Localization of Right Ventricular Outflow Tract Premature Ventricular Complexes Using a Novel Mapping System
Mehmet Ali Elbey1, Sri Harsha Kanuri1, Naushad Shaikh2, Rakesh Gopinathannair1, Dhanunjaya Lakkireddy1
1Kansas City Heart Rhythm Institute (KCHRI), Overland Park, Kansas.2Cardiovascular Associates Inc, Kissimmee, Florida.
Premature ventricular complexes (PVCs) are common in the general population, usuallyasymptomatic, and deemed to be benign in structurally normal hearts. The spectrum of “benign” outflow tract PVCs ranges from single PVCs to recurrent non-sustained ventricular tachycardia (NSVT). Short-coupled right ventricular outflow tract (RVOT) PVCs may trigger polymorphic ventricular tachycardia (VT) in some patients and can be high risk. In many patients, PVCs can be morefrequent and cause symptoms ofpalpitations, shortness of breath, dizziness, and heart failure.In the presence of underlying heart disease, they may indicate an increasedrisk of adverse cardiovascular outcomes. A high PVC burdenmay lead to ventricular dysfunction and worsen underlying cardiomyopathy.PVCs may also be a marker of underlying pathophysiologic processes such as myocarditisand other acquired and inherited infiltrative cardiomyopathies. In this unique case report, we describe the use of a novel non-contact mapping array for mapping RVOT PVCs.
Key Words : Premature ventricular complex, Non-contact mapping system, Radiofrequency catheter ablation, Right ventricular outflow tract, New mapping system, Contact mapping system.
Dhanunjaya Lakkireddy, MD, FACC, FHRS
Executive Medical Director
The Kansas City Heart Rhythm Institute (KCHRI) @ HCA MidWest
Professor of Medicine, the University of Missouri - Columbia
12200, W 106th street, Overland Park Regional Medical Center
Overland Park, KS 66215
Premature ventricular complexes (PVCs) are common in the general population, usually asymptomatic, and deemed to be benign in structurally normal hearts 1. The spectrum of “benign” outflow tract PVCs ranges from single PVCs to recurrent non-sustained ventricular tachycardia (NSVT). Short-coupled right ventricular outflow tract (RVOT) PVCs may trigger polymorphic ventricular tachycardia (VT) in some patients and can be high risk 2. In many patients, PVCs can be more frequent and cause symptoms of palpitations, shortness of breath, dizziness, and heart failure. In the presence of underlying heart disease, they may indicate an increased risk of adverse cardiovascular outcomes. A high PVC burden may lead to ventricular dysfunction and worsen underlying cardiomyopathy. PVCs may also be a marker of underlying pathophysiologic processes such as myocarditis and other acquired and inherited infiltrative cardiomyopathies 1, 3. In this unique case report, we describe the use of a novel non-contact mapping array for mapping RVOT PVCs.
The patient is a 61-year-old male with known recurrent frequent PVCs. His medical history includes non-obstructive coronary artery disease, hypertension, hyperlipidemia, and peripheral artery disease. He underwent heart catheterization in June 2020 which demonstrated a 50% left anterior descending lesion but no significant obstruction. He underwent cardiac positron emission tomography which demonstrated an ejection fraction (EF) of 60% with no myocardial inflammation. The patient was evaluated with thoracic echocardiography before the procedure, left ventricular EF was 60% and the right ventricle cavity, wall thickness, and systolic functions were all normal. An implantable loop recorder was placed in August 2020. ILR interrogation demonstrated a high PVC burden (16.9%). A 12-lead ECG suggested probable RVOT PVC origin. Then we decided to perform a PVC ablation procedure. The patient underwent monitored anesthesia care with the anesthesia. Right and left femoral venous access was obtained using vascular ultrasound guidance. Arterial hemodynamic monitoring was provided through the catheter. An intracardiac echo catheter was advanced via an 11-French sheath into the left femoral vein and positioned in the right atrium. A comprehensive EP study was performed with coronary sinus catheter placement and right ventricular ablation catheter. The non-contact mapping catheter was inserted into the RV and shown with Fluoroscopy ([Figure 1A] and Intracardiac Echocardiography [Figure 1B]. AcQMap (Acutus Medical, CA, USA) and EnSite Precision (Abbott, St Paul, MN, USA) mapping systems were used for 3-D mapping of the ventricular arrhythmia. Based on previous 12-lead ECG and implantable loop recorder monitoring, we anticipated the PVCs to be originating from the RVOT. We correlated the PVC site of origin using the Precision mapping system. Then non-contact mapping catheter was successfully passed through the tricuspid valve because it is handling, and deliverability is easy. We mapped the RV and outflow tract using the AcQMap non-contact mapping system [Figure 2A] and were able to successfully identify and tag the origins of the PVCs. The mapping time at the RV was 180 seconds using the AcQMap non-contact mapping system. Whereas the conventional 3D mapping using the Precision system took close to 14 minutes for the mapping. There were 2 PVCs, one from the RVOT free wall, as well as the second PVC from the RVOT upper septum. We then introduced a contact mapping catheter (HD Grid catheter) [Figure 2B] and similarly mapped the right ventricle and outflow tract localizing the PVCs to the same locations previously identified by the AcQMap system. The earliest local activation timing on the bipolar signal from the tip electrode was provided [Figure 2C]. Both PVCs were successfully mapped using the 3-D map and successfully ablated with a TactiCath Catheter at 40W. Total RF delivery time was for 256 seconds (Abbott, St Paul, MN, USA). At this point, isoproterenol was started with adequate heart rate response. Then a post-procedural programmed stimulation was conducted this did not induce any arrhythmias. Post-ablation intracardiac echo showed no pericardial effusion and RV ejection fraction was unchanged. At this time, we decided to conclude the procedure. The patient’s 8 months post-ablation PVC burden was <0.1% (rare).
Figure 1. The Mapping Catheter is shown with Fluoroscopy (A) and Intracardiac Echocardiography (B) in the RV ventricle. The noncontact mapping catheter was positioned generally in the center of the chamber when performing the electrical mapping. In doing so, the majority of endocardial aspects of the RV will likely be within the optimal range.
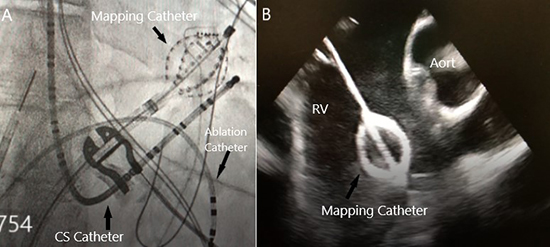
Non-contact mapping system was used in this case due to rare PVCs and we thought it would be fast, not taking a long time over the conventional mapping.
Figure 2. 2A: The AcQMap imaging and mapping system demonstrate the three-dimensional right ventricular anatomy and PVC localization (blue arrow). 2B: The Precision Mapping System demonstrates three-dimensional RV and RVOT anatomy with the HD Grid catheter, 2C: Shows PVC and the earliest activation on intracardiac ECG at the bottom (green arrows).
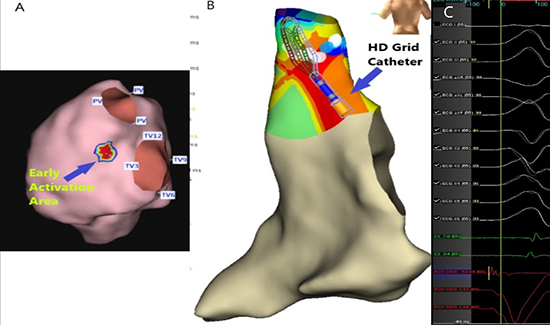
We present the first case of mapping and treatment of PVCs originating from the RVOT using the AcQMap mapping system and AcQMap catheter. The mapping catheter combines 48 ultrasound transducers responsible for reconstructing the chamber anatomy and 48 engineered electrodes which enable charge-density-based activation patterns to be displayed along the endocardial surface of the heart 4. It is the only integrated high-resolution ultrasound-based imaging and non-contact mapping catheter capable of capturing cardiac imaging information in addition to cardiac activation mapping. When combined with the AcQMap mapping system, the catheter also provides maps of most arrhythmias in under 3 minutes, enabling a map to ablate—remap strategy to evaluate therapy effectiveness 5. To map PVCs and VTs, the arrhythmia must occur naturally or be induced during the procedure. Patients can become hemodynamically unstable and rapidly decompensate. Currently marketed contact-based mapping systems require contact with the tissue which can be proarrhythmic. Similarly, there exists a likelihood of missing the arrhythmia due to lack of contact at the activation site. Non-contact mapping is ideally suited to map both PVCs and VTs as it allows for global visualization. One is able to identify and target the origin of the arrhythmia with a single beat and achieve it without disturbing the tissue. However, this mapping system was evaluated in the ventricle in an animal study 6 but has not been reported in human ventricle arrhythmias yet. Although the AcQMap catheter is designed and has regulatory approval for use in the left and right atria to map complex atrial arrhythmias, we elected to use it to map PVCs originating from the RVOT.
This case report suggests that patients presenting with PVCs originating in the RVOT can be quickly and easily mapped using a novel non-contact mapping system to identify the target area to be treated by radiofrequency catheter ablation. With appropriate clinical care, this example demonstrates the potential for safe and effective use of the system, not only in the left and right atria, but also to identify the ventricular origins of PVCs, particularly those with RVOT origin.