Rescue leadless pacemaker implantation in a pacemaker-dependent patient with congenital heart disease and no alternative routes for pacing
Mohamed Sanhoury, Gaetano Fassini, Fabrizio Tundo, Massimo Moltrasio, Valentina Ribatti, Giuseppe Lumia, Flavia Nicoli, Elisabetta Mancini, Annalisa Filtz, Claudio Tondo
Cardiac Arrhythmia Research Centre, Department of Cardiovascular Sciences, University of Milan, Centro Cardiologico Monzino, IRCCS, Via Parea 4, Milan 20138, Italy.
Congenital heart disease patients are considered a unique group of patients regarding their high risk of conduction abnormalities , whether de novo or surgically induced , and the challenges in both implantation and management of device related complications. We present a case of a pacemaker-dependent patient with congenital heart disease who experienced complications of both previous epicardial and transvenous pacing which rendered her a non-suitable candidate of both routes.
Key Words : Congenital heart disease, Transvenous pacing, Epicardial pacing system, Leadless pacemaker.
Correspondence to: Mohamed Sanhoury , Email address: drsanhory@yahoo.com, Tel : +39 3290931694, Fax: +39 0258002782
Patients with congenital heart disease and symptomatic bradyarrhythmias requiring pacemaker implantation are among the most challenging clinical cases, owing to complex anatomy, frequently limited vascular access, higher risk of pacemaker related complications and risk of life-long pacemaker dependency.
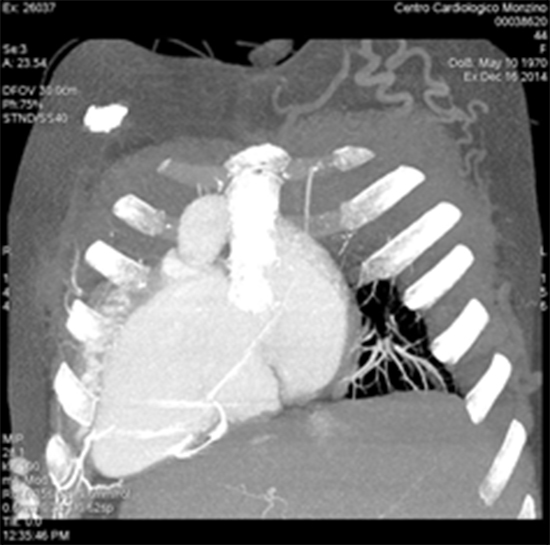
We report a case of a forty-seven years old lady with a history of congenital heart disease (Dextrocardia, situs inversus, double outlet right ventricle and ventricular septal defect). When she was 7 years old, a corrective surgical intervention was complicated by complete A-V block which necessitated the implantation of transvenous endocardial single chamber pacemaker. After the second replacement of the pacemaker generator in 1991, the patient had experienced pacemaker lead malfunction (progressive increase of pacing impedance and threshold) .Multidetector computed tomography (MDCT) revealed complete occlusion of the superior vena cava and innominate veins with extensive venous collaterals ([Figure 1]). The decision was taken to abandon the transvenous lead and implant an epicardial one (Medtronic Legend II) with the battery placed in the right upper abdominal quadrant ([Figure 2]).
Figure 1. MDCT chest reveals extensive collateral formation (arrow) form the venous occlusion
Over the following 10 years, elective generator replacement was done twice due to battery depletion. In 2014 , the patient presented with recurrent dizzy spells. Pacemaker interrogation revealed markedly elevated pacing threshold (4 V at 1.0 msec PW) with near End-Of-Life (EOL) . In the light of none available vascular access for regular transvenous pacing added to the failure of the epicardial pacing system , we decided to implant a leadless pacemaker (Medtronic Micra TM).
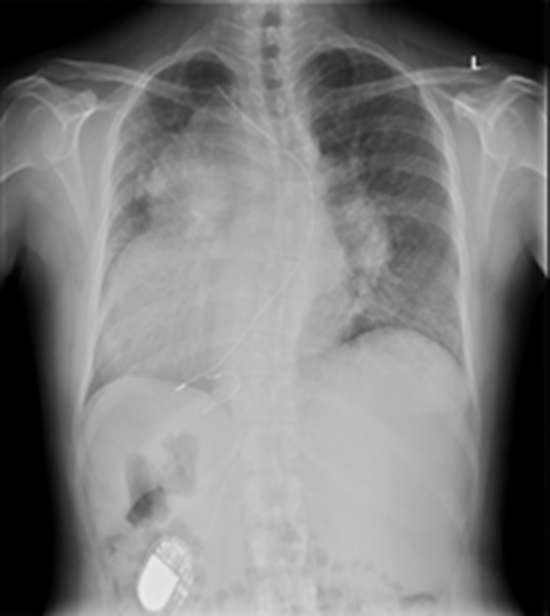
Figure 2. Pre-implantation plain chest X-ray PA view shows abandoned transvenous lead (asterisk) , the failed epicardial lead (arrow head) and pacemaker generator implanted in the right hypochondrium (arrow)
After getting a left femoral vein access and passing dilators of increasing size over a stiff wire, the Micra sheath was advanced to the junction of inferior vena cava (IVC) and right atrium. The device mounted on a steerable catheter was then advanced and manipulated to the right ventricular apical septal area. After confirming good contact with attaining the goose neck shape of the catheter and contrast material injection, the device was deployed and tine stability was confirmed by gentle pulling of the tether ([Figure 3]). Interrogation of the device revealed R wave amplitude of 8 mV and capture threshold of 1.0 V at 0.4 msec PW. The post-procedural course was uneventful with confirmed pacemaker position by chest X-ray ([Figure 4]) and good pacemaker parameters the day after the procedure.
Figure 3. Fluoroscopy images during implantation : (a) contrast injection to confirm the device contact with the ventricular wall, (b) gentle pull back of the catheter to confirm device stability, and (c)Final device position in the apical RV septum
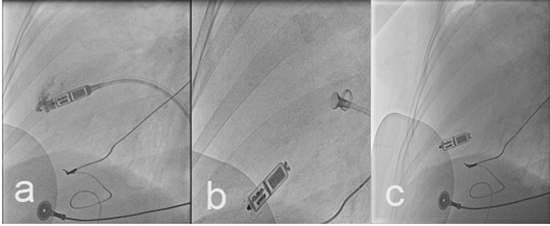
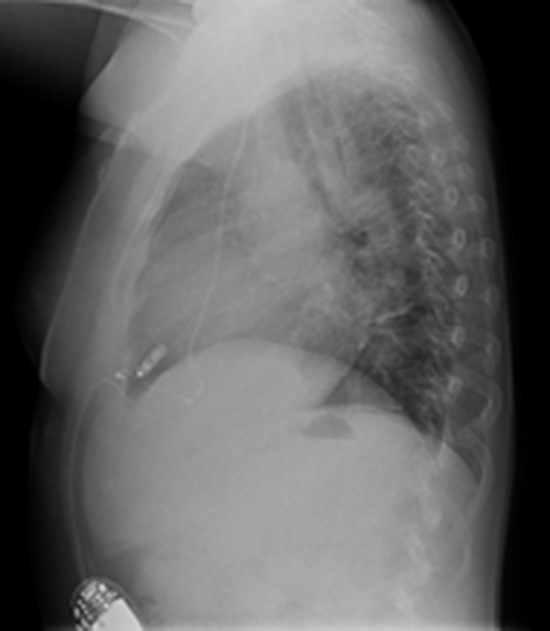
Figure 4. Post-implantation lateral CXR shows the final position of Micra leadless pacemaker (arrow) in the apical RV septum
Congenital heart disease (CHD) patients account for a small proportion of the patients requiring a pacemaker or defibrillator implantation. It was found that the overall long-term pacing- related complication rate in CHD was close to 40 %, compared with 5 % (or 0.5 % per year) in non-CHD. [1] Epicardial pacing system was associated with a higher lead failure rate as observed in our case. Endocardial pacing was found to be more durable but with multiple inherent risks as thromboembolic complications , reported to be more than 2 folds in patients with intracardiac shunts and transvenous endocardial leads [2], and the risk of venous occlusion ranging from 5-10% in long term follow-up studies. The case described in our report is challenging considering both the pacemaker-dependency in addition to absent any other remaining route for pacing lead implantation.
The non-surgical implantation of a small and self-contained single chamber leadless pacemaker mounted on a catheter and advanced through the femoral venous access was proved to be safe and effective by two recent published trials on the two commercially available leadless pacemaker systems. [3],[4] In our case, considering the bilateral venous access block and the failed epicardial pacing system, leadless pacemaker remained to be the only choice for the patient.
Leadless pacemaker implantation may be a reasonable strategy in patients with congenital heart disease and limited approaches for pacing. Some developments such as dual chamber and resynchronization leadless pacing systems as well as long term assessment of this modality in this unique patient population may be needed before being used on a large-scale.
Gaetano Fassini , Fabrizio Tundo and Massimo Moltrasio received consulting fees/honoraria from Medtronic, Inc. Claudio Tondo received consulting fees/honoraria from St. Jude Medical; Medtronic, Inc.; and Boston Scientific Corp.