Inappropriate dosing of direct oral anticoagulants in patients with atrial fibrillation
Katy Lavoie1, Marie-Hélène Turgeon2, Caroline Brais3, Josiane Larochelle4, Lucie Blais5,6, Paul Farand1,7, Geneviève Letemplier1,7, Sylvie Perreault5, Marie-France Beauchesne1,5,7,8
1Faculté de médecine et des sciences de la santé, Université de Sherbrooke, Sherbrooke, Québec, Canada.2Centre hospitalier universitaire de Montréal, Montréal, Canada.3Hôpital du Haut-Richelieu, Saint-Jean-sur-Richelieu, Québec, Canada.4Institut universitaire de gériatrie de Sherbrooke, Sherbrooke, Québec, Canada.5Faculté de pharmacie, Université de Montréal, Montréal, Québec, Canada.6Centre de recherche, Hôpital du Sacré-Cœur de Montréal, Montréal, Québec, Canada.7Centre hospitalier universitaire de Sherbrooke, Sherbrooke, Québec, Canada.8Centre de recherche, Centre hospitalier universitaire de Sherbrooke, Sherbrooke, Québec, Canada.
Atrial fibrillation (AF) is a common cardiovascular disease for which newer oral anticoagulants are available. The main objective of this study was to evaluate the appropriateness in prescriptions of direct oral anticoagulant (DOACs), more specifically apixaban, dabigatran and rivaroxaban. This was a singlecentre, retrospective study conducted in the province of Quebec, Canada. Adult subjects hospitalized between October 2011 and October 2014, with a diagnosis of AF, and a DOAC prescription were included. Data were retrieved from the electronic medical records and prescriptions were evaluated according to appropriateness criteria. A total of 500 subjects were included (235 subjects on dabigatran,222 on rivaroxaban and 43 on apixaban). Overall, 70.4% (95% confidence interval [Cl] 66.4–74.1) of DOAC prescriptions were considered appropriate. About 24% of subjects received an inappropriate dose of apixaban, dabigatran or rivaroxaban. A reduced dose was prescribed in 56.8% of subjects with no clearindication, and 43.2% received a dose that was not reduced while indicated. DOACs were frequentlyprescribed at a dose that was considered inappropriate. There is a need to strengthen dosing recommendations of DOACs in clinical practice.
Key Words : Atrial fibrillation, direct oral anticoagulants, apixaban, dabigatran, rivaroxaban.
Correspondence to: Marie-France Beauchesne
Atrial fibrillation (AF) is the most common type of cardiac arrhythmia. AF affects between 1% and 2% of the population and its prevalence increases with age [1][2]. AF is associated with a three to five-fold increased risk of ischemic stroke, and anticoagulants are effective in preventing thromboembolic events [3][4].
Vitamin k antagonists (warfarin) have mainly been used in AF management, but direct oral anticoagulants (DOACs) such as dabigatran, rivaroxaban and apixaban are increasingly used. DOACs have a rapid onset of action, few food and drug interactions, and blood monitoring is required less frequently when compared with warfarin. However, DOACs are contraindicated in patients with severe renal failure (creatinine clearance [CrCl] <25-30 mL/min), and not all have antidotes to reverse their effect pulation and its prevalence increases with age [5][6].
Selecting the appropriate dosing of DOACs is important to prevent thromboembolic events and reduce the risk of bleeding. Warfarin has been used for several years and its efficacy and long-term safety are well known [5]. However, only a few real-world studies on DOACs have been published [7]-[14]. Short-term studies have confirmed the efficacy and safety of DOACs in AF [15]-[17], but it is essential to evaluate their appropriateness in use. Thus, the main objective of this study was to evaluate overall appropriateness in prescriptions of DOACs in adults with AF.
Study design and study population
A retrospective cohort study was conducted in a single centre (Centre hospitalier universitaire de Sherbrooke, Quebec, Canada). The study population included subjects hospitalized between October 2011 and October 2014, with a diagnosis of AF documented on the discharge summary. Subjects were identified using ICD-10 codes (International Statistical Classification of Diseases and Related Health Problems - 10th revision) for the diagnosis of AF (I48X, I48.XN-001 and I48.XN-002). Medical records were reviewed to determine eligibility (inclusion and exclusion criteria are listed in ([Table 1]). Only the first hospitalization following October 1th 2011, was selected.
Table 1. Inclusion and exclusion criteria
|
Inclusion criteria
|
Exclusion criteria
|
|
At least 18 years of age at the time of hospitalization
Diagnosis of AF documented on the discharge summary
DOAC prescribed during hospitalization and at discharge
Availability of a discharge prescription and the list of usual medications
|
AF associated with: congenital heart disease, uncontrolled hyperthyroidism, illicit drug or drug intoxication
Another indication for anticoagulation such as deep vein thrombosis, pulmonary embolism, orthopedic surgery or hypercoagulable state
AF with a duration of less than 48 hours
Peri-operative AF
|
Data Collection
Electronic medical records were reviewed, more specifically the discharge summary, laboratory data (values that were closest to DOAC prescription), and discharge prescriptions. Data collected for each subject were sociodemographic characteristics (age, gender, weight, height and body mass index), serum creatinine (to estimate renal function using the Cockcroft-Gault equation), liver enzymes, comorbidities, length of stay, healthcare unit of admission, concomitant medications that are contraindicated with DOACs (ketoconazole, itraconazole, voriconazole, posoconazole and ritonavir) or known to increase bleeding risks (antiplatelets and nonsteroidal anti-inflammatory drugs (NSAIDS). Valvular AF was determined when the diagnosis was specified on the discharge summary or in the presence of mitral or tricuspid valve replacement or repair.
Appropriateness in Prescriptions
The appropriateness criteria were developed and reviewed by the anticoagulotherapy committee of our institution. The criteria were mainly based on DOACs product monographs along with the 2012 update of Canadian Society of Cardiology (CSC) guidelines [18]-[22]. The appropriateness criteria used are presented in [Table 2]. A total of 8 appropriateness criteria were evaluated for each subject included in the study.
Table 2. Appropriateness criteria for DOACs prescriptions
|
Criteria
|
|
1. Type of AF
DOACs are prescribed in patients with non-valvular AF (i.e. mitral or tricuspid valve replacement or repair) [18].
|
|
2. Mitral stenosis
In the case of mitral stenosis, DOACS are prescribed in patients with mild to moderate mitral stenosis only (not moderate to severe) [18].
|
|
3. CHADS2 score
When DOACs are prescribed, the CHADS2 score is > 1.
If the CHADS2 score = 0, the CHA2DS2-VASc score must be > 2 or there has been a cardioversion [18].
|
|
4. Heart valves
In the case of heart valves, DOACs are used in patients with a bioprosthetic valve (and not a mechanical valve) [19].
|
|
5. Concomitant medication
DOACs are prescribed in patients that do not use ketoconazole, itraconazole, voriconazole, posaconazole or ritonavir [5, 20-22].
|
|
6. Renal function
Dabigatran and rivaroxaban are prescribed when the estimated CrCl is > 30 mL/min [5, 21, 22].
Apixaban is prescribed when CrCl is > 25 mL/min [20].
|
|
7. Liver enzyme levels
DOACs are prescribed if blood levels of AST and ALT are < twice the upper limit of normal [5].
|
|
8. a. Dose of dabigatran
Dabigatran is prescribed at a reduced dose of 110 mg twice daily if one of the following factors is present:
Weight < 50 kg
Age > 80 years old
Haemorrhagic risk factors: HAS-BLED score > 3 and/or CrCl is between 30 and 49 mL/min [5, 21].
|
|
8. b. Dose of rivaroxaban
Rivaroxaban is prescribed at a reduced dose of 15 mg once daily if CrCl is between 30 and 49 mL/min [5, 22].
|
|
8. c. Dose of apixaban
Apixaban is prescribed at a reduced dose of 2.5 mg twice daily when 2 of the 3 following criteria are present:
Serum creatinine > 133 mol/L
Age > 80 years old
Weight <60kg [5, 20].
|
Legend: CrCl, creatinine clearance; CHADS2: Congestive Heart Failure, Hypertension, Age ≥ 75 years, Diabetes, Stroke; CHA2DS2-VASc: Congestive Heart Failure, Hypertension, Age ≥ 75 years old, Diabetes, Stroke history, Vascular disease, Age between 65 and 74 years, Sex Category; HAS-BLED: Hypertension, Abnormal renal and/or liver function, Stroke, Bleeding, Labile INR, Elderly, Drugs.
Statistical Analyses
Baseline characteristics were described estimating means, medians, standard deviations, ranges and proportions with 95% confidence intervals (CI). The proportion (with 95% CI) of prescriptions that were in accordance with each of the 8 appropriateness criteria was initially calculated. Then, the proportion (with 95% CI) of prescriptions that were in accordance with all appropriateness criteria for a given DOAC was estimated. Statistical analyses were performed using the IBM SPSS Statistics software.
Ethical Considerations
This project was approved by the ethics committee of our institution. Data were kept confidential using a denominalized database.
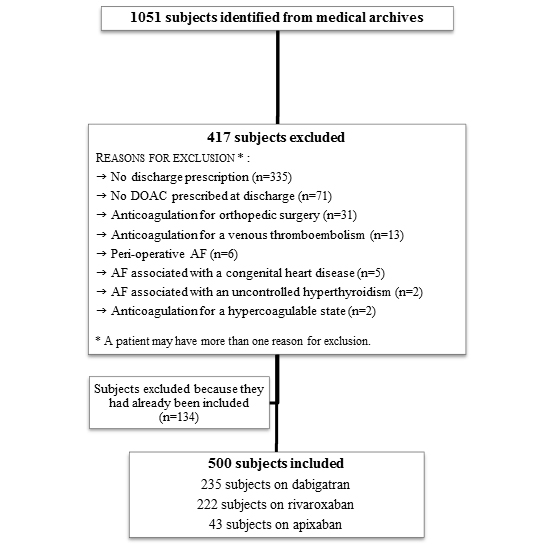
A total of 1051 subjects were initially screened for their eligibility, and 551 were excluded [Figure 1.]. Most of them had an exclusion criterion (n=417) and 134 were previously selected (more than one hospitalization). The main reasons for exclusion were the lack of a discharge prescription (80.3%), DOAC not prescribed at discharge (17.0%) and anticoagulation for an orthopedic surgery (7.4%). Thus, 500 subjects were included, 235 on dabigatran, 222 on rivaroxaban and 43 on apixaban. More patients were on dabigatran and rivaroxaban because they were introduced earlier (October 2011, and March 2013, respectively), than apixaban (January 2014) on the therapeutic formulary. Excluded and included subjects had similar baseline characteristics [].
[Table 3] presents sociodemographic characteristics, hospitalization data, laboratory values, comorbidities, and concomitant drugs. Data on compliance related to each appropriateness criteria are found in [Table 4.]). Subjects were 74 years old on average, and about half were males (52.4%). Most subjects (71%) had a body mass index (BMI) greater than 25.0 kg/m2. Subjects were mainly hospitalized at the cardiology unit (41.0%), followed by internal medicine (21.4%), family medicine (17.0%), neurology (8.6%) and respiratory medicine units (3.4%). Moreover, 63.4% of subjects were previously anticoagulated.
Figure 1. Flow chart for subjects selection.
Supplementary Table 1. Supplementary Table 1
|
Characteristics
|
|
Patients included
(n=500)
n (%) *
|
Patients excluded
(n=417)
n (%) *
|
|
Age (years), mean +- SD
|
|
74.3 +- 11.5
|
70.5 +- 13.5
|
|
Male gender
|
|
262 (52.4)
|
258 (61.9)
|
|
|
|
|
|
|
Hospitalization data
|
|
|
|
|
Length of stay, median; range (days)
|
|
5 ; 1 - 70
|
4 ; 1 - 85
|
|
Care units
|
Cardiology
|
205 (41.0)
|
205 (49.2)
|
|
|
Internal medicine
|
107 (21.4)
|
35 (8.4)
|
|
|
Family medicine
|
85 (17.0)
|
16 (3.8)
|
|
|
Neurology
|
43 (8.6)
|
31 (7.4)
|
|
|
Surgery
|
22 (4.4)
|
37 (8.9)
|
|
|
Respirology
|
17 (3.4)
|
15 (3.6)
|
|
|
Orthopedics
|
1 (0.2)
|
47 (11.3)
|
|
|
Others
|
20 (4.0) 1
|
31 (7.4) 2
|
|
|
|
|
|
|
Drugs
|
|
|
|
|
DOACs user,s type
|
Prevalent
|
317 (63.4)
|
254 (60.9)
|
|
|
Incident
|
148 (29.6)
|
138 (33.1)
|
|
|
Switch
|
35 (7.0)
|
25 (6.0)
|
Legend:* Data are presented as a proportion, unless otherwise noted.¹ Other care units: gastroenterology, nephrology, geriatrics, hematology, ORL, urology, rheumatology, psychiatry and palliative care.² Other care units: geriatrics, gastroenterology, ORL, urology, hematology, neurosurgery, plastic surgery, endocrinology, gynecology, psychiatry, rheumatology and palliative care.
Overall, 70.4% (95%CI 66.4–74.4) of subjects were prescribed a DOAC in accordance with all criteria. A high proportion of subjects (94.6%) had a thromboembolic score (CHADS2 score) equal to or greater than 1. In patients who had a CHADS2 score of 0 (n=27), 13 had a CHA2DS2-VA2Sc score of at least to 2 and/or a cardioversion, and 6 had a surgical ablation. About 3% of patients had valvular AF and 1.0%, moderate to severe mitral stenosis. No patients had a mechanical heart valve, but 2.6%, a bioprosthetic heart valve. Similarly, no patients were prescribed a medication that is contraindicated with DOACs.
Table 3. Baseline characteristics of included patients (n=500)
|
Characteristics
|
|
n (%) *
|
|
Age (years), mean +- SD
|
|
74.3 +- 11.5
|
|
Male gender
|
|
262 (52.4)
|
|
BMI (kg/m2)o
|
< 18.5
|
12 (2.4)
|
|
|
18.5 - 24.9
|
132 (26.6)
|
|
|
25.0 - 29.9
|
164 (33.0)
|
|
|
> 30.0
|
189 (38.0)
|
|
Hospitalization data
|
|
|
|
Length of stay, median; range (days)
|
|
5; 1 - 70
|
|
Health Care units
|
Cardiology
|
205 (41.0)
|
|
|
Internal medicine
|
107 (21.4)
|
|
|
Family medicine
|
85 (17.0)
|
|
|
Neurology
|
43 (8.6)
|
|
|
Surgery
|
22 (4.4)
|
|
|
Respirology
|
17 (3.4)
|
|
|
Others1
|
21 (4.2)
|
|
Laboratory data
|
|
|
|
CrCl (CG), median; range (mL/min) o
|
|
63.9; 20.4 - 120.0
|
|
CrCl < 30 mL/min
|
|
11 (2.2)
|
|
Level of liver enzymes o
|
AST, median ; range (U/L)
|
21; 8-155
|
|
|
ALT, median ; range (U/L)
|
17; 5-529
|
|
Clinical data
|
|
|
|
Atrial flutter
|
|
79 (15.8)
|
|
Valvular AF
|
|
13 (2.6)
|
|
Mitral stenosis
|
Mild to moderate
|
25 (5.0)
|
|
|
Moderate to severe
|
5 (1.0)
|
|
Biological heart valve
|
|
13 (2.6)
|
|
Congestive heart failure
|
|
183 (36.6)
|
|
Hypertension
|
|
397 (79.4)
|
|
Diabetes
|
|
157 (31.4)
|
|
Stroke
|
|
139 (27.8)
|
|
Atherosclerotic heart disease
|
|
212 (42.4)
|
|
Atherosclerotic vascular disease
|
|
84 (16.8)
|
|
Cirrhosis
|
|
5 (5.2)
|
|
CHADS2 > 1
|
|
473 (94.6)
|
|
CHADS2-VA2Sc > 2
|
|
455 (91.0)
|
|
|
|
|
|
Drugs
|
|
|
|
Number of drugs at discharge, median ; range
|
|
10 ; 2 - 8
|
|
Antiplatelets
|
|
82 (16.4)
|
|
NSAIDs
|
|
30 (6.0)
|
|
DOACs user,s type
|
Prevalent
|
317 (63.4)
|
|
|
Incident
|
148 (29.6)
|
|
|
Switch
|
35 (7.0)
|
Legend: BMI, body mass index; CrCl, creatinine clearance; CG, Cockroft-Gault; NSAIDs, nonsteroidal anti-inflammatory drugs.* Data are presented as a proportion, unless otherwise noted.º Presence of patients with missing data: 3 for BMI and 93 for liver enzymes.¹ Other care units: gastroenterology, nephrology, geriatrics, hematology, ORL, orthopedics, urology, rheumatology, psychiatry and palliative care
Table 4. Appropriateness of DOACs prescriptions
|
Criteria
|
|
Appropriateness, n (%)
|
95% CI
|
|
1. Type of AF
|
Appropriate use
|
487/500 (97.4)
|
96.0 - 98.8
|
|
2. Mitral stenosis
|
Appropriate use
|
495/500 (99.0)
|
98.1 - 99.9
|
|
3. CHADS2 score
CHADS2 > 1
CHADS2 = 0
|
Appropriate use
Appropriate use
Total number of appropriate use
|
473/473 (100.0)
13/27 (48.1)
486/500 (97.2)
|
95.8 - 98.7
|
|
4. Heart valves
|
Appropriate use
|
500/500 (100.0)
|
|
|
5. Concomitant medication
|
Appropriate use
|
500/500 (100.0)
|
|
|
6. Renal function
|
|
|
|
|
Dabigatran and rivaroxaban
Apixaban
|
Appropriate use
Appropriate use
Total number of appropriate use
|
446/457 (97.6)
43/43 (100.0)
489/500 (97.8)
|
96.5 - 99.0
|
|
7. Liver enzymes levels
|
Appropriate use
|
395/407 (97.1)
|
95.4 - 98.7
|
|
8. a. Dose of dabigatran
|
|
|
|
|
Dabigatran 110 mg BID
Dabigatran 150 mg BID
|
Appropriate use
Appropriate use
Total number of appropriate use
|
106/130 (81.5)
67/105 (63.8)
173/235 (73.6)
|
67.9 - 79.3
|
|
8. b. Dose of rivaroxaban
|
|
|
|
|
Rivaroxaban 15 mg DIE
Rivaroxaban 20 mg DIE
|
Appropriate use
Appropriate use
Total number of appropriate use
|
31/61 (50.8)
148/161 (91.9)
179/222 (80.6)
|
75.4 - 85.9
|
|
8. c. Dose of apixaban
Apixaban 2,5 mg BID
Apixaban 5 mg BID
|
Appropriate use
Appropriate use
Total number of appropriate use
|
7/20 (35.0)
23/23 (100.0)
30/43 (69.8)
|
55.5 - 84.1
|
|
Overall appropriateness
|
Appropriate use
|
352/500 (70.4)
|
66.4 - 74.4
|
Legend:C-Gault, Cockcroft-Gault; GFR, glomerular filtration rate; AST, aspartate aminotransferase; ALT, alanine aminotransferase; PO, per os; BID, twice a day; QD, once a day
Most subjects on dabigatran or rivaroxaban had an estimated creatinine clearance (CrCl) greater than 30 mL/min (97.8%), and all patients on apixaban had an estimated CrCl greater than 25 mL/min. Liver enzymes values were available for 81.4% of patients, and 97.1% of them had values of AST (aspartate aminotransferase) and ALT (alanine aminotransferase) lower than twice the upper limit of normal.
About a quarter of subjects were prescribed a dosing of dabigatran that was considered inappropriate: 38.7% were prescribed 110 mg without having a characteristic justifying a reduced dose, while 61.3% were prescribed 150 mg when a lower dose was indicated.
For subjects on rivaroxaban, 19.4% were prescribed a dose that was considered inappropriate. About 70% were prescribed a dose of 15 mg while having an estimated CrCl greater than 49 mL/min and 30.2% had an estimated CrCl between 30-49mL/min and were prescribed 20mg. For patients on apixaban, 30.2% received a dose of 2.5 mg without having two characteristics justifying this reduced dose.
DOACs were frequently prescribed at a reduced dose in adults with AF. Up to a third of patients were prescribed a dose of dabigatran that was considered inappropriate. Proportions of inappropriate dosing (without specification for under or overdosing) of dabigatran (14.4%) and rivaroxaban (35.4%) has been reported [7] [13]. A Danish study reported that only 55.5% of subjects were prescribed 150 mg of dabigatran while indicated (a lower dose was used) [14]. Larock et al. reported that the dosage of dabigatran was reduced in 10.1% of subjects with no clear indication, while 4.3% of subjects had a regular dose when a reduction was indicated [12]. Finally, up to a third of subjects were prescribed a dose of apixaban that was considered inappropriate, but some clinicians might reduce the dose in the presence of only one justification factor.
We found a lower proportion of subjects with valvular disease (2.6%) than the ones reported by Larock et al. (7.2%)[12] and Carley et al. (10.9%) [8], but it was similar to the one found by Armbruster et al. (2.8%) [7]. DOACs were rarely prescribed in subjects with moderate to severe mitral stenosis (1.0%), which was also described by Tellor et al. (0.8% of patients with moderate to severe mitral stenosis received rivaroxaban) [13], and Armbruster et al. (0.2% of patients with moderate to severe mitral stenosis received a dose of dabigatran) [7].
Most of our patients had a CHADS2 score greater than or equal to one. At the time of the study, we considered the prescription of DOACs in patients who were 65 years and over with a CHADS2 score of 0 inappropriate (this represents 8 patients), while it is no longer the case[23]
In our study, no subjects with a mechanical heart valve were prescribed a DOAC. In fact, dabigatran was found to increase the risks of thromboembolic and bleeding events in such patients [24].
No subjects received a concomitant medication that is contraindicated with DOACs. In a study conducted in Denmark, one patient (0.02%) was taking ketoconazole or itraconazole at the beginning of treatment with dabigatran, and six patients (0.1%) were prescribed itraconazole while on dabigatran [9].
A low proportion of subjects (2.2%) received a DOAC while having an estimated CrCl lower than 30 mL/min. Previous studies have also reported minimal use of dabigatran in patients with this level of renal function, with proportions varying between 0.4% and 2.9% [7],[10]-[13].
About 20% of subjects were prescribed a DOAC with no recent values of liver enzymes available . This is of concern since DOACs are not recommended in the presence of severe hepatic impairment.
Our study is limited by several factors; a few numbers of patients on apixaban were included, several subjects were excluded because of incomplete discharge prescriptions (but their baseline characteristics were similar to the ones of included subjects), it was conducted in a single centre, and data were collected retrospectively. Also, the appropriateness criteria are based on limited evidence. Furthermore, factors influencing dose selection may not have been documented in the medical chart. DOACs are increasingly prescribed and physicians are now more familiar with their use, which could influence results from future studies on this subject.
Dabigatran, rivaroxaban and apixaban were commonly prescribed with a dose that was considered inappropriate. Our results support the need to implement measures to promote appropriate dosing of DAOCs. Further studies should investigate underdosing of DOACs in a larger setting.
We acknowledge the contribution of Martin Turgeon and the anticoagulotherapy committee of the Centre hospitalier universitaire de Sherbrooke for reviewing our appropriateness criteria.
This study was funded by Bayer, Boehringer Ingelheim and Bristol-Myers Squibb/Pfizer alliance.
MFB received funding from Bayer, Boehringer Ingelheim and Bristol-Myers Squibb/Pfizer alliance for investigator initiated research projects. Besides the source of funding for this study, all other authors have no conflicts of interest to declare, in relation with this issues discussed in this manuscript.