Strategies to Improve Safety and Efficacy of Atrial Fibrillation Ablation Using Electrode Multipolar Phased RF PVAC™ Catheter: a Case Report
Fabio Dorfman, MD, Cristiano Dietrich, MD, PhD, Paulo Costa, MD, Evandro Sbaraini, MD, Rafael Abt, MD, Dalmo Moreira, MD, PhD, Cézar Mesas, MD, PhD*
Instituto de Cardiologia e Ritmologia de São Paulo, ICRESP, Mogi das Cruzes University, Mogi das Cruzes, Brazil.*Londrina State University, Londrina, Brazil.
Phased radiofrequency ablation with a single catheter technique, using a 9-electrode circumferential catheter, is a viable approach to pulmonary vein isolation for the treatment of atrial fibrillation. However, creating effective transmural lesions with such technique, while avoiding serious complications like atrioesophageal fistula, can be difficult. This case illustrates a challenging scenario, where catheter maneuvers fail to allow safe radiofrequency delivery, due to esophageal temperature rise, despite extensive navigating maneuvers. Changing the bipolar-to-unipolar ratio of energy delivery, from 2:1 to 4:1, allowed the creation of effective lesions, avoiding excessive increase in esophageal temperature.
Key Words : Multipolar, Phased, PVAC, Ablation.
Correspondence to: Cézar Eumann Mesas, Rua Espírito Santo, 1443, postal code 86020-420, Londrina - PR, Brazil.
Atrial fibrillation (AF) is the most common supraventricular tachyarrhythmia in clinical practice. Pulmonary veins (PVs) isolation is an effective treatment for recurrent, symptomatic, drug-refractory AF. The most widely used approach, with point-by-point ablation at the PV antrum, is a complex and time-consuming procedure. A single catheter technique, with phased radiofrequency (RF) delivery using a 9-electrode circumferential catheter (PVAC), has emerged as a viable alternative.[1] Despite the advantages of this approach, creating permanent, transmural lesions, while avoiding serious complications like atrioesophageal fistula, can be challenging.
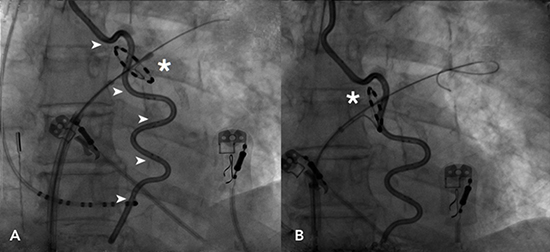
A 72-year-old female was referred to catheter ablation due to symptomatic paroxysmal AF, refractory to amiodarone. Her CHA2DS2VASc score was 3 and the procedure was performed under general anesthesia and therapeutic anticoagulation with warfarin (INR value ≥ 2). After a single venous femoral access, heparin was administrated (100U/kg bolus) before left atrium access, and continued throughout the procedure, to maintain ACT levels ≥350 s. A single transeptal puncture was performed, guided by fluoroscopy and intracardiac echocardiography. Luminal esophageal temperature (LET) was continuously monitored with a multi-sensor esophageal temperature probe (Circa Scientific, Park City, UT, USA). Electrical isolation was achieved in all four PVs using the multipolar circular ablation catheter PVACTM Gold and the GENius generator (Phased RF system, Medtronic Ablation Frontiers, Carlsbad, CA, USA). The RF generator delivered duty-cycled unipolar and bipolar energy between the selected electrodes pair, targeting each PV antrum, in a temperature-controlled, power-limited fashion (60°C, maximum 10 W). During ablation of the left superior PV, RF delivery was limited by consistent elevation of LET (35.7 to 38.5°C). After many attempts of repositioning the catheter (Fig.1), complete isolation was achieved by increasing the bipolar-to-unipolar ratio of energy from 2:1 to 4:1 at the electrode pair associated with LET rise, which remained below 37.8°C throughout ablation.
Figure 1. Fluoroscopy shows the PVAC catheter at the left superior PV antrum (asterisk), in two different positions and orientations (A and B). After several maneuvers (rotating, pulling and pushing, sliding), RF delivery still caused consistent elevation of esophageal temperature, up to 38.5°C, monitored by the multi-sensor esophageal temperature probe (arrowheads)
This case illustrates a challenging situation, common to most approaches to AF ablation, but with aspects specific to this new technology. Although there is still a debate regarding its utility to avoid esophageal damage, monitoring of LET is used by most electrophysiologist to titrate RF energy or reduce delivery duration. We used a multi-sensor esophageal temperature probe, that enhances esophageal coverage with 12-point temperature sensing.[2]
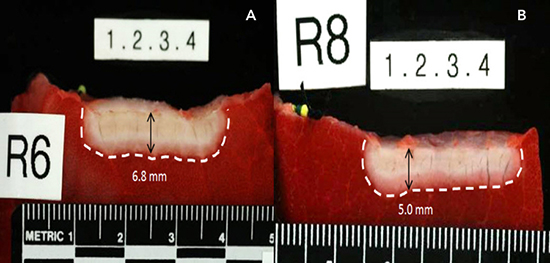
Once an area of LET increase is identified, maneuvers to navigate the PVAC aiming to find safer positions (steering, rotating, pulling and pushing, sliding) are tried first. If this fails, changing the energy settings by delivering RF in different bipolar-to-unipolar ratio, can reduce the lesion depth while assuring transmural lesion (fig. 2). This can be explained by the fact that unipolar RF current flows from the electrode to the reference patch, causing deeper lesions, while bipolar current flows between two adjacent electrodes, creating more superficial lesions.[3,4] In our case, changing the ratio from 2:1 to 4:1 enabled us to safely isolate the left superior PV.
Figure 2. Different lesion depth on porcine thigh muscle preparation with energy delivered on a 2:1 bipolar-to-unipolar ratio (A) and 4:1 ratio (B). Courtesy by Medtronic
Multipolar phased PVAC catheter ablation is a feasible and safe option for PV isolation. Monitoring of esophageal temperature is important to minimize the risk of esophageal injury. When catheter maneuvers fail to allow safe RF delivery due to esophageal temperature rise, changing the bipolar-to-unipolar ratio to 4:1 can influence the depth of lesions, enabling the creation of more superficial yet effective lesions.