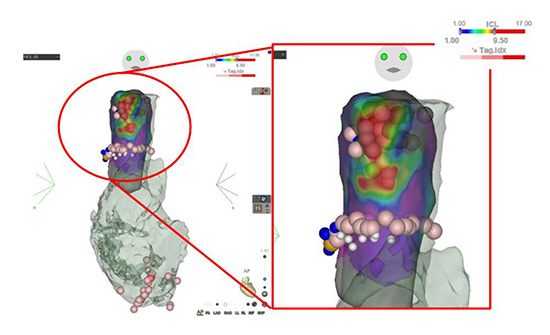
The complex fractionated atrial electrogram (CFAE) module of the CARTO system reveals that the anterior aspect of the SVC demonstrates CFAE with high ICL values. Areas of ICL > 9.5 are indicated in red.
The present patient required no less than three sessions of ablation for paroxysmal AF in a structurally normal heart. Predictors of late recurrence of AF are reported to include hypertension, LA size, LA volume, atrial tissue fibrosis, ablation strategies, and natriuretic peptides 11. However, most cases of paroxysmal AF can be cured without multiple sessions, and the present case had none of the predictors of AF recurrence listed above. In such as the present case, the presence of hidden AF triggers or drivers at an unusual site (such as the SVC) may need to be considered Eliminating these hidden causes may be the only way to free the patient from AF.
Figure 3. Ablation of fibrillatory activation in the superior vena cava (SVC).
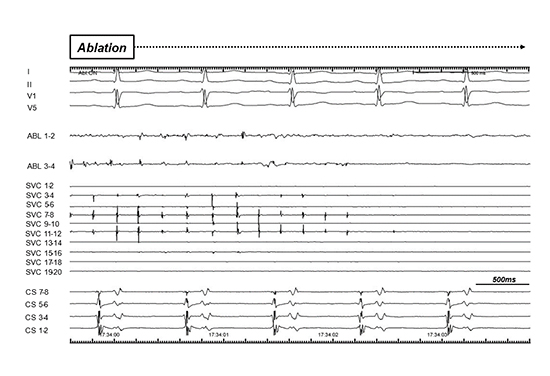
In an area with high ICL values, fragmented continuous potentials were recorded at distal pairs of electrodes of the ablation catheter. The application of 25 W radio frequency to this site terminated the fibrillatory activation in 3 seconds.
It is well known that ectopic beats originating from the SVC can initiate AF. It has been also reported that SVC tachycardia can be a driver of AF 7-8. In the present case, the atrium resumed a sinus rhythm spontaneously upon completion of the SVC isolation and AF could no longer be induced. However, there was residual fibrillatory activity in the SVC. These findings suggest that fibrillatory activity in the SVC had acted not only as a trigger but also as a driver of AF. Furthermore, SVC electrical reconnection after circumferential isolation is considered to be relatively common: Miyazaki et al. reported that it is observed in as many as 74% of patients undergoing repeated AF ablation 12. Even in the present case, SVC electrical reconnection was confirmed in the last session. Thus, elimination of the SVC activity was deemed essential to cure AF in the present case.
In previous reports 7-8, organized potentials or tachycardia that were presumed to be drivers of AF were observed in the isolated SVC. In these cases, the ablation target site could be identified relatively easily with a conventional activation mapping technique with and/or without a 3D mapping system. However, in the present case, the residual potential showed fibrillatory activity without repeatability in its cycle length and activation pattern. Thus, the conventional activation mapping technique could not be applied for ablation of the residual potential in our case.
Nademanee et al. reported that targeting CFAE could be an effective primary strategy for AF ablation 9, based on the hypothesis that CFAE areas are critical sites for AF perpetuation and can serve as target sites for AF ablation. The strongest advantage of CFAE ablation is that it enables operators to identify the therapeutic target even under the condition of fibrillatory activity. The CFAE module in the CARTO system can localize CFAE visually, and guides the operator to the candidate sites for ablation 10.
However, it is also reported that the efficacy of CFAE-based ablation in the atrium was limited 13. The iatrogenic area of arrhythmogenesis caused by extensive ablation and the low selectivity in targeting of an individual patient’s specific arrhythmic substrate were mentioned as the reasons for the lack of benefit associated with additional ablation. In the present case, the residual fibrillatory activities was localized and anchored at the anterior aspect of SVC, and these activities were termination after ablation to the limited areas with high ICL. We speculated that the efficacy of CFAE mapping in the SVC of the present case was based on the stability and localization of the targeted fibrillatory activities.
ICL is a unique parameter of the CARTO system. It is a measure of the interval of each component of fragmented potentials according to the parameter setting. In ICL mode, the number of components of fragmented potentials matching the parameter setting is counted as the ICL, and the total number of ICLs is calculated at each point. A high ICL value indicates a high density of components of fragmented potentials at the site of interest 14-15. It has been suggested that CFAE could be produced by multiple rotors serving as an AF driver 16. In the present study, we targeted SVC sites in which CFAE was expressed as high ICL values. Ablation of these sites successfully terminated the residual fibrillatory activity that had acted as the driver of AF in the present case.
SVC stenosis by the circumferential isolation has been reported in few cases and is considered to be relatively rare 17, however, its potential risk should be considered. It was reported that the SVC stenosis occurred by the widespread ablation to the entire circumference of the SVC-right atrium junction, presumably through spread of interstitial edema through the contiguous tissue space 18. The high ICL area in this case was limited at the anterior aspect of the SVC, and there was no notable anatomical structure. Therefore, we considered the ablation on the area was of low risk of SVC stenosis.
Ectopic activity from the SVC may act as a trigger and drive AF in cases of ablation-refractory AF. In the situation that residual SVC activity shows a fibrillatory pattern, which prevents use of the conventional activation mapping technique, the ICL map of the CARTO system can identify the target site of ablation in the isolated SVC.