Clinical Role of Dominant Frequency Measurements in Atrial Fibrillation Ablation – A Systematic Review
Lucaz Gadenz1, Javad Hashemi1, Mohammad Hassan Shariat1, Lorne Gula2, Damian P Redfearn1
1Queen's University, Kingston, Ontario, Canada.2London Health Sciences, London, Ontario, Canada.
Experimental data strongly supports a role for high-frequency sources in the perpetuation of atrial fibrillation, it follows that identification of areas exhibiting spectra containing high dominant frequencies (DF) may represent perpetuating sources and targeted elimination might terminate AF. The aim of this review is to present and critically appraise the literature on DF studied in association with AF ablation.
A systematic review was done including the PubMed (MEDLINE), Cochrane Central Register of Controlled Trials (Central), Scientific Electronic Library Online (SciELO), and HighWire Press databases. The searches were made by combining the terms “Dominant Frequency”, “Atrial Fibrillation”, and “Catheter Ablation” and their translations for the English and non-English based databases. Ten articles were selected from a total of 327 articles found after the initial search. The ablation strategy varied, most studies performed pulmonary vein isolation alone or associated with complex fractionated atrial electrogram ablation with or without an additional intervention. The use or not of DF sites as ablation target was distinguishable between the articles. Four articles ablated DF sites as a major intervention or in addition to a traditional approach. The remaining 6 articles assessed DF sites pre and post ablation and associated these data with clinical outcome.
No prior study has systematically comprised information for clinical use of DF. The current literature supports global DF as a useful marker of ablation outcome; however direct intervention targeting DF appears premature with mixed results and too few studies.
Key Words : Dominant Frequency, Atrial Fibrillation, Signal processing, Catheter ablation, Systematic Review.
Correspondence to: Damian Redfearn MB ChB MD MRCPI FRCPC Professor of Medicine
Queen’s University Heart Rhythm Service
Kingston General Hospital, 76 Stuart Street, Kingston K7L 2V7 Telephone: 613 549 6666 ext 3377 Fax 613 548 1387
Atrial fibrillation (AF) is the most common supraventricular arrhythmia in clinical practice with both increasing prevalence and incidence in a global pattern[1]. Catheter ablation is an accepted, widely employed therapy with good outcomes in paroxysmal AF and less optimal outcomes in persistent AF[2]. Empiric approaches, such as pulmonary vein isolation (PVI), ablation lines, and electrogram based approaches have been used with reasonable efficacy[3]. ‘Targeting’ approaches have traditionally used feature extraction of electrocardiogram signals recorded during AF. The most widely employed technique is ablation of complex fractionated atrial electrograms (CFAEs).
Fractionated electrograms are believed to represent sites of reentry that facilitate maintenance of AF[4] . CFAEs are defined as low-voltage fractionated electrograms with a cycle length <120 ms[5] . Studies have suggested that CFAEs can potentially characterize areas with wavefront collision, functional conduction block, wave break and wave fusion[6]. These sites with CFAE appear temporo-spatially stable[7] and ablation at these sites may also result in AF termination[7]-[9].
However, lack of standardization and the frequent use of the mean cycle length (CFEmean) as a poor surrogate for CFAEs, has led to conflicting data and favoring a simple, empiric circumferential ablation[2]. An alternative targeting method is identification of the spatial distribution of excitation frequencies during AF by spectral analysis of intracardiac electrograms[10]. This method is referred to as Dominant Frequency (DF) in the literature. Unlike CFAE, that is most often presented as a time domain feature of the electrograms (the mean cycle length); DF approaches contemplate the frequency contents of the electrograms. Dominant frequency is defined as the highest magnitude sinusoidal component of the electrogram and is found by decomposing the electrograms into a finite number of sinusoidal constituents and finding the one with the highest magnitude.
The DF is quantified in hertz and is variable between electrograms recorded at different anatomic sites[9]. Despite the differences between the DF and CFAE, some experimental studies have claimed that high DF activity at the posterior left atrium is the cause of fractionation of wave fronts in its immediate surroundings, resulting in CFAEs[11].
Theoretically, DF indicates the source of fibrillatory activity by recognizing the signal with a higher frequency compared with its surroundings[2]; it appears to be associated with perpetuating rotors in pre-clinical studies[12], [13]. As experimental data has strongly supported a role for high-frequency sources in the perpetuation of AF, it follows that identification and elimination of areas exhibiting spectra containing high dominant frequencies (DF) may represent perpetuating sources, might terminate AF[9], [14]. The aim of this review is to present and critically appraise the literature on DF studied in association with AF ablation.
The databases used for this review include PubMed (MEDLINE), Cochrane Central Register of
Controlled Trials (Central), Scientific Electronic Library Online (SciELO), and HighWire Press. The searches combined the terms “Dominant Frequency”, “Atrial Fibrillation”, and “Catheter Ablation” for the English based databases. The same terms and their translations were used for the non-English based databases. Articles published in English, Portuguese, and Spanish were accepted to this review. Randomized clinical trials, non-randomized clinical trials, cohort studies, case-control studies, and others interventional and observational studies were included. Guidelines, editorials, review articles, meta analysis and poster presentations were excluded.
For an article to be included in this review it must have met all the following criteria: (1) Assessment of intracardiac DF for LA or RA or both; (2) Reported ablation outcomes; (3) The DF measurements must be correlated to outcomes in any way; (4) a follow-up period of ≥ 6 months must be present. Articles linking DF to geometry, signal processing or spatiotemporal parameters without clinical correlations were excluded from this review. The number of participants and publication data was not used as exclusion criteria.
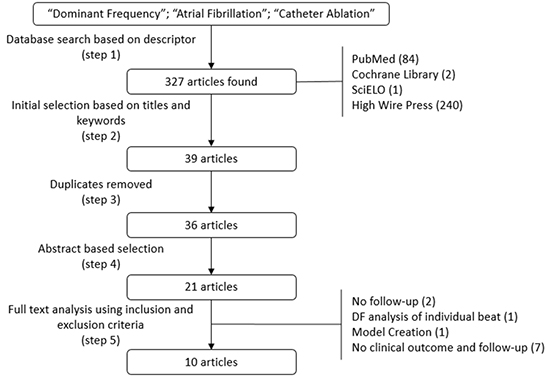
This review was based on the following steps: Database search based on descriptor (Step 1); Initial selection based on titles and keywords (Step 2); Duplicates removal (Step 3); Abstract based selection (Step 4); and Full-text analysis using inclusion and exclusion criteria (Step 5). The methodology used in this review is summarized in [Figure 1].
Figure 1. Steps followed for the systematic review.
Studies passed through the fifth step were selected for data extraction. Relevant information related to study design, type of population, follow-up period, DF assessment, ablation procedures (strategy, use of DF sites for ablation, and anatomic regions assessed), outcome periods (short and long term), complications, and correlations between DF assessment and clinical prognosis were assessed.
A total of 327 articles were identified at the initial step (84 from PubMed, 2 from Cochrane Library, 1 from SciELO, and 240 from HighWire Press). Thirty-nine potential articles remained after Step 2. Duplicates were removed in Step 3 and, 21 articles resulted after abstract selection in Step 4. The selection in Step 5 identified 10 articles which passed through quality assessment and data extraction. The reasons for exclusion of the 11 studies are listed in [Table 1].
Table 1. List of excluded articles and respective reasons.
|
Author and year
|
Title
|
Exclusion
|
|
Bencsik et al 2009
|
Acute effects of complex fractionated atrial electrogram ablation on dominant frequency and regulatory index for the fibrillatory process
|
No follow-up.
|
|
Biviano et al 2015
|
Atrial electrogram discordance during baseline vs reinduced atrial fibrillation: Potential ramifications for ablation procedures
|
No clinical outcome and follow-up.
|
|
Garibaldi et al 2012
|
Predicting catheter ablation outcome in persistent atrial fibrillation using atrial dominant frequency and related spectral features
|
No follow-up.
|
|
Grzeda et al 2009
|
Complex fractionated atrial electrograms: Properties of time-domain versus frequency-domain methods
|
Model creation.
|
|
Lee et al 2011
|
Relationship among complex signals, short cycle length activity, and dominant frequency in patients with long-lasting persistent AF: A high-density epicardial mapping study in humans
|
No clinical outcome and follow-up.
|
|
Lin et al 2012
|
A Prospective, Randomized Comparison of Modified Pulmonary Vein Isolation Versus Conventional Pulmonary Vein Isolation in Patients with Paroxysmal Atrial Fibrillation
|
DF analysis of individual beat.
|
|
Salinet et al 2014
|
Distinctive patterns of dominant frequency trajectory behavior in drug-refractory persistent atrial fibrillation: Preliminary characterization of spatiotemporal instability
|
No clinical outcome and follow-up.
|
|
Takahashi et al 2006
|
Organization of frequency spectra of atrial fibrillation: Relevance to radiofrequency catheter ablation
|
No clinical outcome and follow-up.
|
|
Takahashi et al 2008
|
Characterization of Electrograms Associated With Termination of Chronic Atrial Fibrillation by Catheter Ablation
|
No clinical outcome and follow-up.
|
|
Tuan et al 2011
|
Regional fractionation and dominant frequency in persistent atrial fibrillation: Effects of left atrial ablation and evidence of spatial relationship
|
No clinical outcome and follow-up.
|
|
Yokoyama et al 2009
|
Paroxysmal atrial fibrillation maintained by nonpulmonary vein sources can be predicted by dominant frequency analysis of atriopulmonary electrograms
|
No clinical outcome and follow-up.
|
DF = Dominant Frequency
From the 10 selected studies, 1 was a randomized controlled trial (RCT)[15], and 9 were non randomized studies (NRS)[16]-[24]. There were six studies with paroxysmal and persistent AF patients[15]-[18], [20], [21], and 4 studies with only persistent AF patients[19], [22]-[24]. One article was from 2006[18], two from 2009[16], [20],two from 2010[19], [23], two from 2011[22], [24], one from 2013[17], and one from 2014[15]. There were 3 studies which classified AF as persistent and long-lasting or long-standing persistent[17], [19], [24], and 2 studies which classified AF as paroxysmal and non-paroxysmal[20], [21]. The sample size varied among the articles with 40 the smallest[21] and 232 the highest[15] study population.
The ablation strategy varied; studies reported PVI alone or associated with CFAE ablation with or without an additional intervention such as empiric linear lesions. Four articles involved ablation of DF sites as a major intervention or in addition to an empiric approach[15]-[17], [22], one of which was a randomized clinical trial[15]. The remaining six articles assessed DF sites pre and post ablation and associated DF values with clinical recurrence.
The shortest mean follow-up was 9±6 months[18]. The regions selected for DF measurements varied, however, all articles involved LA assessment of DF. Five studies also measured DF in the RA[15], [16], [20], [21]. In addition, three articles assessed DF extracted from the atrial component of lead V1 of the 12-lead electrocardiogram (ECG)[18], [23], [24]. We empirically divided studies into outcomes that reported DF values as markers of interventional success and studies that targeted high DF sites with the aim to improve procedural outcomes. [Table 2] and [Table 3] present the selected articles according to whether high DF was used as an ablation target.
Table 2. List included of articles that did not target dominant frequency.
|
Author and year
|
DF definition
|
Type of Population
|
Ablation
|
Clinical correlation
|
Areas assessed
|
Follow-up
|
|
*Lemola et al 2006
|
The frequency of the highest peak in the periodogram (0.5-60Hz)
|
Paroxysmal (49) and Persistent (35) AF.
|
CPVA
EGA
|
Acute decrease in the DF of AF after EGA was associated with a favorable clinical outcome in patients with persistent AF.
|
V1, CS
|
9 ± 6 months
|
|
*Lin et al 2010
|
DF gradient >20%
|
Persistent (20) and Long-lasting Persistent (30) AF.
|
PVI + Linear Ablation + CFE + Non-PV Ectopies
|
Presence of an intra-LA DF gradient predicted the SR maintenance rate.
|
LA
|
11 ± 6.5 months
|
|
*Lo et al 2009
|
The normalized largest peak identified in the spectrum of 2-30Hz
|
Paroxysmal (33) and Non-paroxysmal (52) AF
|
PVI + Linear Ablation + CFAE
|
Lower mean LA DF, lower mean RA DF, and lower max RA were associated with acute AF termination.
|
LA, RA, PV
|
13 ± 8 months
|
|
*Okumura et al 2012
|
The largest peak frequency of the resulting spectrum (3-30Hz)
|
Paroxysmal (24) and Non-paroxysmal (16) AF
|
CPVI
|
Higher DF in the LA, non-paroxysmal AF and longer AF duration were independent predictor of AF recurrence.
|
LA, RA, PV, CS
|
12.3 months
|
|
**Yoshida et al 2010
|
The frequency of the highest peak in the periodogram (0.5-20 Hz)
|
Persistent (100) AF
|
APVI + CFAE
|
≥ 11% decrease in DF had the highest accuracy in predicting freedom from atrial arrhythmias.
|
V1, CS
|
14 ± 3 months
|
|
**Yoshida et al 2011
|
The frequency of the highest peak in the periodogram (0.5-20 Hz)
|
Persistent (42) and Long-standing Persistent (37) AF
|
PVI + CFE + Linear Ablation
|
DF in LAA was associated with AF termination.
|
LAA, V1, CS
|
14 ± 7 months
|
AF = Atrial Fibrillation; APVI = Antral Pulmonary Vein Isolation; CFE = Complex Fractionated Electrograms; CFAE = Complex Fractionated Atrial Electrograms; CPVA = Circumferential Pulmonary Vein Ablation; CPVI = Circumferential Pulmonary Vein Isolation; CS = Coronary Sinus; DF = Dominant Frequency; EGA = Electrogram-Guided Ablation; LA = Left Atrium; LAA = Left Atrial Appendage; NRS = Non Randomized Study; PV = Pulmonary Vein; PVI = Pulmonary Vein Isolation; and RA = Right Atrium. (*Prospective study, **Prospective and Retrospective study)
Table 3. List of articles that ablated on dominant frequency sites.
|
Author and year
|
DF definition
|
Type of Population
|
Ablation
|
Clinical Correlation
|
Areas assessed
|
Follow-up
|
|
*Atienza et al 2009
|
DFmax ≥20% of its surroundings
|
Paroxysmal (32) and Persistent (18) AF.
|
DFmax + CPVI
|
Independent predictors of freedom of AF were ablation of DFmax sites and paroxysmal AF.
|
PV-PLAW, LA, CS, RA
|
9.3 ± 5.4 months
|
|
§Atienza et al 2014
|
DFmax ≥20% of its surroundings
|
Paroxysmal (115) and Persistent (117) AF
|
CPVI only
HFSA only
CPVI + HFSA
|
Freedom was seen in 79% of CPVI and 81% of HFSA in paroxysmal AF, and in 65% of CPVI and 69% of CPVI + HFSA in persistent AF patients at 1 year.
|
LA, CS, RA
|
12 months
|
|
*Kumagai et al 2013
|
DF ≥ 8Hz with RI >0.2
|
Paroxysmal (23) AF, Persistent (9) AF and Longstanding Persistent (18) AF.
|
PVI
PVI + DF
PVI + DF + CFAE
|
AF terminated by ablation at high-DF sites (9.9 ± 0.7 Hz) in 11 of 18 patients.
|
LA, RA, CS
|
12 months
|
|
**Verma et al 2011
|
Group I: DF ≥ 8Hz with RI >0.2
Group II: DF higher than the mean atrial DF with RI >0.2
|
Persistent (80) AF
|
CFAE-based
DF + PVAI Control PVAI
|
After 1 year, DF + PVAI resulted in freedom in 17/30 patients (57%) off antiarrhythmic therapy after 1 procedure.
|
LA
|
12 ± 2 months
|
AF = Atrial Fibrillation; CFAE = Complex Fractionated Atrial Electrograms; CPVI = Circumferential Pulmonary Vein Isolation; CS = Coronary Sinus; DF = Dominant Frequency; HFSA = High-Frequency Source Ablation; LA = Left Atrium; PVAI = Pulmonary Vein Antral Isolation; PV-PLAW = Pulmonary Veins-Posterior Left Atrial Wall; PVI = Pulmonary Vein Isolation; RA = Right Atrium; RI = Regularity Index. (*Prospective study, **Prospective and Retrospective study, § Randomized Controlled Trial)
DOMINANT FREQUENCY VALUES AS A MARKER OF PROCEDURAL SUCCESS
In the following studies, DF values were employed as markers of procedural success rather than as targets for ablation. We describe the results of studies reporting outcome associated with DF values.
With a sample size of 84 patients, Lemola et al 2006[18] performed a circumferential PVI (CPVI) or an electrogram-guided ablation (EGA) in persistent AF and paroxysmal AF groups and correlated the changes in DF with clinical outcome. They simultaneously analyzed the spectral component of atrial activity from lead V1 with electrograms at the base of the left atrial appendage (LAA), on the anterior left atrial wall, and coronary sinus (CS). In both ablation approaches, EGA or CPVI significantly decreased the mean DF in the LAA and in the CS. Contrary to another similar study[23], termination of AF during ablation did not appear related to the percent DF reduction in the LA, CS or V1 values. In persistent AF patients who underwent EGA, a greater decrease in DF values in the CS and V1 was associated with freedom from recurrent AF.
In 2009, Lo et al aimed to associate different parameters with the prediction of AF termination and long-term outcome[20]. In their work, a PVI and a linear ablation at the anterior roof and lateral mitral isthmus were performed in 83 patients with an additional CFAE approach if AF was not terminated. Although smaller LA size and lower RA mean DF were the only parameters that independently predicted AF termination, other parameters such as lower mean DF at LA and RA, and lower max RA DF were also associated with acute AF termination. In addition, a larger LA diameter and presence of RA ectopy were independent predictors of late AF recurrence.
Similarly, in 2011, in order to determine if electrical and structural remodeling measurements influence outcomes in AF ablation, Yoshida et al[24] performed a PVI followed by a CFAE ablation in 79 persistent or longstanding persistent AF patients. Additional linear ablation was performed if AF persisted after PVI. Electrograms recorded prior to ablation in the LAA, and CS, and surface lead V1 were used for analysis. Patients who did not achieve AF termination had significantly higher DF in the LAA and lead V1. In a multivariate analysis, DF in the LAA was associated with AF termination. After a mean follow up of 14 ± 7 months, patients with AF recurrence had a higher DF recorded in the LAA than patients without recurrence (6.8 ± 0.7 Hz vs. 6.3 ± 0.7 Hz; P=0.03).
In a prior study from 2010, the same group examined change in DF from surface lead V1 atrial activity and the CS electrograms during the ablation procedure of persistent AF patients (PVI + CFAE)[23] and correlated with acute and long-term clinical success. Ablation terminated AF in 39 patients and 61 patients required cardioversion. The mean percentage decrease in DF was 9.8% ± 8.8% in V1 and 10.0% ± 6.8% in the CS. A decrease ≥11% in DF (V1) and AF termination during ablation procedure were both independent predictors of freedom from recurrent atrial arrhythmias after a single procedure. Also, the percent decrease in DF of AF was significantly correlated with the RFA duration directed applied to CFAE sites.
In 2012, Okumura et al[21] identified CFAEs and DF characteristics from responders and non responders, relative to AF termination during the procedure and patients with and without AF recurrences during follow-up. From the 40 patients, 19 were CPVI responders and 21 were CPVI non-responders. In the non-responders group, the DF from the LAA, LA body, and CS were significantly higher than the CPVI responders. The recurrent AF group presented significantly higher prevalence of non-paroxysmal AF patients (66.7% vs. 28.6%), longer mean AF duration (97.3 vs. 42.3 months), and higher mean DF in the LA body (6.89 ± 0.71 vs. 6.14 ± 0.85 Hz) in comparison to the non-recurrent AF group in a mean follow-up of 12.3 months. In addition, they found that those parameters (higher DF in the LA, non paroxysmal AF and longer AF duration) were independent predictors of AF recurrence.
Lin et al (2010)[19] performed PVI and linear ablation in 50 persistent AF patients and examined the effects of ablation on the patterns of DF distribution. Additional CFAE ablation was performed if AF was not terminated. Non-PV ectopy was later focally ablated in all patients. AF termination and clinical outcome were not assessed in 10 patients because the endpoint was not reached after PVI. As a result, AF terminated in 19 of the 40 patients and termination was significantly higher in patients with higher DF gradient recorded across the LA (DF max was >20% of the average DF in the LA) when compared to patients with lower DF gradient (<20% of the average DF). The isolation of DF max sites resulted in a final procedural AF termination rate of 71% after all continuous CFAE were eliminated. The DF gradient was predictive of long-term success.
DOMINANT FREQUENCY AS A MODIFIABLE TARGET FOR ABLATION
In the studies described below, high DF sites mapped within the atria were targeted for ablation and outcomes were associated with this strategy.
Initially, in 2009, Atienza et al[16] performed a catheter ablation of all DFmax sites prior to PVI in 50 patients to evaluate the safety and long-term outcome of ablation DFmax sites. In total 50 paroxysmal and persistent AF patients participated in this study. They associated higher probability of remaining free from atrial arrhythmias and fibrillation with ablation of DF sites. Recurrences of AF were higher in patients with remaining DF sites after the procedure (50% vs. 77%; P=.05). In their work, a predictor of a better outcome for persistent AF patients was found to be the presence of an LA-to-RA gradient before ablation. Moreover, ablation of DF sites and paroxysmal AF were independent predictors of long-term AF freedom.
The same group tested the hypothesis that targeted ablation of high DF sites referred to as high frequency source ablation (HFSA) would be as effective as empiric PVI procedures. In 2014, Atienza et al[15] reported a non-significant improvement in recurrence of AF or AT at 1 year in the HFSA arm in paroxysmal AF (79% vs. 81% for AF, and 72% vs. 76% for AF/AT, respectively) and in persistent AF (65% vs. 69% for AF, and 63% vs. 67% for AF/AT, respectively). Similarly, the time to the first recurrence after a single procedure and after redo procedures did not differ in both paroxysmal and persistent AF patients. It was highlighted that a number of patients from the HFSA groups (18 in the HFSA alone and 26 in the CPVI + HFSA) were spared from ablation due to safety concerns (HFS located inside LAA, and proximity to structures) and that additional ablation lines in the control group may have increased efficacy. Nonetheless, a high DF target strategy appeared no better than empiric PVI in both paroxysmal and persistent patients.
Kumagai et al 2013[17] studied 50 patients of which 18 exhibited procedural AF termination. Of those, 14 had PVI only, three had additional high DF ablation, and one had PVI + DF ablation + CFAE, which was the only persistent patient to achieve termination. Eight of the 14 paroxysmal patients demonstrating AF termination had high DF sites ablated during within the PVI circles and three patients at LA septum. Overall, 11 of 18 patients reached AF termination due to ablation in high DF sites in this study. Only DF sites ≥8 Hz were considered for ablation. In total, 31 high-DF sites were ablated in paroxysmal AF and 130 in persistent AF. Also, LA-to-RA DF gradient in patients with AF termination was not significant compared with patients without AF termination after the procedure.
Verma et al 2011[22] performed PVI after DF-guided ablation in 30 persistent AF patients based on retrospective observations of 20 AF patients group who underwent previous CFAE-based ablation and later compared with 30 case-matched patients from a control PVI group. Only two patients achieved AF termination with DF ablation alone and two others patients reached AF termination after additional PVI. The remaining 26 required cardioversion. After a mean of 12 ± 2 months, 17/30 patients (57%) from the DF + PVI group achieved AF freedom without drugs after the first procedure which was not significantly different from the control PVI group which achieved 60% AF freedom. On retrospective analysis of the CFAE-based group, AF termination occurred in regions where high DF (8-25 Hz) overlapped with CFAE (characterized by a mean cycle length of 40-120 ms). They observed that 89 ± 8% of the high DF regions overlapped with CFAE, however only 48 ± 27% of the CFAE areas overlapped with high DF regions.
In this systematic review, we examined the clinical use of DF in regard to catheter ablation for atrial fibrillation. No prior study has systematically reviewed this topic to evaluate the relevance of DF in the clinical field. Since the late nineties, experimental studies have speculated on the potential use of high DF sites for ablation[9], [25], [26] given the strong experimental association with AF perpetuation. The mechanism of persistent AF in humans remains elusive but recent data strongly supports maintenance by rotors with high DF[25]. From the analysis of the presented clinical articles, two major findings can be described: (1) measured DF appears to correlate with acute and chronic procedural outcomes. (2) Ablation on DF sites did not convincingly improve procedural success. Other findings include: (1) high mean DF across the LA at the beginning of ablation is associated with poor outcome, (2) decrease in DF value in CS and V1 during the procedure is associated with less recurrence, (3) reduction ≥11% in surface ECG derived DF during the ablation procedure is associated with freedom from AF recurrence in persistent AF, and (4) higher pre-ablation intra-LA DF gradient is predictive of long-term SR maintenance.
From the analyzed articles, it is clear that there is a correlation between DF and ablation outcome. Theoretically, high DF indicates the source of fibrillatory activity by recognizing the signal with a higher frequency compared with its surroundings[27], it appears to be associated with perpetuating rotors. It follows that a reduction in the fibrillatory activity should occur when this value reduces and observational studies appear to support this. Therefore, DF behaves as a marker of ablation success, with a reduction in both surface and intracardiac (LA and CS) measures indicative of better outcome. Some studies presented higher termination when ablating CFAE areas adjacent to high DF values[19], [21] this correlation between CFAE and DF indicates that the intraprocedural reduction of DF may present a useful marker of optimal ablation as an alternative to AF termination, although this potential has not been examined prospectively.
High DF sites as a target for ablation
The data presented by the second group of articles were somewhat contradictive. AAblating high DF sites prior to CPVI presented beneficial results for both paroxysmal and persistent patients in an early work by Atienza et al in 2009[16]; however, no such conclusion could be inferred from the RCT performed by the same group in 2014[15]. Supporting this, additional high DF-based ablation on PVAI of 30 persistent AF patients did not improve on outcome as indicated by Verma et al 2011[22]. Moreover, Kumagai et al in 2013[17] reported lower AF termination in patients that combined high DF-guided and CFAE ablation after PVI. The discrepancy of these results might be related to certain differences in studies characteristics. For high DF definition, two articles[15], [16] based their maximum DF value in comparison to its surroundings, therefore their inclusion criteria did not involve a previously defined value. While the other two[17], [22] defined a cutoff value of 8Hz for high DF ablation, this isolates the possibility of recognizing lower amplitude DF sites with the same influence on neighboring tissue that sites with DF ≥8Hz might be expected to have. The absence of a control group in three of the four studies[16], [17], [22] impairs the validation and comparison of their results. These four studies were quite heterogeneous in demographics, two studies had a mean population age of 55 ± 9 years[15], [16] and the other two of 64.8 ± 11.1 years[17], [22]. In addition, Verma et al had higher mean AF duration of 7.4 years[22] , higher than the 60.9 ± 67.6[17], 19.9 ± 11 months[16], and 4.13 years[15] years from the remaining three studies. The comparison of the results from those articles might not be justified since age and AF duration were associated with worse procedural outcomes[24].
Why does ablation of high DF sites not provide better results?
This apparent paradox might be explained by understanding the limitations of frequency analysis. First, a reduction in globally measured DF is associated with good clinical outcome but it does not follow that ablation of specific high DF sites results in the same. The dilemma of DF use lies on its variability due to changes in amplitudes, and morphology, which could be the effect of noise and far-field potentials[27]. Complexity of the signal morphology, as present in CFAE, and relative amplitude can affect the power spectrum and therefore the DF value, the morphology of discrete, narrow electrograms will have minimal impact on DF value, however low amplitude, broad CFAE electrograms will impair reproducibility of DF measurement and are less reliable[28]. Bipolar electrograms and signal duration ≥2 seconds are recommended to reduce those influences, as well as signal processing prior to fast Fourier transform (FFT), averaging multiple signals, and evaluation of power spectrum consistency[29]. Most studies follow those recommendations but vary considerably in the duration of signal examined and present data as a single value that is not representative of the time variant signal. For example, the two studies authored by Atenzia[15], [16] appear to show conflicting results, however the electroanatomic mapping system and processing of DF values were entirely different in the two studies and may go some way to explaining the discrepancy. Thus, targeting high DF is vulnerable to many confounding factors, not limited to signal processing issues but also bystander collision, pivot of wave fronts as well as meandering driver sources; indeed, it is quite conceivable that collision between two driver wave fronts will result in a higher DF value than either driver alone. However, it can be understood that if one driver is eliminated the overall DF drops due to a reduction in the collision and therefore reduction in global DF can quite readily be a marker of source reduction both directly and indirectly. Trying to target high DF may not yield the same result as both measurement error and confounding ablation of collision sites will not impact sources. In addition, in the case of meandering rotors, the shift in the observed frequency due to relative motion of the catheter and the rotor (Doppler Shift) can complicate the interpretation of high DF sites[30]. Even in the case of presence of a stationary rotor and neglecting the targeting error due to catheter navigation inaccuracies, the ablation at the pivoting center of the rotor may only decrease the rotation rate of the rotor, the required ablation strategy for rotors is unknown.
Put simply, these data indicate that DF can be used as a marker to predict outcome and recurrences; however, targeting this marker might not be the solution to increase ablation success with current signal processing techniques. High DF at specific site might be an indicator of a high frequency driving source, or it might be passive and just result of collision or overlap of different wave fronts[31].
Despite the disappointing results of targeting there remains good evidence that DF is a reflection of intraprocedural substrate change, as such it may provide a marker of the need for additional substrate ablation after PVI, and as such, a marker of optimal substrate ablation, avoiding proarrhythmia and complications associated with attempting to ablate until SR is achieved. Furthermore, contemporary signal processing techniques should be investigated to differentiate collision, nonstationary phenomena and anchored sources of AF perpetuation. Research using analysis of wavebreak and longer recording periods will attempt to resolve this issue to allow more specific targeting and avoid bystander regions. More work needs to be done to improve the accuracy of DF measurement and site differentiation.
Not all studies presented full representation of the atria, some presented only a specific region or ‘global’ metric from surface ECG or CS. Therefore, we could not provide a direct quantitative comparison of data and the results were provided in a qualitative manner. Restricted to the design of our study, no statistical calculation and summary from demographics were provided. However, important information was provided relevant for this review. No exclusion was done based on quality assessment, for this reason, the results and conclusion presented from the analyzed articles can be biased and may not represent the optimal situation. Some studies presented a small sample size[16]-[19], [21], [22] that reduces the external validity of their results. In addition, most studies used the same individual as a control for itself in a retrospective comparison[17]-[21], [23], [24] which also alters the validation of the results.
No prior study has systematically comprised information for clinical use of DF. Our systematic literature review supports DF as a useful marker of ablation outcome; however direct intervention targeting DF appears premature with mixed results and too few studies. Further research is warranted to explore this relationship Further research is warranted to explore this relationship and the development of more selective ablation procedures to avoid unnecessary complications and improve the outcome of targeted approach. Current DF methodology and targeting is insufficient to be used as a clinical tool but shows great promise as signal processing methods evolve.
The study design, data collection, analysis and interpretation and drafting of the article was developed by Lucas Gadenz with supervision and orientation from Dr. Javad Hashemi PhD and Dr. Damian P Redfearn MD. Dr. Mohammad Hassan Shariat PhD performed adjustments with addition of relevant information for the final draft. Dr. Damian P Redfearn MD and Dr Lorne Gula MD made direct contributions to the article’s final draft. Critical revision was performed by Lorne Gula MD and all authors. The article’s submitted version was approved by all authors.