Fluoroless Catheter Ablation of Atrial Fibrillation: Integration of Intracardiac Echocardiography and Cartosound Module
Enes Elvin Gul11*, Zahra Azizi1*, Pouria Alipour1, Sohaib Haseeb2, Rebecca Malcolm1, Maria Terricabras1, Paula Sanchez Somonte1, Bernice Tsang1, Yaariv Khaykin1, Zaev Wulffhart1, Atul Verma1, Alfredo Pantano1
1Division of Cardiology, Southlake Regional Health Centre, University of Toronto, Toronto, Ontario, Canada.2College of Medicine and Dentistry, James Cook University, Townsville, Queensland, Australia.*Contributed equally as first co-authors.
To evaluate the feasibility, safety, and clinical efficacy of non-fluoroscopic radiofrequency catheter ablation of atrial fibrillation (AF) in comparison to traditional fluoroscopy-guided ablation in a local Canadian community cohort.
We retrospectively studied consecutive patients with paroxysmal and persistent AF undergoing pulmonary vein isolation (PVI) guided by intracardiac echocardiography (ICE) and Carto system (CartoSound module). ICE-guided PVI without fluoroscopy (Zero-fluoro group) was performed in 116 patients, and conventional fluoroscopy-guided PVI (Traditional group) was performed in 131 patients.
Two hundred and forty-seven patients with AF (60.7% male; mean age: 62.2 ± 10.6 years; paroxysmal AF =63.1%) who underwent PVI were studied. Mean procedure times were similar between both groups (136.8±33.4 minutes in the zero-fluoro group vs. 144.3±44.9 minutes in the traditional group; p=0.2). Acute PVI was achieved in all patients. Survival from early AF recurrence was 85% and 81% in the zero-fluoro and traditional groups, respectively (p = 0.06). Survival from late AF recurrence (12-months) between the zero-fluoro and traditional groups was also similar (p=0.1). Moreover, there were no significant differences between complication rates, including hematoma (p = 0.2) and tamponade (p = 1),between both groups.
Zero-fluoroscopy ICE and CartoSound-guided AF ablation may be safe and feasible in patients undergoing PVI compared to conventional fluoroscopy-guided ablation.
Key Words : .
Alfredo Pantano,
Southlake Regional Health Centre
602-581 Davis Drive, Newmarket, ON, L3Y 2P6, Canada
Radiofrequency ablation (RFA) of atrial fibrillation (AF) is performed under fluoroscopic guidance and, therefore, carries a risk of radiation for both the patients and the medicalstaff attending to the procedure1–4. Although three-dimensional (3D) mapping and newer imaging modalities have allowedfor catheter visualization and successful ablation, there has been increasing interest to use minimal or zero fluoroscopy during AF ablation to limit radiation dose exposure 5–7. Recently, fluoroless RFA procedures guided by intracardiac echocardiography (ICE) and 3D mapping were safely performed with high success for complex arrhythmias 8–12. To better understand the applicability of these findings in a local Canadian population, the present analysis examined the feasibility, safety, and clinical efficacy of non-fluoroscopic RFA in comparison to traditional fluoroscopy-guided ablation in patients with AF.
Study population and protocol
From October 2017 to November 2020, 247 consecutive patients undergoing RFA for symptomatic, drug-refractory paroxysmal AF (PAF) or persistent AF were retrospectively studied. To minimize interoperator bias, one operator (AP) conducted RFA. Our center has performed Fluoroless AF ablations as the standard of care and first option since 2019. Cases before and after the implementation of the Fluoroless AF program were compared. DuringFluoroless ablations, the operator and other medical staff attending to the procedure did not wear lead aprons and the fluoroscopy system was inactivated and removed. The operator (A.P) has been practicing Fluoroless AF ablation for two years. The study was approved by the local Institutional Research Ethics Board.
Procedure duration, acute procedural success, complications, and recurrence rates were recorded. Procedure duration was recorded from initial venous access until final sheath removal after the completion of the RFA procedure. Acute procedural success was determined by both entrance and exit blocks of the pulmonary veins. Recurrence was defined by the occurrence of arrhythmia after the blanking period (three months) following RFA.
Pre- and intra-procedural description
Transesophageal echocardiography was performed to rule out intracardiac thrombus in the left atria and the left atrial appendage. Antiarrhythmic drugs were discontinued for approximately five half-lives except for amiodarone, which was discontinued for >2 weeks before the RFA procedure. Patients were asked to hold oral anticoagulation 24 hours before the procedure and a bolus of heparin (100 U/kg) was administered intravenously before the transseptal puncture. Intravenous heparin was administered to target an activated clotting time above 300 seconds.
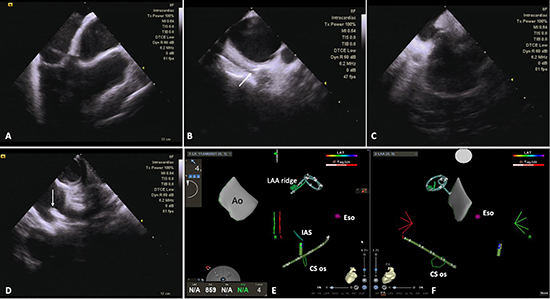
Under sterile draping in the usual fashion, three 9 French venous sheaths were placed in the right femoral groin. An 8-French Soundstar catheter (Biosense Webster, Irvine, CA) was inserted into the right atrium. After activation of the Carto Sound module, the following anatomical landmarks were marked in the mapping system: aorta, interatrial septum, coronary sinus ostium, and left atrial appendage ridge. An esophageal temperature probe was placed adjacent to the posterior wall of the left atrium and the level of the tip of the probe was marked on the mapping system with the help of ICE and CartoSound [Figure 1]. A diagnostic catheter (IBI Inquiry, Abbott Medical) was placed into the coronary sinus under ICE, EGM, and CartoSound guidance. Transseptal access was obtained under ICE guidance as follows: the wire was advanced into the right atrium, and once identified, directed into the superior vena cava (SVC). A long SL-1 sheath and dilator (Abbott Medical) were advanced over the wire into the SVC. After the wire was removed, a transseptal puncture needle (Brocken brough, Abbott Medical) was advanced to within the dilator tip. The unit was pulled inferiorly and directed to a particular region of the interatrial septum. Once “tenting” was observed,a puncture was performed and confirmed with both ICE visualization and microbubble injection. Single transseptal access was obtained in all patients ([Figure 2] and Supplementary Video). In case of failure to obtain proper tenting of the septum, we used an ablation catheter to direct the SL1 sheath to the septum, which was marked on the mapping system. The ablation catheter was then removed and replaced with a dilator and BRK needle. Transseptal puncture was performed afterward. Then,a circular Lasso catheter (Biosense Webster, Irvine, CA) was placed in the left atrium and detailed electro-anatomical mapping of the left atrium was performed with the CARTO 3 system (Biosense Webster, Irvine, CA). Following the completion of mapping, the Lasso catheter was exchanged with a contact-force (CF) sensing bidirectional Thermocool Smart Touch catheter (Biosense Webster, Irvine, CA) to start the RFA procedure. Point-by-point wide antral circumferential ablation with RF was performed in all patients. RF energy was applied at 30 to 35 Watts in the anterior aspect of pulmonary veins; however, in the posterior aspect of the pulmonary veins, RF energy was applied at 20-25 Watts due to the close proximity to the esophagus. Targeted CF was 10-20 grams except in areas that were in close proximity to the esophagus where targeted CF was 5-10 grams. Ablation index was used (450 for the anterior wall, 350-400 for the posterior wall). If AF persisted after isolation, electrical cardioversion was performed to restore sinus rhythm. Additional lesion delivery was left at the operator's discretionand could have included LA roofline, LA posterior wall isolation, and mitral isthmus line. It is worth mentioning that CartoSound was used in order to guide the Fluoroless procedure, including annotation of adjacent tissues (esophagus, aorta, interatrial septum, LAA ridge), transseptal puncture, catheter stability, and catheter insertions (coronary sinus catheter). However, the LA shell was created with the Lasso catheter. Additionally, the Lasso catheter was used to check the PV isolations at the end of the procedure.
Figure 1. Illustratrive description of fluoroless AF ablation (ICE+CartoSound). A:Main view showing aortic root, tricuspide valve, RV, and RVOT; B: Clockwise rotation of ICE catheter showing posterior aspect of left atrium along with esophagus and tip of esophagial probe (arrow); C: Interatrial septum; D: View of LAA ridge (arrow) between LAA and LSPV; E-F: Carto images of anatomical landmarks obtained by ICE. Left lateral and PA views showing aorta (Ao), interatrial septum (IAS), coronary sinus ostium (CS os), left atrial appendage ridge (LAA ridge), and tip of esophagial probe (Eso).
For the traditional fluoroscopy group, the transseptal puncture was performed with ICE and fluoroscopy guidance using standard projections and contrast agents.
Figure 2. Visualization of superior vena cava (A) and wire in the SVC (B). Tenting of the septum is visulazied (C).

Catheters were removed at the end of the procedure, and venous hemostas is was provided by a figure-of-eight suture with a three-way stop cock technique.
AF recurrence was defined as any documented episode of AF/atrial tachycardia >30 seconds. The first 90 days (3 months) immediately following the RFA procedure were considered to be the blanking period. Early recurrence was defined as any recurrence of AF/AT in 90 days. Late recurrence was defined as any recurrence at 12 months. Patients were evaluated at the outpatient clinic at 3, 6, and 12 months after the RFA procedure. A 12-lead electrocardiogram (ECG) and 48-hour Holter data were collected. Additional ECG monitoring was instructed if patients exhibited symptoms suggestive of AF. Anti arrhythmic drugs were discontinued in all patients after the blanking period, and oral anticoagulation was continued according to their CHA2DS2VASc score.
Baseline characteristics were described as mean ±standard deviation for continuous variables and frequencies and percentages for categorical variables. Comparisons between the zero-fluoro and traditional groups were made using the Student’s t-test or using the chi-squared test or their non-parametric counterparts, as appropriate. Kaplan Meier analyses were performed to analyze the survival free from early and late arrhythmia recurrence and to compare arrhythmia recurrence between patients in the zero-fluoro and traditional groups during follow-up. Cox proportional-hazards models were developed to compare the risk of early and late recurrence of arrhythmia between the two groups during follow-up. All models were adjusted for age, gender, left atrial diameter, ejection fraction, body mass index, and comorbidities regardless of their significance in the univariate model. All statistical analyses were performed using R Studio (Version 1.2.5042) and R version (Version 4.0.3) with two-sided statistical significance set at an alpha = 0.05.
Overall, 247 patients with AF who underwent PVI for AF were studied. The mean age was 62.2± 10.6 years;60.7% were male, and 63.1% had PAF. ICE-guided PVI without fluoroscopy (Zero-fluoro group) was performed in 116 patients (47%), and conventional fluoroscopy-guided PVI (Traditional group) was performed in 131 patients (53%). Baseline characteristics of the population are summarized in [Table 1].The mean fluoroscopy time in the traditional group was 12.5 ± 9.8 minutes. Patients in the traditional group had more permanent pacemakers compared to the zero-fluoro group (16.4% vs. 3.1%; p = <0.001). Otherwise, there were no significant differences in baseline characteristics before ablation between the groups. A chronological description of similar studies describing Fluoroless RFA of AF is depicted in [Table 2].
Table 1. Baseline characteristics of patients undergoing traditional and zero-fluoro ablation of atrial fibrillation.
|
Total(n = 247) |
Zero-fluoro(n = 116) |
Traditional(n = 131) |
P-Value |
| Age (years) |
62.2 (10.6) |
61.9 (10.6) |
62.6 (10.6) |
0.5 |
| Gender, male |
150 (60.7%) |
86 (65.6%) |
64 (55.2%) |
0.1 |
| PAF |
152 (63.1%) |
78 (62.4%) |
74 (63.8%) |
0.9 |
| Comorbidities |
|
|
|
|
| Hypertension |
129 (52.4%) |
65 (50%) |
64 (55.2%) |
0.4 |
| Diabetes |
28 (11.4%) |
14 (10.8%) |
14 (12.1%) |
0.9 |
| Sleep Apnea |
67 (27.2%) |
34 (26.2%) |
33 (28.4%) |
0.7 |
| Stroke |
9 (3.7%) |
3 (2.3%) |
6 (5.2%) |
0.3 |
| CAD |
38(15.4%) |
16(12.3%) |
22(19%) |
0.2 |
| Ischemic CM |
16(6.5%) |
5(3.8%) |
11(9.5%) |
0.1 |
| HCM |
7(2.8%) |
1(0.8%) |
6(5.2%) |
0.09 |
| BMI (kg/m2) |
29.8 (5.5) |
29.84 (6.2) |
29.65 (4.6) |
0.7 |
| Ejection Fractiona |
|
|
|
|
| 1 |
204 (87.2%) |
108 (90.8%) |
96 (83.5%) |
0.08 |
| 2 |
20 (8.5%) |
5 (4.2%) |
15 (13%) |
|
| 3 |
6 (2.6%) |
3 (2.5%) |
3 (2.6%) |
|
| 4 |
4 (1.7%) |
3 (2.5%) |
1 (0.9%) |
|
| LA size (mm) |
41.6 (5.5) |
41.5 (5.2) |
41.7 (5.7) |
0.7 |
| Implanted device |
|
|
|
|
| PPM |
23 (9.3%) |
4 (3.1%) |
19 (16.4%) |
<0.001 |
| ICD |
6 (2.4%) |
1 (0.8%) |
5 (4.3%) |
0.1 |
a Ejection Fraction: 1 = Normal = LVEF 50% to 70% (midpoint 60%); 2 = Mild dysfunction = LVEF 40% to 49% (midpoint 45%); 3 = Moderate dysfunction = LVEF 30% to 39% (midpoint 35%); Severe dysfunction = LVEF less than 30%.
Table 2. Previous studies reporting on fluoroless catheter ablation for atrial fibrillation.
| Study |
No. of fluoroless ablations |
Setting |
Study design |
Type of AF |
3D mapping system |
Utilization of ICE |
Energy usage |
Patients with cardiac devices (%) |
Procedure duration(min) |
Complications |
| Ferguson et al. (2009) 11 |
n = 21
No control group |
USA |
Cohort study, Prospective |
PAF + PeAF |
Yes (EnSite NavX) |
Yes(100%) |
RF |
NA |
208 (188 to 221) |
|
| Reddy et al. (2010) 12 |
n = 20
No control group |
USA |
Cohort study, Prospective |
PAF |
Yes (EnSite NavX) |
Yes (100%) |
RF |
15 |
244 ± 75 |
|
| Bulava et al. (2015) 9 |
Zero-fluoro group, n = 40
Traditional group (Control), n = 40 |
Czech Republic |
Single-center RCT |
PAF |
Yes (CARTO) |
Yes (45%) |
RF |
NA |
92.5 ± 22.9 |
|
| Sánchez et al. (2016) 19 |
n = 56
No control group |
USA |
Cohort study, Retrospective |
PAF + PeAF |
Yes (EnSite NavX) |
Yes (70.4%) |
RF |
10 |
126 ± 50 |
|
| Percell et al. (2016) 20 |
n = 20 |
USA |
Cohort study, Retrospective |
PAF + PeAF |
Yes (EnSite Precision or CARTO) |
Yes (100%) |
RF or cryoballoon |
NA |
210 |
Tamponade (1.2% in the fluorless group) |
| Razminia et al. (2017) 13 |
n = 186
No control group |
USA |
Cohort study, Retrospective |
PAF + PeAF + longstanding PeAF |
Yes (EnSite NavX or CARTO) |
Yes (100%) |
RF or cryoballoon |
10 |
194.4 |
2 pts with tamponade, 1 pt with atrioesophageal fistula |
| Liu et al. (2018) 21 |
Zero-fluoro group, n = 200
Traditional group (Control), n = 50 |
USA |
Cohort study, Retrospective |
PAF + PeAF |
No |
Yes (100%) |
RF |
9.5 |
90.3 ± 17.7 |
1 pt with partial phrenic palsy, 1 pt with pseudoaneurysm |
| Lyan et al. (2018) 15 |
Zero-fluoro group, n = 245
Traditional group (Control), n = 236 |
Russia andKazakhstan |
Cohort study, Retrospective |
PAF |
Yes (CARTO) |
Yes (100%) |
RF |
2.4 |
108.8 ± 18.2 |
3 pts with tamponade in the Fluoroless group |
| Sommer et al. (2018) 14 |
n = 1000
No control group |
Germany |
Clinical Registry |
PAF + PeAF + longstanding PeAF |
Yes (EnSite Precision) |
No |
RF |
NA |
120 ± 40.4 |
Femoral pseudoaneurysm (n= 10), Pericardial effusion (n = 7), arteriovenous fistula (n= 1), phrenic nerve palsy (n= 1), stroke (n= 1) |
| Cha et al. (2020) 18 |
Zero-fluoro group, n = 30
Traditional group (Control), n = 30 |
South Korea |
Cohort study, Retrospective |
PAF + PeAF |
Yes (CARTO) |
Yes (100%) |
RF |
NA |
163.9 ± 59.7 |
1 pt with pericardial effusion in the Fluoroless group |
| Salam et al. (2020) 22 |
n = 325
No control group |
USA |
Case series |
PAF + PeAF |
Yes (CARTO) |
Yes (100%) |
RF |
NA |
134.0 ± 30.5 |
|
| Zei et al. (2020) 23 |
Zero-fluoro group, n = 100
Traditional group, n = 60 |
USA |
Clinical Registry |
PAF + PeAF |
Yes (CARTO) |
Yes (100%) |
RF |
1 |
192 ± 37 |
1 pt with TIA in the Fluoroless group |
| Jan et al. (2020) 24 |
n = 144
No control group |
Slovenia |
Cohort study, Prospective |
PAF |
Yes (EnSite NavX or CARTO) |
Yes (100%) |
RF |
NA |
175 ± 40 |
4 pts with pericardial effusion w/o tamponade, 2 pts with pseudoaneurysm of femoral artery, 1 pt with tamponade, 1 pt with transient phrenic nerve injury |
| Žižek et al. (2020) 25 |
n = 451
No control group |
Slovenia |
Cohort study, Retrospective |
PeAF |
Yes (EnSite NavX or CARTO) |
Yes (100%) |
RF |
5.4 |
161 ± 64 |
6 pts with pericardial effusion, 2 pts with PSA intervention, 1 pt with tamponade, 1 pt with transient phrenic palsy, 1 pt with CIED lead dislocation |
| Lurie et al. (2020) 16 |
Zero-fluoro group, n = 147
Traditional group (Control), n = 176 |
Canada |
Cohort study, Retrospective |
PAF + PeAF |
Yes (EnSite or CARTO or Rhythmia) |
Yes (100 %) |
RF |
2.7 |
176 ± 46 |
2 pts with pericardial effusion or tamponade, 1 pt with atrioesophageal fistula in the Fluoroless group |
| Present study (2020) |
Zero-fluoro group, n = 116
Traditional group (Control), n = 131 |
Canada |
Cohort study, Retrospective |
PAF + PeAF |
Yes (CARTO) |
Yes (100 %) |
RF |
2 |
136.85 ± 33.4 |
1 pt with tamponade in the Fluoroless group |
Abbreviations: AF = atrial fibrillation; CIED = cardiac implantable electronic devices; NA = not available; PAF = paroxysmal atrial fibrillation; PeAF = persistent atrial fibrillation; PVI = pulmonary vein isolation; PSA = pseudoaneurysm; RF = radiofrequency; RCT = randomized clinical trial; TIA = transient ischemic attack
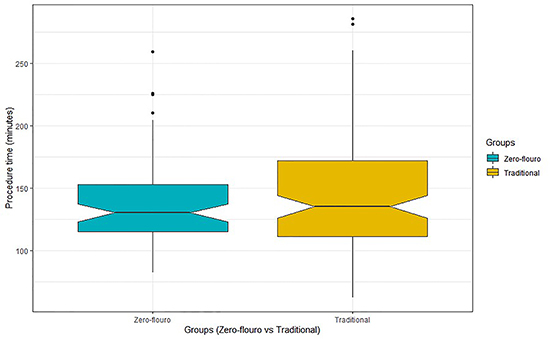
ICE was used in all patients. Initial rhythm, site of ablation, and need for direct current cardioversion post-RFA were similar between both groups. In all cases, RFA was successfully performed. Procedure-related details are summarized in [Table 3]. Mean procedure times were similar between both groups (136.8 ± 33.4 minutes in the zero-fluoro group vs. 144.3 ± 44.9 minutes in the traditional group; p = 0.2) [Figure 3].
Table 3. Procedural details of patients undergoing traditional and zero-fluoroablation of atrial fibrillation.
|
Total |
Zero-fluoro |
Traditional |
P-Value |
| General Anesthesia |
140(57.4%) |
104(79.4%) |
36(31.9%) |
<0.001 |
| Initial Rhythm |
|
|
|
0.9 |
| SR |
175(71.7%) |
92(70.8%) |
83(72.8%) |
|
| AT |
4(1.6%) |
2(1.5%) |
2(1.8%) |
|
| AF |
65(26.6%) |
36(27.7%) |
29(25.4%) |
|
| Additional lesion delivery |
|
|
|
|
| LA Roof Line |
4(1.6%) |
0(0%) |
4(3.5%) |
0.1 |
| LA Posterior Wall |
5(2%) |
1(0.8%) |
4(3.5%) |
0.3 |
| Mitral Isthmus |
2(0.8%) |
0(0%) |
2(1.7%) |
0.4 |
| Concurrent AFL |
4(1.6%) |
0(0%) |
4(3.5%) |
0.1 |
| DCC Post procedure |
111(44.9%) |
55(42%) |
56(48.3%) |
0.3 |
| Procedure duration, min |
140.5 ± 39.1 |
136.8 ± 33.4 |
144.3 ± 44.9 |
0.2 |
Abbreviations: AF = Atrial Fibrillation; AFL = Atrial Flutter; AT = Atrial Tachycardia; DCC = Direct Current Cardioversion; ICE = Intracardiac Echocardiography; LA = Left Atrial; SR = Sinus Rhythm
Figure 3. Box plot of the total procedure time between zero-fluoro and traditional AF ablation groups.
There were no significant differences between complication rates, including hematoma and tamponade, between both groups. The incidence of hematoma was 1 (0.8%) in the zero-fluoro group compared to 4 (3.4%) in the traditional group (p = 0.2). Moreover, the incidence of tamponade was 1 (0.8%) in the zero fluoro group and none in the traditional group (p = 1).The one tamponade in the zero-fluoro group occurred during LA ablation, likely due to a steam pop in the anterior aspect of the RSPV. No other complications occurred in this group.
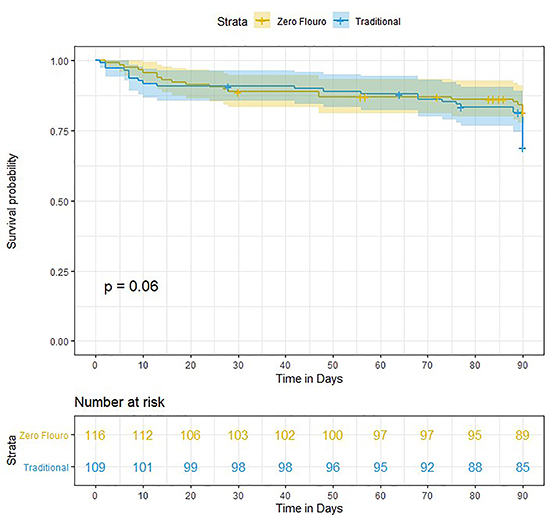
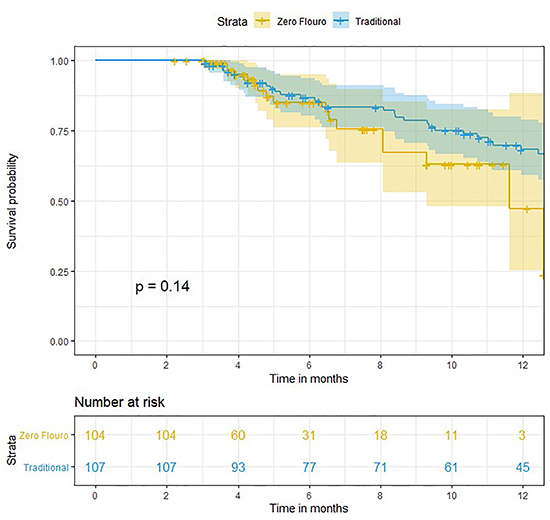
Survival from early AF recurrence was 85% and 81% in the zero-fluoro and traditional groups, respectively (p=0.06) [Figure 4]. Twelve-month survival rates from late AF recurrence were50% in the zero-fluoro group and 68% in the traditional group, respectively [Figure 5]. Moreover, survival from late AF recurrence (12-months) in the zero-fluoro and traditional groups were not significantly different (p=0.1).
Figure 4. Survival curve for early recurrence of AF in the zero-fluoro and traditional groups.
A Cox proportional-hazards multivariate analysis of the variables, including age, gender, type of AF (PAF vs. persistent AF), presence of hypertension, diabetes, left atrial size, left ventricular ejection fraction, and body mass index showed no significant differences in the risk of early and late AF recurrence (12-months) between the zero-fluoro and traditional groups during follow-up (Traditional vs. Zero-fluoro: early recurrence: Hazard ratio (HR) = 1.7[0.9-3.2], late recurrence: HR0.5[0.2-1.02]).
Figure 5. Survival curve for late recurrence of AF in the zero-fluoro and traditional groups.
In this single-center study of a local Canadian community, we showed that zero-fluoroscopy RFAhad similar efficacy and safety profiles compared to the traditional fluoroscopy-guided approach. Furthermore, the procedure duration between zero-fluoroscopy and traditional groups was comparable. Freedom from AF was also comparable between both groups. In this study, we showed the safety and feasibilityintegration of imaging (ICE and CartoSound) and EGM guidance in order to perform fluoroless catheter ablation in patients with PAF and persistent AF.
An increasing number of studieshave recently reported on the feasibility and safety of zero-fluoroscopy AF ablation [Table 2].Ferguson et al.first demonstrated this technique in 21 patients with AF usingrotational ICE and 3D mapping. The mean procedure duration was 208 minutes, and there were no complications11.This was followed by Reddy et al.,who described a series of 20 consecutive patients who underwent PVI for PAF using zero-fluoroscopy. The procedure duration was 244 ± 75 minutes, with 97% acute procedural success12. The only randomized clinical trial to comparethe nonfluoroscopic approachwith the traditional approach was performed in 80 patients with PAFin a single-center experience. The authors found no differences in the proceduraloutcomes or overall safety between the two groups 9. Most of these studies have been limited by their small scaleor a predominant focus on acute procedural outcomes. Razminia et al. reported 5-year experience of zero-fluoroscopy ablation by using ICE and the Ensite system and demonstrated that it was feasible and safe 13. The largest study employing zero-fluoroscopy during AF ablation was conducted by Sommer et al. with their experience of 1,000 patients treated without fluoroscopy with the Mediguide system 14. ICE was not used to guide the procedure. Another large-scale studyreporting on the long-term safety and efficacy following fluoroless AF ablation retrospectively reviewed 481 consecutive patients with PAF15. These authors compared ICE-guided PVI (n = 245) with conventional fluoroscopy-guided PVI (n = 236) and reported no differences in procedural outcomes, complication rates, and AF recurrence. Based on this evidence, zero-fluoroscopy AF ablation utilizing ICE and 3D mapping appears to be feasible, safe, and effective compared to fluoroscopy-guided ablation. Our cohort included 247 consecutive patients with PAF or persistent AF and included 131 patients in the fluoroscopy-guided RF ablation group and 116 patients in the zero-fluoroscopy ablation group. CARTO 3D mapping was utilized during all our procedures.Lurie et al. recently detailed their first-ever Canadian experience with 323 consecutive AF patients. Of these, 176 patients were treated with fluoroscopy-guided RF ablation, and 147 patients were treated with zero-fluoroscopy ablation and utilized3D mapping systems (EnSite, n = 175; CARTO, n = 144; Rhythmia, n = 3). Minimal fluoroscopy was used in 17 patients in the zero-fluoroscopy group (median 3 seconds, IQR 1.2 – 4.8)16. Both studies (Lurie et al. and our cohort) included patients with cardiac devices as well. In comparison with other previous studies, our cohort revealed high acute procedural success rate of 100%, acomplication rate of 0.9%, and aprocedure durationof 136.85 ± 33.4 minutes. The low complication rate with the zero-fluoroscopy technique may be due to visualizing the guide wires and the catheters while advancing into the heart to avoid injuries.
It has been reported that the learning curve for zero-fluoroscopy AF ablation occurs over the first 20 cases 17.For an experienced operator, zero-fluoroscopy AF ablation might be achieved safely and feasibly within 5 to 10 cases 18.Our recommendation, based on experience, is for operators with less experience to avoid Fluoroless AF ablation in patients with cardiac devices in order to prevent any inadvertent lead issues such as dislodgement. Operators experienced in the utilization of ICE may not view this as a limitation. We believe that if the operators perform zero-fluoroscopy procedures as routine practice, adverse effects of ionizing radiation could be significantly reduced in both patients and the medical staff.
The study was performed by a single operator at a single center, and therefore results of this study may not be applicable to other centers. For context, three operators perform around 450 to 500 AF ablations a year; however, only one operator in the practice performs Fluoroless AF ablation. Another limitation was our study’s retrospective and nonrandomized design. There were very few patients with cardiac devices in the zero-fluoroscopy arm; therefore, more data are required before adopting a zero-fluoroscopy approach in patients with cardiac devices. Detailed procedure measures such as time to transseptal puncture and left atrial dwell time were not recorded, which could differ between zero-fluoroscopy and traditional groups. Another limitation was that an esophageal temperature probe (Smiths Medical, Minneapolis, MN, USA) with a single sensor was used. Only one point was marked on the map. This may not be sufficient since the esophagus has a longer course. Lastly, this technique may not be adaptable to other mapping systems as the CARTOSOUND® Module with SOUNDSTAR® Catheter are only compatible with CARTO® 3 System and proprietary to Biosense Webster.
Fluoroless RFA was associated with similar safety and efficacy profiles as the traditional AF ablation. In addition,AF ablation without fluoroscopy did not prolong the total mean procedure time or compromise the recurrence rate during long-term follow-up.