Clinical Utility of Intravenous Nifekalant Injection during Radiofrequency catheter Ablation for Persistent Atrial Fibrillation
Tetsuma Kawaji1, Satoshi Shizuta2, Shintaro Yamagami2, Takanori Aizawa2, Akihiro Komasa2, Takashi Yoshizawa2, Masashi Kato1, Takafumi Yokomatsu1, Shinji Miki1, Koh Ono2, Takeshi Kimura2
1Department of Cardiology, Mitsubishi Kyoto Hospital.2Department of Cardiovascular Medicine, Graduate School of Medicine, Kyoto University.
Radiofrequency catheter ablation (RFCA) for persistent atrial fibrillation (AF) is still challenging even in RFCA-era for AF. The aim of this study was to assess the clinical utility of nifekalant, a pure potassium channel blocker,during RFCA for persistent AF.
We retrospectively enrolled 157 consecutive persistentAF patientsundergoing first RFCA procedure withcomplex fractionated atrial electrogram (CFAE)ablation after pulmonary veins isolation and compared outcomes between patientswith (NFK group: N=79) and without (No-NFK group: N=78)additional CFAE ablation using intravenous nifekalant (0.3mg/kg). Primary endpoint was 24-month atrial arrhythmia-free survival post ablation.The prevalence of AF terminationwas significantly higher in NFK group than No-NFK group (64.6% versus 7.7%, P<0.001). Arrhythmia-free survival, however, was not significantly different between 2 groups (61.5% versus 54.1%, P=0.63).There was no significant difference between 2 groups in the prevalence of recurrent atrial tachycardia(25.0% versus 23.5%, P=0.89). Arrhythmia-free survivalin patients with AF termination during procedure was significantly higher thanthose without (73.0% versus 41.0%, P=0.002; adjusted hazard ratio 0.48, 95% confidence interval 0.17-0.84, P=0.02) amongNFK group,but not amongNo-NFK group (66.7% versus 53.2%, P=0.53).
Intravenous nifekalant injection during additional CFAE ablation did not improve sinus maintenancerate after RFCA procedure for AF, but AF termination by nifekalant injection could be a clinical predictor of better success rates after procedure.
Key Words : Spersistentatrial Fibrillation, Radiofrequency Catheter Ablation, Complex Fractionated Atrial Electrogram, Nifekalant.
Correspondence to: Tetsuma Kawaji,
Department of Cardiology, Mitsubishi Kyoto Hospital, 1 KatsuraGosho-cho, Nishikyo-ku, Kyoto 615-8087 Japan
Pulmonary veins isolation (PVI) is well-established treatment for paroxysmal atrial fibrillation (AF) to eliminate the triggers of AF[1-3].Guidelines recommended catheter ablation as second-line therapy for paroxysmal AF patientsrefractory to antiarrhythmic drugs (AADs) (class I)and as first-line therapy for selected symptomatic patients (class IIa)[4-5] .In contrast, catheter ablation for persistent AF is still challenging withlow sinus maintenance rate after procedure, although several studies demonstrated the superiority of catheter ablation for persistent AF over conventional treatment with AADs in sinus maintenance[6,7].Therefore, clinical utility of additional strategiesbeyond PVI to target atrial substrate such as ablation of complex fractionated atrial electrograms (CFAEs), ganglion plexus, electrical rotors and block lines have been developed, but their efficacy in addition to PVI is controversial[8-11].
AADs, especially class III AADs,increased action potential duration andsome studies had reported injection of these drugs during ablation procedure reducedCFAEsites.[12-14] We had started to use nifekalant, a pure class III AAD, during additional CFAE ablation after PVI in catheter ablation for persistent AF to detect critical CFAE sites remained even after prolongation of action potential duration by nifekalant. The aim of this study is to evaluate clinical utility of intravenous nifekalant injection during additional CFAE ablation by comparing the success rates between with and without additional CFAE ablation using intravenous nifekalantin consecutive persistent AF patients undergoing radiofrequency catheter ablation.
A total of 157 consecutive persistent AF patients undergoing first radiofrequency catheter ablation with additional CFAE ablation after PVI between October 2013 and March 2016 in Kyoto university hospital were consecutively included in our analysis [Figure 1].Intravenous injection of nifekalant during CFAE ablation was performed in 79 patients (NFK group)and the other 78 patients did not receive nifekalant during CFAE ablation (No-NFK group). The decision to use nifekalant during CFAE ablation was left to the discretion of the operators before procedure. Both groupswere further subdivided into patients with or without AF termination duringCFAE ablation.Persistent AF patients who showed sinus conversionduring PVI and did not receive CFAE ablation were excluded in this study.
Figure 1. Study population
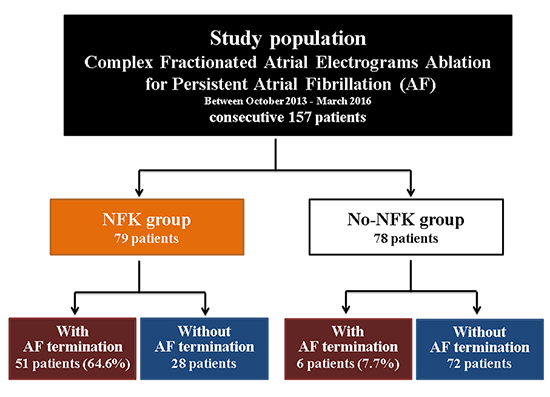
AF=atrial fibrillation
The present study protocol was approved by the ethics committee in Kyoto University Hospital. Written informed consent for the catheter ablation procedure and follow-up were obtained from all the patients.Follow-up information was obtained by hospital-chart review and/or telephone contact with the patient, relatives, and/or referring practitioners.
Extensive encircling PVI was performed with a 3.5-mm tip irrigation catheter (NAVISTAR THERMOCOOL, Biosense Webster, CA, USA) and 20-pollar circular-shaped catheters (Lasso, Biosense Webster or Orbiter PV, C.R. Bard Electrophysiology, Lowell, MA, USA). After PVI, we targeted the points with CFAE, defined as intervals >120 ms and voltages <0.05 mV,[15]around septal and inferiormitral annulusin the direction of ostium of left atrial appendagewhere are common CFAE sites and have potential of inferior bilateral Ganglionated Plexi or the vein/ligament of Marshall implicated in trigger and maintenance of AF for persistent AF [Figure 2A][15-17].In NFK group, nifekalant (0.3mg/kg) was intravenously injected after routine CFAE ablation and we targeted still remained CFAE pointson both atrium, appendage or coronary sinus. Meanwhile, further CFAE ablation was not performed in no-NFK group. CFAE ablation with a power control model with a temperature setting of 40-43℃and maximal power of 25-30 W was delivered for 30-40 seconds. We performed external electrical cardioversion for persistent AF even after CFAE ablationin both groups.Additional ablation for atrial tachycardia (AT) was performed only when AF converted to sustained AT. Linear left atrial ablations such as mitral isthmus line and left atrial roof line ablation were added if needed to terminate sustained AT. Tricuspid valve isthmus ablation was routinely performed regardless of the presence of typical atrial flutter.
Figure 2A. . A representative case of atrial fibrillation termination during procedure in NFK group.A) CARTOTM image with ablation points,A 66-year-old man with long-standing atrial fibrillation and left ventricular dysfunction received radiofrequency catheter ablation. During procedure, we ablated sites of complex fractionated atrial electrogram (CFAE) from mitral annulus to ostium of left atrial appendage (purple points)after pulmonary veins isolation.The electrical excitation of coronary sinus was organized and, then, the cycle length was constantly prolonged by intravenously injected nifekalant (0.3mg/kg)
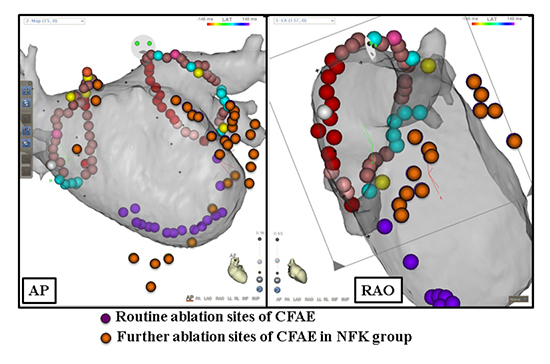
AP=antero-posterior view, d=distal, CFAE=complex fractionated atrial electrogram, CS=coronary sinus, HRA=high right atrium, p=proximal, RAO=right anterior oblique view
Definitions and Outcome Measures
Persistent AFwas defined as AF lasting beyond 7 days and further divided into 2 group: early-persistent AF (lasting <1 year) and long-standing AF (lasting>1 year).AF termination during procedure was defined as transition directly from AF to sinus rhythm or AT.
The primary endpoint was 24-month recurrent atrial arrhythmia-free survival with a blanking period of 3 months post ablation procedures. A 12-lead electrocardiogram was routinely measured at each clinical visit and 24-hour Holter monitoring was recommended at 3-, 6-, 12-month and yearly thereafter, especially in asymptomatic patients. Recurrent atrial arrhythmias were defined as those lasting >30 seconds or requiring repeat procedures. Clinically detected recurrent ATs were defined as regular atrial tachycardia having apparent and constant P waves in electrocardiogram. Other irregular atrial tachycardia having vague P waves was classified into recurrent AF.
Categorical variables were presented as number and percentage and were compared with the chi-square test when appropriate; otherwise, we used Fisher’s exact test. Continuous variables were presented as mean and standard deviation, and were compared using the Student’s t-test. We used the Kaplan-Meier method to estimate 24-month atrial arrhythmia-free rate, and assessed the difference with the log-rank test. The impact of AF termination on recurrent atrial arrhythmia after procedure in NFK group was evaluated by multivariable analysis using the Cox proportional hazard model with the following patients and procedural variables: long-standing AF, female gender, presence of diabetes, and left atrial diameter beyond 50mm in transthoracic echocardiography.Statistical analyses were performed using JMP 10 (SAS Institute Inc, Cary, NC) software. All the analyses were two-tailed, and P value <0.05 was considered statistically significant.
Mean age of the present study population was 66 years and 77% of patients were male
[Table 1]. CHADS2 and CHA2DS2-VASc score were relatively low (1.8 and 2.9, respectively). The prevalence of long-standing AF was 40%. Left atrium was dilated (46mm) and the prevalence of severely dilated left atrium beyond 50mm was 21%.The baseline characteristics between NFK and Non-NFK group were not significantly different, except for the prevalence of hypertension. The prevalence of AF termination during procedure was significantly higher in NFK group than No-NFK group (64.6% versus 7.7%, P<0.001). A representative case of AF termination in NFK group was described in [Figure 2B]. Procedure time for CFAE ablation was significantly higher and relatively more additional block line ablation was required in NFK group than No-NFK group, although total procedure time was not significantly different between 2 groups (204min versus 197min, P=0.20).
Figure 2B. A representative case of atrial fibrillation termination during procedure in NFK group.B) intracardiac electrocardiograms Still remained CFAE ablation, furthermore,was continued at left atrial appendage, anterior left atrium and coronary sinus (orange points). When we ablated sites of CFAEs at right atrial septum, atrial fibrillation was converted to sinus rhythm. We added tricuspid valve isthmus ablation and finished procedure. Sinus rhythm has been maintained for 906 days and left ventricular function is recovered.
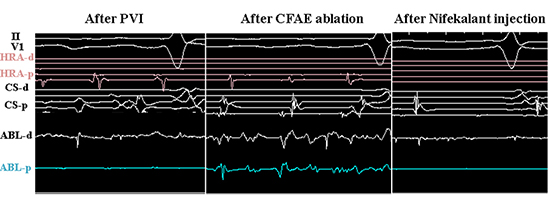
AP=antero-posterior view, d=distal, CFAE=complex fractionated atrial electrogram, CS=coronary sinus, HRA=high right atrium, p=proximal, RAO=right anterior oblique view
Table 1. Patient and Procedure Characteristics Compared between NFK group and No-NFK group.
|
All
N=157
|
NFK group
N=79
|
NFK group
N=79
|
P value |
| Age (years) |
65.7±9.0 |
65.9±10.2 |
65.9±10.2 |
0.72 |
| Age ≧75 years |
23 (14.7%) |
16 (20.3%) |
7 (11.4%) |
0.04 |
| Female gender |
36 (22.9%) |
19 (24.1%) |
17 (21.8%) |
0.74 |
| History of heart failure |
25 (15.9%) |
16 (20.3%) |
9 (11.5%) |
0.13 |
| Hypertension |
68 (43.3%) |
38 (48.1%) |
51 (65.4%) |
0.03 |
| Diabetes |
26 (16.6%) |
15 (19.0%) |
11 (14.1%) |
0.41 |
| Ischemic Stroke
|
13 (8.3%) |
6 (7.6%) |
7 (9.0%) |
0.75 |
| Vascular disease |
15 (9.6%) |
10 (12.7%) |
5 (6.4%) |
0.18 |
| CHA2DS2 score |
1.8±1.4 |
1.7±1.4 |
1.9±1.4 |
0.50 |
| CHA2DS2-VASc score |
2.9±1.7 |
2.8±1.8 |
2.9±1.6 |
0.75 |
| Long-standing atrial fibrillation |
62 (39.5%) |
30 (38.0%) |
32 (41.0%) |
0.70 |
| Echocardiography |
| Left ventricular ejection fraction (%) |
60.5±13.3 |
59.0±14.7 |
61.9±11.6 |
0.18 |
| Left atrial diameter (mm) |
45.7±6.0 |
46.1±6.3 |
45.2±5.8 |
0.34 |
| >50 mm |
33 (21.2%) |
19 (24.4%) |
14 (18.0%) |
0.33 |
| Procedure characteristics |
| Total procedure time (minutes) |
201±38 |
204±41 |
197±35 |
0.20 |
| Time for CFAE ablation (minutes) |
46.1±6.3 |
55±24 |
42±21 |
<0.001 |
| Superior vena cava isolation |
0 (0.0%) |
0 (0.0%) |
0 (0.0%) |
- |
| Block line ablation |
15 (9.6%) |
11 (13.9%) |
4 (5.1%) |
0.06 |
| AF termination |
57 (36.3%) |
51 (64.6%) |
6 (7.7%) |
<0.001 |
Categorical variables are presented as number (percentage). Continuous variables are presented as mean ± SD.CFAE= complex fractionated atrial electrogram.
In NFK group, total procedure time was significantly shorter in patients with AF termination during procedure despite the high prevalence of additional block line ablation than those without (Supplementary
[Table 1]). There was no significant difference in any of baseline and procedure characteristics between patients with and without AF termination during procedure in No-NFK groups (Supplementary
[Table 2]).
Table 2. Independent risk factors for recurrent atrial tachyarrhythmia in NFK group
|
|
Univariate |
|
|
Multivariable |
|
| Variables |
HR |
95% CI |
P value |
HR |
95% CI |
P value |
| Female gender |
1.59 |
0.68-3.41 |
0.27 |
2.05 |
0.76-5.15 |
0.15 |
| Diabetes |
0.42 |
0.10-1.21 |
0.12 |
1.05 |
0.15-1.98 |
0.48 |
| Left atrial diameter >50 mm |
2.30 |
1.01-4.97 |
0.047 |
2.53 |
0.03 |
0.48 |
| AF termination |
0.33 |
0.15-0.69 |
0.004 |
0.38 |
0.17-0.84 |
0.02 |
| Long-standing AF |
1.94 |
0.92-4.11 |
0.08 |
2.18 |
0.91-5.26 |
0.08 |
AF=atrial fibrillation, CI=confidence interval, and HR=hazard ratio.
Recurrent atrial arrhythmia with or without nifekekalant
Mean follow-up duration was 686±296 days. Recurrent atrial arrhythmia-free survival rateafter the first procedure in NFK group was 73.0% at 6-month, 66.1% at 12-month, 61.5% at 18-month, and 61.5% at 24-month,which was equivalent with that in No-NFK group: 74.3% at 6-month, 63.9% at 12-month, 59.6% at 18-month, and 54.1% at 24-month(P=0.63) [Figure 3A]. 7 out of 28 patients (25.0%) with recurrent atrial arrhythmia in NFK group and 8 out of 34 patients (23.5%) in No-NFK group were documented ATs(P=0.89).The arrhythmia-free survival rate after the last procedure was also not significantly different between NFK group and No-NFK group (75.9% versus 75.6% at 24 months, P=0.83) [Figure 3B]. Furthermore, the comparable arrhythmia-free rates between NFK group and No-NFK group werepreserved in both patients with early-persistent and long-standing AF (Supplementary [Figure 1]).
Figure 3. Recurrent arrhythmia-free survival between NFK group and No-NFK group A) after the first procedure ,and B) after the last procedure
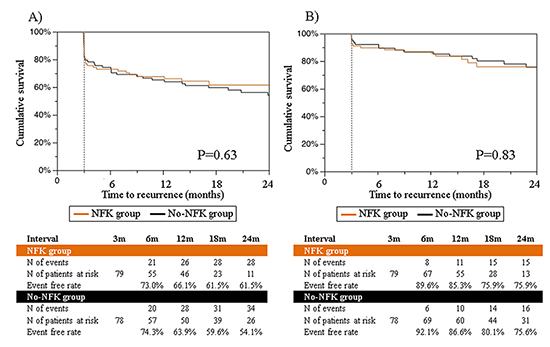
AF termination during procedure
Among 79 patients in NFK group, recurrent atrial arrhythmia-free survival rate after the first procedure in patients with AF termination during procedure was 81.9% at 6-month, 79.8% at 12-month, 73.0% at 18-month, and 73.0% at 24-month, which was significantly higher than those without: 56.9% at 6-month, 41.0% at 12-month, 41.0% at 18-month, and 41.0% at 24-month (P=0.002) (Figure4-A). AF termination during procedure, furthermore, was a significant predictor for arrhythmia-free success after procedure in multivariable analysis among NFK group (hazard ratio 0.48, 95% confidence interval 0.17-0.84, P=0.02)
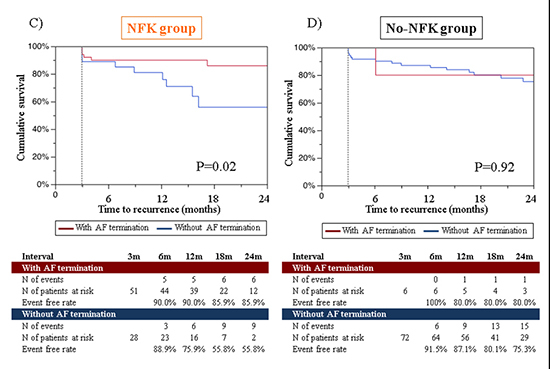
[Table 2]. The prevalence of recurrent ATs was significantly higher in patients with AF termination than those without (50.0% versus 6.3%, P=0.01). Among 78 patients in No-NFK group, in contrast, 24-month arrhythmia-free rate after the first procedure was not significantly different between patients with and without AF termination during procedure (66.7% versus 53.2%, P=0.53) [Figure 4A,Figure 4B]. The prevalence of recurrent ATs was twice, but not significantly higher in patients with AF termination than those without (50.0% versus 21.9%, P=0.42).Recurrent atrial arrhythmia-free survival rate after the last procedure was also significantly different between patients with and without AF termination in NFK group (85.9% versus 55.8%, P=0.02), but not in No-NFK group (80.8% versus 75.3%, P=0.92) [Figure 4C,Figure 4D].The better arrhythmia-free rates in patients with AF termination were more pronounced in long-standing AF (Supplementary [Figure 2]).
Figure 4A. Recurrent arrhythmia-free survival after procedure between patients with and without AF termination in NFK group and No-NFK group A),B)after the first procedure
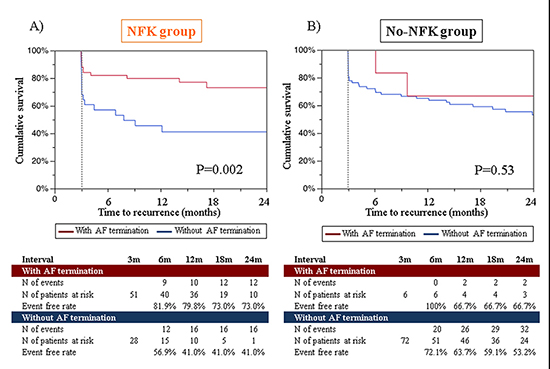
Figure 4B. Recurrent arrhythmia-free survival after procedure between patients with and without AF termination in NFK group and No-NFK group C),D) after the last procedure
This study demonstrated clinical utility of intravenous nifekalant injection during CFAE ablation in consecutive 157 patients undergoing catheter ablation for persistent AF patients. We found thatthe strategy did not improve sinus maintenance rate after procedure despite further CFAE ablation on PVI, although AF termination during CFAE ablation with nifekalant was associated with favorable outcomes after procedure.
CFAEs were described by K. Nademanee, et al in 2004 and several previous studies showed that substrate modification targeted to CFAEs improved arrhythmia-free rate after catheter ablation procedure[15,18,[19]. Conversely, broad ablation of all CFAEs could result in greater tissue destruction during catheter ablation for persistent AF and induce complex ATs after procedure[8,20,[21].STAR AF II trial, a recent randomized trial comparing three approaches to substrate ablation for persistent AF, showed no efficacy of CFAE and left atrial linear ablations[9].Y. Lin et al., however, reported limited CFAE ablation, targeted areas of mean CFAE interval <60milliseconds, improved arrhythmia-free survival after procedure compared with ablation of extensive CFAEs, defined as CFAE interval <120milliseconds[22].In substrate modification, we should detect critical contributor on maintenance of AF and avoid unnecessary tissue destruction so as not to induce complex ATs.
In the present study, weassessed the utility of further CFAE ablation with nifekalant. Nifekalant, developed in Japan, increases effective refractory period via blockade of cardiac delayed rectifier of potassium current and is called a pure class III AAD like ibutilideabroad because they are distinguished from most other class III AAD blocking sodium and calcium current as well as potassium current[12-14,[23].These drugs are usually used for ventricular arrhythmia, buthave potential to convert current-onset atrial arrhythmia to sinus rhythm by increasing effective refractory period[24,25].Regarding radiofrequency catheter ablation for AF, several small studies showed injection of these drugs after PVI provided certain rate of sinus conversion during procedure even in persistent AF or reduced areas of CFAE potential during persistent AF[14,26-28].Therefore, we believed still electrically fragmented sites where satisfied CFAE criteria even after increased effective refractory period by injection of these drugs might be critical contributors on maintenance of AF and CFAE ablation targeted these critical sites could reduce AF recurrence without increased ATs. CFAE ablation with nifekalant, however, did not improve clinical outcomes after procedure in this study, consistent with previous studies of CFAE ablation after injection of ibutilideornifekalant[27,28].Further studies would be desired to validate the utility of substrate ablation, including CFAE, electrophysiological rotor, and low voltage ablation, in combination with these drugs.
Previous studies had reported the favorable outcomes after AF termination during stepwise ablation with CFAE ablation, although Kochhauser, et al. recently reported poor correlation between AF termination and favorable prognosis[8,29-30].In the present study, meanwhile, AF terminationwas increased by nifekalant injection and predicted favorable prognosis after procedure in NFK group.The prevalence of ATs among recurrent atrial tachyarrhythmias was increased in patients with AF termination but the incidence of recurrent atrial tachyarrhythmias including AF and ATs was decreased in total. Furthermore, the favorable impact of AF termination by nifekalant was preserved even after repeat procedures.Therefore, AF terminationbynifekalant might prove proper substrate modificationand might indicate us the timing we should stop further substrate modification. The stepwise approach requires drastic ablation until AF is terminated,whilenifekalant injection during substrate ablation could be a clinical indicator to detect patients with favorable outcomes after procedure withminimum tissue destruction by reducing unnecessary ablation.
There are several limitations that should be considered. First and most importantly, our study population, especially patients with AF termination in No-NFK group, was small and we might underestimate each impact on clinical success. Further study is needed to establish the utility of nifekalant during catheter ablation for persistent AF. Second, this study was a non-randomized control trial and whether or not operators use nifekalant left to the discretion of them.However, most patients were ablated by one main operator and most baseline characteristicswere,fortunately, not significantly different between NFK and Non-NFK group.Finally, generalizing our results to populations outside Japan should be done with caution because study population was all Japanese and effect of nifekalant has been proven mainly for Japanese.
Intravenous nifekalant injection during additional CFAE ablation did not improve sinus maintenance rate after RFCA procedure for AF, but AF termination by nifekalant injection could be a clinical predictor of better outcomes after procedure.