Effect of SLGT2 Inhibitors on Patients with Atrial Fibrillation
Justin Haloot1, Lucijana Krokar1, Auroa Badin1
1University of Texas Health San Antonio, San Antonio, TX.
Sodium glucose cotransporter 2 (SGLT2) inhibitors have been associated with various cardiovascular benefits. There is limited data examining the effect of these medications on atrial fibrillation (AF) associated clinical outcomes. We compared ischemic stroke, acute coronary syndrome (ACS), cardioversion, and all-cause mortality outcomes in AF patients on SGLT2 inhibitors to propensity matched controls.
We conducted a retrospective study with a global medical research network database. AF patients were identified via ICD codes that must have been present for at least one month. Patients on SGLT2 inhibitors were identified as those on dapagliflozin, empagliflozin, or canagliflozin for at least one month. AF patients on SGLT2 inhibitors were propensity matched to those not on SGLT2 inhibitors based on age, race, ethnicity, cardiovascular comorbidities, valvular disease, pulmonary disease, urinary diseases, cardiovascular procedures, cardiovascular medications, and anticoagulants. We examined incidence of ischemic stroke, at least one ACS episode, cardioversion, and all-cause mortality.
In 26,269 AF patients, SGLT2 inhibitors were associated with lower risk of cardioversion (HR 0.921, 95% CI 0.841 - 0.999, p = 0.0245) and all-cause mortality (HR 0.676, 95% CI 0.635 - 0.721, p < 0.0001). However, there was an association with increased risk for ischemic stroke (HR 1.081, 95% CI 1.012 - 1.154, p 0.0201). There was no clear association with ACS events.
In patients with AF, use of SGLT2 inhibitors was associated with a lower risk of cardioversion and all-cause mortality and higher probability of survival based on Kaplan-Meier analysis.
Key Words : .
Justin Haloot, DO, MS
University of Texas Health San Antonio
Department of Internal Medicine
7703 Floyd Curl Dr.
San Antonio, TX 78229
Diabetes mellitus (DM) is a major cardiovascular risk factor; it is associated with increased cardiovascular events, mortality, and hospitalization 1. DM is also an independent risk factor for atrial fibrillation (AF) 2. It is associated with increase AF burden, lower quality of life, and worse AF associated morbidities 3-5. While the pathophysiologyis not fully understood, it is thought to be due to hyperglycemic-induced myocardial remodeling and expansion of epicardial adipose tissue. This leads to inflammation-related cardiac fibrosis and changes in electrical conduction that may lead to AF 5,6. Sodium glucose cotransporter 2 (SGLT2) inhibitors were developed to selectively inhibit these transporters, found exclusively in the proximal convoluted tubule of the kidneys, thereby preventing glucose reabsorption, increasing urinary glucose excretion, and decrease blood glucose levels 7. Several trials, including the EMPA-REGOUTCOME 8, CANVAS 9, DECLARE-TIMI 58 10, CREDENCE 11, DAPA-HF 12, and EMPEROR-Reduced 13trials, have demonstrated various cardiovascular benefits of these medications. However, none of these trials formally evaluated SGLT2 inhibitor effects on atrial fibrillation.
A post-hoc analysis of the EMPA-REG OUTCOME trial found that in patients with type 2 DM and established cardiovascular disease, irrespective of AF presence, empagliflozin reduced heart failure (HF) related hospitalizations and renal events 14. With regards to SGLT2 inhibitor direct effects on AF, a post-hoc analysis of the DECLARE-TIMI 58 trial found that dapagliflozin reduced incidence of AF and atrial flutter related events regardless of prior history of AF or atrial flutter in type 2 DM patients 15. Recently, analysis of the CREDENCE trial examined the effect of canagliflozin on stroke and AF in type 2 DM patients with diabetic kidney disease 16. This analysis found no significant effect of canagliflozin on risk of hemorrhagic stroke, total stroke, and AF. However, the study was not powered to evaluate this specific effect. A meta-analysis of 16 trials found that SGLT2 inhibitors in patient with type 2 DM may reduce atrial fibrillation and atrial flutter as well as all-cause mortality 17. Currently, there has been no study examining the direct effect of SGLT2 inhibitors on atrial fibrillation related outcomes and complications, regardless of diabetes status.
Therefore, we utilized a large medical research database to explore the effect of SGLT2 inhibitors on atrial fibrillation related outcomes including stroke, acute coronary syndromes, cardioversion, and all-cause mortality in patients with atrial fibrillation. The goal of our study was to compare these outcomes in AF patients on SGLT2 inhibitors to propensity score matched patients with AF not on SGLT2 inhibitors.
Data Availability Statement
TriNetX global research network contains aggregate de-identified data from anonymously participating Healthcare Organizations (HCOs), mainly in the United States, but also throughout the world. TriNetX is compliant with the Health Insurance Portability and Accountability Act (HIPAA) and data displayed are in aggregate form, containing only de-identified data. Data can be accessed via TriNetX research network at https://live.trinetx.com. There may be costs and agreement forms needed to obtain data.
TriNetX(Cambridge, MA) is a global health research network with access to electronic medical records, including diagnoses, procedures, medications, laboratory values, and genomic information from 53 health care organizations (academic medical centers, physician practices, and community hospitals) with approximately 63,500,569 patients greater than or equal to 18 years old as of August 18, 2021. It is compliant with HIPAA, the US federal law which protects the privacy and security of healthcare data. Any data displayed on the TriNetX Platform in aggregate form, or any patient level data provided in a data set, only contains de-identified data as per the de-identification standard defined in HIPAA Privacy Rule.
This is a retrospective observational study conducted via data from TriNetX Research Network. Inclusion criteria was: (1) patients having atrial fibrillation (AF) based on the International Classification of Diseases (ICD), Ninth and Tenth Revisions, Clinical Modification codes (I48.91, I48.0, I48.2, I48.1, I48.20, I48.19, I48.21, I48.11, I48.9, I48.21, I48.11)that was (2) present for at least one month. For the SGLT2 inhibitor cohort, we included (3) patients on SGLT2 inhibitors empagliflozin, dapagliflozin, or canagliflozinand (4) were on the medication for at least one month. These three were chosen due to being the most studied medications. The control cohort was designated as AF patients that were not on empagliflozin, dapagliflozin, or canagliflozin. We excluded patients that had a diagnosis of atrial flutter.
Outcomes were based on ICD codes and Common Procedure Coding System (CPT) codes that occurred at least one month after the diagnosis of AF with SGLT2 inhibitors or AF without SGLT2 inhibitors. Outcomes included ischemic stroke (I63, I63.50, G46.4, G46.3, I63.40, I63.239, I63.30, I63.139, I66.09, I63.139, I66.09, I63.59, I63.22, I63.219, I63.019, I63.20), acute coronary syndrome including unstable angina, non-ST elevation myocardial infarction, and ST elevation myocardial infarction (I20.0, I21.3, I21.1, I21.0, I21.19, I21.09, I21.2, I21.29, I21.11, I21.02, I21.21, I21.01, I21, I21.4, I25.110, I25.710, I25.700, I25.720, I25.790, I25.750, I25.760, I25.730), cardioversion (250980009, 5A2204Z, 1012978, 92960, 92961),and all-cause mortality.
TriNetX was accessed on August18, 2021, and an anonymous data set from January 1, 2016, to August 18, 2021, was obtained. SGLT2 inhibitor cohort consisted of all patients greater than or equal to 18 years old with the diagnosis of AF, based on ICD codes above, and on either empagliflozin, dapagliflozin, or canagliflozin for at least 30 days. The control cohort was composed of all patients greater than or equal to 18 years old with the diagnosis of AFand not on empagliflozin, dapagliflozin, or canagliflozin. 48 participating health-care organizations had data available for this patient population. Afterwards, propensity-scored matching was conducted and outcomes were collected.
Statistical analysis was conducted via the TriNetX online platform. Categorical variables were compared utilizing chi-square tests and continuous variables were compared utilizing independent sample t-tests. Propensity score matching at a 1:1 ratio was conducted with logistic regression to control for current age, age at AF diagnosis, gender, race, ethnicity, hypertensive diseases, hypertensive heart disease, ischemic heart disease, heart failure, valvular heart disease, metabolic syndrome (including, but not limited to hyperlipidemia, obesity, and overweight), type 2 diabetes mellitus, cerebrovascular disease, chronic kidney disease, respiratory disease, nervous system disease, neoplasms, disease of the urinary tract, cardiovascular procedures (including but not limited to echocardiography, cardiac catheterization, revascularization, coronary artery bypass grafts), cardiovascular medications (including, but not limited to: beta blockers, calcium channel blockers, antiarrhythmic drugs, antianginals), and anticoagulants (including, but not limited to: direct oral anticoagulants, warfarin, and heparin).
Cox proportioned hazardsratio with 95% confidence intervals (CI) for incidence of stroke, acute coronary syndrome (ACS), cardioversion, ablation, and all-cause mortality were generated after propensity score matching. Kaplan-Meier analysis was conducted to estimate all-cause mortality. Statistical significance was set at p < 0.05.
A total of 48 HCOs provided data for patients with AF on SGLT2 inhibitors and not on SGLT2 inhibitors. There were 26,294 patients with AF and an SGLT2 inhibitor and 1,368,518 patients with AF and not on an SGLT2 inhibitor. After propensity score matching, both cohorts had a size of 26,269 patients [Table 1]. The average age at the diagnosis of atrial fibrillation for patients in the SGLT2 inhibitor cohort and no SGLT2 inhibitor group was 66.3 + 10.5 years and 64.2 + 12.7 years respectively. The majority in each cohort were of male gender (68.5% and 70.7% respectively). 80% of patients in both cohorts had hypertension, approximately 79% had type 2 diabetes mellitus, 77% had metabolic diseases, 61% had nervous system diseases, and 57% had respiratory system diseases. Approximately 92% were on cardiovascular medications and 73% were on anticoagulants [Table 2].
Table 1. Baseline Characteristics of the AF PatientsWith and Without SGLT2 Inhibitors Before and AfterPropensity-Score Matching
|
Initial Populations |
Propensity Score Matched Populations |
|
AF and SGLT2i (n=26,294) |
AF and No SGLT2i (n=1,368,518) |
P-Value |
AF and SGLT2 (n=26,269) |
AF and No SGLT2 (n=26,269) |
P-Value |
| Age (years) at Dx of AF (mean + SD) |
66.2 + 10.5 |
71.3 + 12.9 |
< 0.0001 |
66.3 + 10.5 |
64.2 + 12.7 |
< 0.0001 |
| Gender |
| Male |
68.5% |
56.1% |
< 0.0001 |
68.5% |
70.7% |
< 0.0001 |
| Female |
29.9% |
43.4% |
< 0.0001 |
29.9% |
28.0% |
< 0.0001 |
| Unknown |
1.5% |
0.5% |
< 0.0001 |
1.5% |
1.3% |
0.0347 |
| Race/Ethnicity |
| White |
75.6% |
79.6% |
< 0.0001 |
75.7% |
76.9% |
0.0004 |
| Black |
12.5% |
8.7% |
< 0.0001 |
12.5% |
11.9% |
0.0809 |
| Asian |
1.3% |
1.4% |
0.6303 |
1.3% |
1.2% |
0.3536 |
| Hispanic/Latino |
4.9% |
3.2% |
< 0.0001 |
4.8% |
4.6% |
0.1878 |
| Co-Morbidities |
| HTN |
80.5% |
43.5% |
< 0.0001 |
80.5% |
78.3% |
< 0.0001 |
| Ischemic Heart Disease |
54.4% |
22.8% |
< 0.0001 |
54.4% |
53.4% |
0.0177 |
| Heart Failure |
43.4% |
15.1% |
< 0.0001 |
43.4% |
41.8% |
0.0002 |
| Metabolic Diseases |
77.6% |
39.3% |
< 0.0001 |
77.6% |
75.4% |
< 0.0001 |
| Nonrheumatic Mitral Valvular Disease |
20.8% |
8.8% |
< 0.0001 |
20.8% |
19.7% |
0.0015 |
| Nonrheumatic Aortic Valve Disease |
13.8% |
7.3% |
< 0.0001 |
13.8% |
12.6% |
< 0.0001 |
| Chronic Rheumatic Heart Disease |
13.5% |
5.7% |
< 0.0001 |
13.5% |
12.6% |
0.0013 |
| Type 2 Diabetes Mellitus |
77.7% |
17.4% |
< 0.0001 |
77.7% |
77.7% |
0.8421 |
| Cerebrovascular Disease |
21.2% |
11.7% |
< 0.0001 |
21.2% |
19.9% |
0.0003 |
| Chronic Kidney Disease |
25.2% |
11.4% |
< 0.0001 |
25.2% |
23.9% |
0.0004 |
| Respiratory Disease |
58.1% |
32.2% |
< 0.0001 |
58.1% |
56.2% |
< 0.0001 |
| Nervous System Disease |
61.3% |
32.1% |
< 0.0001 |
61.3% |
59.95% |
0.0017 |
| Neoplasms |
34.1% |
21.9% |
< 0.0001 |
34.1% |
33.1% |
0.0114 |
| Others |
| Cardiovascular Procedures |
67.7% |
39.5% |
< 0.0001 |
67.7% |
66.0% |
< 0.0001 |
| Cardiovascular Medications |
91.7% |
57.0% |
< 0.0001 |
91.7% |
91.7% |
0.8867 |
| Anticoagulants |
73.2% |
36.1% |
< 0.0001 |
73.2% |
73.6% |
0.2731 |
AF = Atrial Fibrillation, SGLT2i = Sodium-Glucose Co-Transporter 2 inhibitor, Dx = Diagnosis, SD = Standard Deviation,HTN = Hypertension
In the cohorts of 26,269 AF patients, SGLT2 inhibitors were associated with a higher risk of ischemic stroke (HR 1.081, 95% CI 1.012 – 1.154, p 0.0201) based on cox proportioned hazards model. SGLT2 inhibitors were associated with a higher risk of acute coronary syndrome (HR 1.04, 95% CI 0.99 – 1.10, p 0.1458), although this did not reach statistical significance. AF patients on SGLT2 inhibitors were also associated with lower risk for cardioversion (HR 0.912, 95% CI 0.841 - 0.999, p 0.0248). Lastly, SGLT2 inhibitors in AF patients were associated with lower all-cause mortality (HR 0.676, 95% CI 0.635 - 0.721, p < 0.0001) [Table 3]. Kaplan-Meier analysis demonstrated survival probability was higher in patients with AF and SGLT2 inhibitors (74.1% vs. 67.6%, p < 0.0001) [Figure 1].
Figure 1. Kaplan-Meier Survival Curve for AF Patients With and Without SGLT2 Inhibitors
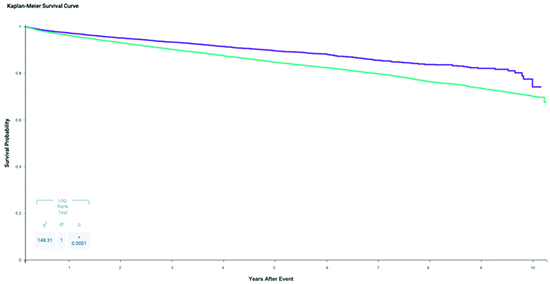
Purple = Atrial Fibrillation patients with SGLT2 Inhibitors
This is the first observational study to examine the effect of SGLT2 inhibitors and outcomes on patients with atrial fibrillation. The main findings of this study show that patients with AF on these medications were associated with lower risk of cardioversion and all-cause mortality. There appears to be a higher tendency toward an ischemic stroke and acute coronary syndrome.
Table 2. Cardiovascular Medications in Atrial Fibrillation Patients With and Without SGLT2 InhibitorsBefore Propensity Matching
| Medications |
AF and SGLT2i (n=26,294) |
AF and No SGLT2i (n=1,368,518) |
| Cardiovascular Medications |
92% |
63% |
| Antilipemic Agents |
78% |
38% |
| Atorvastatin |
55% |
23% |
| Simvastatin |
19% |
12% |
| Rosuvastatin |
18% |
6% |
| Pravastatin |
13% |
6% |
| Fenofibrate |
9% |
2% |
| Ezetimibe |
8% |
3% |
| Beta Blockers/ Related |
77% |
43% |
| Metoprolol |
57% |
30% |
| Carvedilol |
27% |
9% |
| Labetalol |
12% |
6% |
| Diuretics |
68% |
35% |
| Loop Diuretics |
54% |
24% |
| Thiazides |
36% |
18% |
| Potassium Sparing Diuretics |
27% |
8% |
| ACE Inhibitors |
52% |
24% |
| Lisinopril |
43% |
20% |
| Enalapril |
4% |
2% |
| Benazepril |
3% |
2% |
| Calcium Channel Blockers |
47% |
27% |
| Amlodipine |
28% |
17% |
| Diltiazem |
18% |
9% |
| Verapamil |
9% |
3% |
| Nifedipine |
3% |
2% |
| Angiotensin II Inhibitors |
42% |
15% |
| Antianginals |
35% |
14% |
| Nitroglycerin |
30% |
12% |
| Isosorbide |
11% |
4% |
| Isosorbide Dinitrate |
5% |
1% |
| Ranolazine |
3% |
1% |
AF = Atrial Fibrillation, SGLT2i = Sodium-Glucose Co-Transporter 2 inhibitor, ACE = Angiotensin Converting Enzyme
Atrial fibrillation is associated with increased risk of stroke, systolic embolism, and death 18,19. Currently, the CHADS2VASc score has been utilized to assess risk of stroke in AF patients 20, 21. Diabetes mellitus is one of the risk factors that is associated with a 2-fold risk of stroke alone 22. SGLT2 inhibitors in diabetic patients has been evaluated and found to have no significant differences in stroke risk from previous meta-analysis of 32 trials 23. A recent post-hoc analysis of 142 patients from the CREDENCE trial found that there was no clear effect of SGLT2 inhibitors on stroke risk in patients with AF and diabetic kidney disease, but the study was not powered to assess this outcome 24. Our study shows AF patients on SGLT2 inhibitors may be associated with a higher risk of ischemic stroke and appears to agreewith the previous study. There have been previous data demonstrating glucose-lowering agents have limited effect on stroke prevention 35. In previous studies, empagliflozin has been associated with systolic blood pressure reduction and hemoconcentration that may increase the risk for ischemic stroke 36. Further data will be needed to examine this association and relationship.
Table 3. Outcomes of AF Patients With and Without SGLT2 Inhibitors After Propensity-Score Matching
|
HR |
95% CI |
P-Value |
| Ischemic Stroke |
1.08 |
1.01 – 1.15 |
0.0201 |
| ACS |
1.04 |
0.99 – 1.10 |
0.1458 |
| Cardioversion |
0.91 |
0.84 – 0.99 |
0.0248 |
| Mortality |
0.68 |
0.64 – 0.72 |
< 0.0001 |
HR = Hazards Ratio, CI = Confidence Interval, ACS = Acute Coronary Syndrome
The most likely hypothesis for the increased risk of ischemic stroke is the blood pressure lowering effect associated with SGLT2 inhibitors. Lower blood pressure poses a risk for hypovolemia and hypotension leading to hypoperfusion and ischemic stroke. The major mechanism for this blood pressure effect may be related to natriuresis, osmotic diuresis, and reduction in body weight 37, 38
There have been multiple trials with results on major adverse cardiovascular events, including myocardial infarction (MI). The EMPA-REG OUTCOME trial demonstrated 14% relative risk reduction in major adverse cardiovascular events (MACE) including MI 8. Specifically for myocardial infarction, there was no statistically significant difference in findings. The CANVAS trial demonstrated 14% relative risk reduction of a major adverse cardiovascular event but also demonstrated no statistically significant difference in MI events 9,32. Similarly, the CREDENCE trial demonstrated a relative risk reduction in cardiovascular death, MI, or stroke 11. The DECLARE-TIMI 58 provided the largest trial and still found a non-statistically significant risk reduction in MACE, including MI 15. Our study demonstrated a non-significant association of SGLT2 inhibitors and ACS events in patients with atrial fibrillation. Again, this may be related to diabetic related microvascular disease. Currently, SGLT2 inhibitors have been thought to have anti-atherosclerotic effects through glucose control, blood pressure control, and weight loss 39. However, patients on SGLT2 inhibitors may have already worsened diabetic microvascular disease that may be confounding the outcome of ACS events. Further trials will be needed to examine the association with ACS events and SGLT2 inhibitors.
Currently, direct-current cardioversion is a Class 1 recommendation for pursuing rhythm-control strategies for AF patients 33. The findings of this study present that AF patients taking SLGT2 inhibitors are associated with lower risk of cardioversion compared to AF patients not taking these medications (HR 0.921, 95% CI 0.841 - 0.999, p = 0.0245). Therefore, this study presents new data that SGLT2 inhibitors are associated with lower risk for cardioversion and may affect the morbidity of AF patients. Further data should be explored to closely examine the association and relationship.
With regards to mechanism, there are currently animal models that have found SGLT2 inhibitors to suppress inflammation and oxidative stress in diabetic rodents 40. A hypothesis is that this decreases inflammation and myocardial fibrosis. This decrease in fibrosis can lead to less alterationin the electrical properties of the atrial tissue and could be a possible explanation for the associated lower risk for cardioversion.
SGLT2 inhibitors have been associated with lower cardiovascular mortality in various patient populations including diabetes, chronic kidney disease, and heart failure 8-13. Similarly, our study demonstrated that SGLT2 inhibitors are associated with lower mortality in AF patients (HR 0.676, 95% CI 0.635 - 0.721, p < 0.0001) compared to those not on them. This provides further evidence that supports previous studies, including a meta-analysis demonstrating an association of SGLT2 inhibitors with lower risk of sudden cardiac death 33.
There are multiple hypotheses as to the overall mortality benefit in cardiovascular patients. SGLT2 inhibitors have been associated with lower blood pressure, increased diuresis, improved cardiac energy metabolism, decreased inflammation, weight loss, improved glucose control, improved cardiac remodeling, preventing ischemic myocardial injury, decreased epicardial fat, and decreased oxidative stress 41. These effects are likely to extend to atrial fibrillation patients, who are also likely to have many similar cardiovascular morbidities that SGLT2 inhibitors are known to impact. In addition, SGLT2 inhibitors may affect left atrial appendage (LAA) remodeling either directly, by reducing cardiac remodeling, or indirectly, by lowing blood pressure and in turn LAA pressure. Further studies are needed to explain the mechanism of SGLT2 inhibitors impact on atrial fibrillation related mortality.
Aside from the inherent limitation of retrospective analysis there are several other limitations to this study. First, these results are based on aggregate data of ICD and CPT codes. There is a possibility of underreporting patients due to timing and number of diagnosis codes as well as CPT codes. We attempted to address these issues by utilizing time constraints when possible. For example, we included only patients with ICD codes for atrial fibrillation that was present for at least one month. Similarly, we only included patients that were on SGLT2 inhibitors for at least one month. This also limited the evaluation of cardiovascular specific mortality as there is no specific code for that. Second, when utilizing the TriNetX data platform, we are limited to use the platform analytical power and limiting us to their calculations and methods. Thirdly, we were unable to directly compare dapagliflozin, canagliflozin, and empagliflozin due to the limitation of the analytical software. Another limitation of this study was the inability to account for socioeconomic status (SES) risk factors in atrial fibrillation. It has been established that SES plays a role in AF burden 34 and plays a role in access to SGLT2 inhibitors. Despite these limitations, and with a comprehensive propensity score matching to any possible confounding factors, this study does provide evidence of the benefit of SGLT2 inhibitors in AF patients, and it should pave the way for prospective trials to provide further proof of this association.
In 26,269 patients with atrial fibrillation, use of SGLT2 inhibitors was associated with lower risk of all-cause mortality. This study provides further evidence for the benefits of SGLT2 inhibitors in patients with atrial fibrillation. Further data should be examined prior to determination of the full effect of SGLT2 inhibitors for this patient population.