Emergency Covered Stent Implantation For Rupture Of A Pulmonary Vein After Balloon Angioplasty For Postinterventional Pulmonary Vein Stenosis
Sebastian Hilbert, MD1, Silke John, MD1, Frank-Thomas Riede, MD2, Arash Arya, MD1, Ingo Paetsch, MD1, Cosima Jahnke, MD1, Gerhard Hindricks, MD1, Andreas Bollmann, MD, PhD1
1Department of Electrophysiology, University Leipzig - Heart Center, Leipzig, Germany.2Department of Pediatric Cardiology, University Leipzig - Heart Center, Leipzig, Germany.
Pulmonary vein (PV) stenosis is a known complication of PV isolation procedures for atrial fibrillation. We describe in this report a case of emergency covered stent implantation for rupture of a PV after balloon angioplasty for postinterventional PV stenosis occlusion. Focus is on stent implantation and on a novel aspect of magnetic resonance imaging for postprocedural outcome evaluation. A focused review of the current literature regarding ongoing limitations of PV stenosis treatment is provided.
Key Words : Pulmonary Vein Stenosis, Balloon Angioplasty, Pulmonary, Vein Rupture, Pericardial Effusion, Covered Stent, MR Angiography.
Correspondence to: Sebastian Hilbert, Department of Electrophysiology, Heart Center Leipzig, Strümpellstr. 39, 04289 Leipzig, Germany.
With the rise of radiofrequency ablation for atrial fibrillation pulmonary vein stenosis (PVS) has become an important complication to consider. Event rates vary considerably depending on post-procedural screening and pulmonary vein isolation (PVI) technique. In recent publications event rates were reported to be as low as 0.5-2%.[1,2] Historically treatment has often been associated with less than desirable outcome. More recently an interventional approach has been described[3,4] which caters for some but not all procedural difficulties. Furthermore there are some complications, which can occur during dilatation of the pulmonary vein. Here we describe one of these complications, rupture of the pulmonary vein, the management of this problem by stenting and considerations for follow-up by CMR angiography.
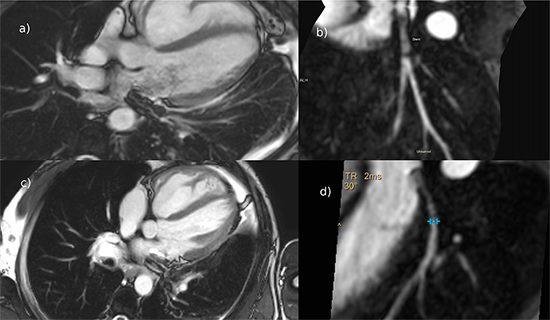
A 43 year old male patient was referred to our hospital for balloon angioplasty (BPVA) of a recurrent stenosis of the left inferior pulmonary vein (LIPV) stenosis as diagnosed by MR angiography (Figure 1a-b). He had undergone a BPVA procedure 3 month previously for pulmonary vein stenosis after pulmonary vein isolation for atrial fibrillation. The patient was symptomatic with severe dyspnea on exertion and hemoptysis. Angiography of the LIPV following fluoroscopy-guided transseptal puncture and placement of a steerable sheath (Agilis, St. Jude Medical) confirmed subtotal stenosis. We then performed balloon dilatation (6x20mm balloon at 16atm for 20 seconds and 8x20mm balloon at 14atm for 14 seconds, Maverick, Boston Scientific). Shortly thereafter, the patient developed acute pericardial tamponade (Video 1) due to rupture of the LIPV (Figure 2a,d; Video 2). Pericardial effusion was confirmed by echocardiography and treated by pericardial drainage. The patient stabilized under continuous autotransfusion and catecholamine support. A covered stent (Advanta V12 Atrium 10x38mm) was deployed (Figure 2c,f) in the LIPV. Subsequent angiography showed no further contrast extravasation (Video 3). Postinterventional course was uneventful. MR angiography after 2 days and six weeks confirmed a patent LIPV. The implanted stent type resulted in artifacts on CMR cine-imaging (SSFP sequence, Figure 3a,c) but allowed evaluation of stent patency on contrast-enhanced CMR angiography (Figure 1c, Figure 3b,d).[5]
Figure 1: CMR-angiography before and after the procedure. Before the procedure the left inferior and right inferior pulmonary vein show very late filling (a). Realtime bolus tracking of the contrast medium (b) demonstrated significantly earlier filling of the right compared to the left lung vessels/parenchyma (bright) thereby indicating limited drainage via the left inferior pulmonary vein. After stenting (day 2: c,d; after six weeks: e,f) the left and right inferior vein fill simultaneously indicating improved lung perfusion/drainage. Note the lack of perfusion of the left upper lobe resulting from a chronically occluded LSPV. The covered stent in the left inferior pulmonary vein is clearly visible (c,e)
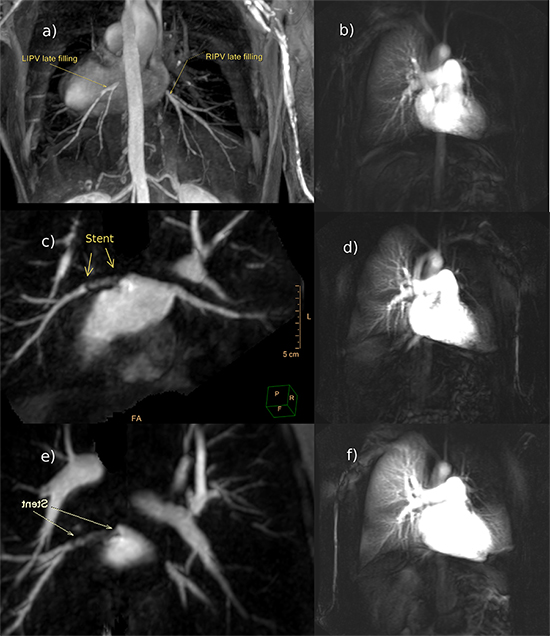
Figure 2. Left anterior oblique (a-c) and right anterior oblique projection with caudal angulation (d-f)) Selective angiography of the left inferior pulmonary vein (LIPV) revealing contrast extravasation at the site of pulmonary vein rupture immediately after balloon dilatation (a, d). Implantation of a stent (b, e) and final stent position (c, f) are shown. Also note pericardial drainage (e, f)
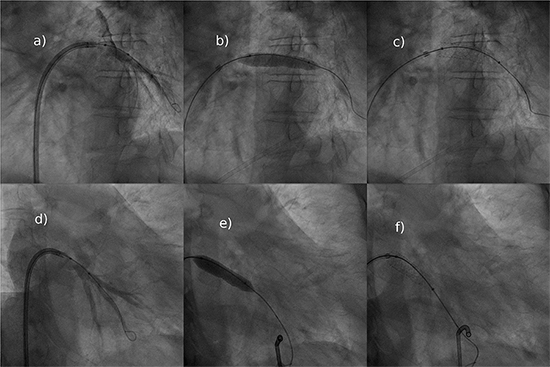
Figure 3. Cardiac magnetic resonance imaging on day 2 after stent placement (a-b) and during six-weeks-follow-up (c-d): imaging artefacts related to the covered stent of the left inferior pulmonary vein vary with the imaging sequence used and may allow for only limited in stent lumen visibility (SSFP-cine sequence, a, c). On contrast enhanced CMR angiography however, luminal visibility was improved and together with depiction of peripheral pulmonary vein branches revealed a vastly patent stent at follow up (day 2, b; six weeks after the procedure, d)
For prevention of stent-thrombosis and stroke the patient received dual antiplatelet therapy (clopidogrel 75mg and aspirin 100mg o.p.d.) and novel oral anticoagulant (apixaban 2,5mg o.p.d).
Six weeks after the procedure CMR angiography revealed a patent stent and the patient had markedly improved (Video 4).
PV stenosis is an increasing problem and rupture of PVs during BPVA may result in potentially lethal bleeding and hence, interventionalists must be prepared to handle such an emergency situation. Balloon angioplasty for treatment of PVS with or without stent placement for prevention of restenosis[6] as well as covered stent implantation for rupture of the ascending aorta after balloon angioplasty[7] has been reported. However, currently no data regarding the use of covered stents in the setting of pulmonary vein rupture are available. Treatment of PV rupture with a covered stent not only provides a treatment option for the complication but can potentially help maintain vessel patency. However stent availability is limited and point of care availability of covered stents with the correct dimensions can decide the fate of a patient.
No standard treatment for PVS exists. Especially subtotal or total occlusions are difficult to manage. Balloon dilatation - alone or in combination with stent placement - has evolved as recommended treatment. There is few data for stents (bare-metal or drug-eluting) regarding the safety and the potential to withstand restenosis. In the low flow system of the PVs stents smaller then 1cm seem to be at higher risk of occlusion.[6] There is no hard evidence that would support a preference of DES in this setting. However experimental data shows that PTA with a drug eluting balloon immediately following intentional ablation of the PV ostia reduces PV stenosis compared to ablation without DE-balloon treatment. Clearly randomized trials are needed. Besides the high incidence of unfavorable outcomes (e.g. restenosis or recurring occlusion) one has to keep in mind that these procedures are associated with a significant risk of procedural complications. Stent placement can result in stent thrombosis which potentially leads to a stroke. Anticoagulation regimens in this setting are a constant matter of debate.
CMR-based follow-up to detect restenosis or occlusion is recommended over CT-angiography or even direct fluoroscopic evaluation. CMR angiography is less invasive and contrast media is usually well tolerated even in patients with impaired renal function. Careful stent selection is important as imaging artifacts caused by stents made from stainless steel tend to obscure the stent lumen. Stents made from tantalum or nitinol usually allow for the evaluation of the stent lumen.[5] As observed in our case not only stent material but also imaging sequence plays a role in stent visibility.
Several viable options for the treatment of pulmonary vein stenosis exist. In the event of periprocedural PV rupture placement of a covered stent has the potential to avoid cardiac surgery. Exact knowledge of the PV's diameter is of utmost importance for the selection of an appropriate stent. The issue of anticoagulation in this setting is largely unsolved and warrants further studies. CMR imaging may be used to effectively diagnose PV stenosis and postinterventional angiographic success.