Does Left Atrial Appendage Exclusion by an Epicardial Clipinfluence Left Atrial Hemodynamics? Pilot Results of Invasive Intra-Cardiac Measurements
Samuel Heuts1*, John H. Heijmans2*, Mark La Meir13, Bart Maesen14
1Department of Cardiothoracic Surgery, Maastricht University Medical Center, Maastricht, the Netherlands.2Department of Anesthesiology and Pain Management, Maastricht University Medical Center, Maastricht, the Netherlands.3Department of Cardiac Surgery, Brussels University Hospital, Brussels, Belgium.4Cardiovascular Research Institute Maastricht (CARIM), Maastricht University, Maastricht, the Netherlands.*denotes equal contribution.
Left atrial appendage (LAA) exclusion is the cornerstone of stroke prevention in surgical treatment of atrial fibrillation (AF). Still, little is known about the direct hemodynamic consequences of LAA closure. In the current pilot study, where we aimed to evaluate these consequences in patients undergoing hybrid AF ablation with LAA exclusion by an atrial clip, seven patients were included. Hemodynamic and intracardiac pressure measurements such as systemic, pulmonary artery (PA), central venous and LA pressure, cardiac output and indexed left ventricular stroke volume (LVSVi) were measured directly before (T0) and after (T1), and 10 minutes after (T2) LAA closure. We found no differences between all timepoints for LA pressure, PA pressure and LVSVi. As such, this is the first study describing the direct hemodynamic consequences of LAA exclusion. LAA exclusion by use of an atrial clip is safe and does not directly affect hemodynamic and intracardiac pressures.
Key Words : Atrial fibrillation, Radiofrequency Ablation, Natriuretic Peptides, Left Atrial Ejection Fraction, Left Atrial Strain.
Bart Maesen, MD, PhD
Department of Cardiothoracic Surgery
Maastricht University Medical Center+
6229 HX, Maastricht, the Netherlands
The left atrial appendage (LAA) is the most common site of thrombus formation in patients with non-valvular atrial fibrillation (AF).1 Therefore, LAA exclusion, either through amputation, stapling or clipping, is the corner stone in the surgical treatment of AF to reduce stroke risk.2 It is known that LAA closure can result in lower systolic blood pressure on the long term3 and it is suggested that it can induce volume overload by neuro-hormonal modulation on the longer term as the LAA is the predominant site of atrial natriuretic peptide (ANP) production in the heart.4 However, the acute direct hemodynamic effects of its closure remain unknown. Therefore, the aim of the current pilot study was to report on the direct hemodynamic consequences of LAA closure evaluated by hemodynamic and invasive intracardiac pressure measurements during hybrid AF ablation.5
Seven patients undergoing hybrid AF ablation were included in the current study (see [Table 1]). Patient were referred for paroxysmal or persistent AF. Our local ethics committee approved the prespecified study protocol (METC 14-5-078, dated October 10th, 2014).
Table 1. Baseline and surgical characteristics
|
Sex |
Age (years) |
BSA (m2) |
LVEF (%) |
LAvol (mL) |
LAVI (mL/m2) |
Rhythm at incision |
Clip size (mm) |
| Patient 1 |
M |
68 |
1.96 |
56 |
127 |
65 |
AF |
35 |
| Patient 2 |
M |
62 |
2.02 |
58 |
102 |
60 |
AF |
35 |
| Patient 3 |
M |
66 |
2.31 |
43 |
89 |
39 |
SR |
40 |
| Patient 4 |
M |
55 |
2.20 |
58 |
66 |
30 |
AF |
35 |
| Patient 5 |
M |
69 |
1.94 |
60 |
95 |
49 |
AF |
35 |
| Patient 6 |
F |
68 |
1.77 |
66 |
81 |
46 |
AF |
35 |
| Patient 7 |
M |
66 |
2.08 |
56 |
78 |
38 |
SR |
35 |
AF: atrial fibrillation, BSA: body surface are, LAVI: indexed left atrial volume, LAvol: left atrial volume, LVEF: left ventricular ejection fraction, SR: sinus rhythm
The surgical procedure was described in detail elsewhere.6 Before incision, the absence of LAA thrombus is confirmed by trans-esophageal echocardiography (TEE). After surgical AF ablation consisting of bilateral pulmonary vein isolation with use of a biparietal bipolar radiofrequency (RF) ablation clamp (OLL2/OSL2 Isolator Synergy Access Clamp, Synergy System, AtriCure), and superior and inferior connecting lesions (so-called ‘box-lesion’) with use of unidirectional bipolar RF device (Coolrail, Atricure) via bilateral thoracoscopy, the LAA is excluded using the Atriclip Pro device (Atricure, Mason, OH, USA). Hereafter, endocardial validation of epicardial lesions and touch-up ablation, if needed, is performed. The creation of additional lesions is left at the discretion of the electrophysiologist (EP).
Echocardiographic evaluation
All patients underwent pre-operative trans-thoracic echocardiography with extensive atrial and ventricular function and dimension evaluation [Table 1]. Per-procedural echocardiographic evaluation was performed with TEE (Epiq 5, Philips, Eindhoven, the Netherlands).
General anesthesia was provided by total intravenous anesthesia (TIVA) using propofol (diprivan, hypnoticum) and sufentanil (opioid) with a low dose of norepinephrine (in the range of 0.05 mcg/kg/min). Before LAA exclusion, it was important to maintain a steady and stable hemodynamic condition (in terms of heart rate, blood pressure and cardiac output (CO)). Therefore, hemodynamic parameters were observed during a 10 minute period, and when stable, we proceeded with LAA exclusion. During LAA exclusion and intracardiac pressure measurements, no interventions such as volume-loading (preload) or administration of additional vasoactive medication (afterload) were necessary.
Pressure and hemodynamic measurements
Systemic arterial pressure was registered by a left radial artery catheter (Arrow, Teleflex, Athlone, Ireland), central venous pressure by a central venous catheter in the right jugular vein. Pulmonary artery (PA) pressures were measured by a Swan-Ganz PA catheter (Edwards Lifesciences, Irvine, CA, USA). Cardiac output (CO) and left ventricular stroke volume (LVSV) were assessed based on arterial pressure derived monitoring (MostCareUp, Vygon, Padua, Italy). The pressure analytical method used to derive the LVSV has been described extensively elsewhere.7 Cardiac output was calculated from LVSV and heart rate using the formula: CO(liters per minute)=LVSV (liters)× heart rate (beats per minute).
For the intra-cardiac pressure measurements, transseptal puncture was performed by a flexible introducer and needle, through the right femoral vein (Agilix NXT, Abott, Lake Bluff, IL, USA). Intra-atrial location of the pressure wire allowed for precise and direct LA pressure measurements. All measurements were performed just before LA exclusion (T0), 1 minute after LA exclusion (T1) and 10 minutes after LA exclusion (T2). All patients were in sinus rhythm (without need of cardioversion) at the time of LAA exclusion and intra-cardiac pressure measurements.
Given the small study population, all data were presented as median and corresponding ranges. Testing for differences at the different prespecified timepoints was performed by use of the Wilcoxon-signed rank test. A two-sided p-value of <0.05 was considered statistically significant. Testing was performed for T0 versus T1, T1 versus T2 and T0 versus T2. Due to the sample size, data were also presented in graphs at the different timepoints for visual assessment. All statistical analyses were performed in SPSS for Mac (SPSS version 27, Armonk, NY, USA). Graphs were realized using GraphPad Prism version 7 (GraphPad software, La Jolla, CA, USA).
Median age of the patient population was 66 years (range 55-69), and included one female (14%). All patients underwent stand-alone hybrid ablation without concomitant procedures. Clip size was determined intra-operatively by assessing the LAA size with the bipolar linear RF device (Atricure). All but one patient received a size 35mm clip (86%). All patients were referred for paroxysmal or persistent AF, 5 patients were in AF at the beginning of the surgery (72%), 2 in sinus rhythm (SR, 28%). At the time of LAA exclusion and intra-cardiac pressure measurements, all patients were in SR.
Median systolic LA pressure at baseline (T0) was 22mmHg (range 13-30), 1 minute after LAA exclusion (T1) 24mmHg (range 15-31mmHg) and 10 minutes after LAA exclusion (T2) 22mmHg (range 15-29mmHg). There were no significant differences in LA pressure between the different timepoints T0 versus T1, T1 versus T2 or T0 versus T2 (p=0.131, p=0.339 and p=0.865 respectively, [Figure 1A], [Table 2]).
Figure 1. Graphical representation of the different invasive pressure and hemodynamic measurements
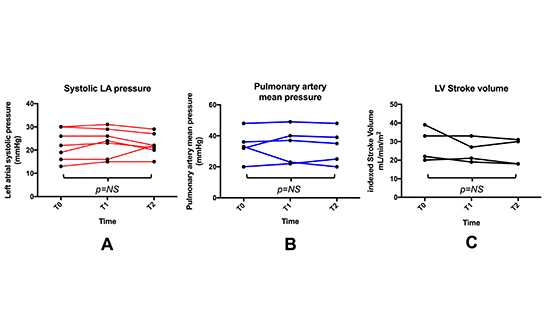
NS: non significant
Table 2. Pressure measurements and hemodynamic parameters
|
T0 |
T1 |
T2 |
p-value |
| Systolic LA pressure |
22 mmHg [13-30] |
24 mmHg [15-31] |
22 mmHg [15-29] |
0.865 |
| Mean PAP |
33 mmHg [20-48] |
37 mmHg [22-49] |
35 mmHg [20-48] |
1.000 |
| Indexed LVSV |
28 mL/m2 [20-39] |
24 mL/m2 [19-33] |
24 mL/m2 [18-31] |
0.066 |
| Systemic MAP |
83 mmHg [74-90] |
88 mmHg [72-104] |
88 mmHg [73-100] |
0.345 |
| CVP |
19 mmHg [7-27] |
23 mmHg [6-24] |
23 mmHg [6-24] |
0.496 |
| SvO2 |
73% [64-77] |
78% [72-78] |
76% [72-78] |
0.368 |
Data is presented as median and corresponding [ranges], p-value presented for T0 versus T2.
Mean PA pressure at T0 was 33mmHg (20-48mmHg), at T1 37mmHg (22-49mmHg) and at T2 35mmHg (20-48mmHg). There were no differences in mean PA pressures between the timepoints (p=0.498, p=0.414 and p=1.000 respectively, [Figure 1B], [Table 2]).
LVSV was measured and corrected for body size (LVSVi) by use of the body surface area (BSA). At baseline, LVSVi was 28mL/m2 (20-39mL/m2), at T1 24mL/m2 (19-33mL/m2), and at T2 24mL/m2 (18-31mL/m2). There were no significant differences in LVSVi at the different timepoints and overall (p=0.285, p=0.581, p=0.066 respectively, [Figure 1C], [Table 2]).
Other hemodynamic measurements, such as mean systemic arterial pressure (p=0.345), central venous pressure (p=0.496) and central venous oxygen saturation (p=0.368) did not exhibit a statistical difference between T0 and T2 ([Figure 1], D-F, [Table 2]).
In the setting of hybrid AF ablation, we have the unique possibility of simultaneous measurement of intracardiac pressures, without adding risk to the procedure.6 As such, the current pilot study is the first to perform intracardiac pressure measurements intra-operatively. We aimed to evaluate the direct hemodynamic consequences of LAA exclusion in these patients. Surprisingly, we did not observe any significant changes in hemodynamic parameters or intracardiac pressure measurements just before, directly after, and 10 minutes after release of the atrial clip.
Neurohormonal, the LAA is the main site of ANP production, and as such, it is perceived that its exclusion can lead to volume overload in the days following surgery. In addition, the LAA also has a contractile function and takes part in the LA contraction process, especially in patients in SR. In patients in SR, LAA ejection fraction (EF) was measured to be 46%, while in the same patients in AF, EF was 26%, demonstrated in an elegant study by Akosah et al.8.
Potentially, as LAA contractile function is already diminished in patients in AF – five of seven patients in this study were in AF at the time of surgery – its exclusion does not significantly affect LA contractile function in general. Still, our findings seem in line with a previous study by de Maat et al.9, where LA function was measured only by echocardiography in patients with paroxysmal AF, undergoing this procedure.
The current pilot study included a modest number of patients, which were predominantly in AF. Therefore, it cannot be ruled out that LAA exclusion in larger patient cohorts in SR, does affect hemodynamics. Furthermore, the LAA comes in different sizes and variations, which was not assessed in the current study. It might be possible that its exclusion does have consequences in larger sized LAA’s.
This is the first study demonstrating the direct hemodynamic consequences of LAA exclusion by intracardiac pressure measurements. Although this pilot study population was modest in numbers, LAA exclusion by use of an atrial clip appeared to be safe, without affecting hemodynamic and intracardiac pressures. Future studies with larger patient cohorts are warranted to corroborate these findings.