The Cost Effectiveness of LAA Exclusion
Bahij Kreidieh, Moisés Rodríguez-Mañero, Sergio H. Ibarra-Cortez, Paul Schurmann, Miguel Valderrábano
Methodist DeBakey Heart and Vascular Center and Methodist Hospital Research Institute, The Methodist Hospital, Houston, Texas.
Left atrial appendage (LAA) exclusion strategies are increasingly utilized for stroke prevention in lieu of oral anticoagulants. Reductions in bleeding risk and long-term compliance issues bundled with comparable stroke prevention benefits have made these interventions increasingly attractive. Unfortunately, healthcare funding remains limited. Comparative cost economic analyses are therefore critical in optimizing resource allocation. In this review we seek to discourse the cost economics analysis of LAA exclusion over available therapeutic alternatives (warfarin and the new oral anticoagulants (NOACs)).
Key Words : Left Atrial Appendage Exclusion, Cost Effectiveness, Watchman,Lariat.
Correspondence to: Miguel Valderrábano,
Associate Professor of Medicine, Weill College of Medicine, Cornell University
Adjunct Associate Professor of Medicine, Baylor College of Medicine
Director, Division of Cardiac Electrophysiology, Department of Cardiology Houston Methodist Hospital
6550 Fannin, Suite 1901 Smith Tower.
Atrial fibrillation (AF) is an abnormal heart rhythm characterized by rapid, disorganized activation (fibrillation) of the left and right atria of the heart. It is solely responsible for 15% of 700 000 strokes occurring in the United States each year.1 Multiple pharmacologic therapies are employed for stroke prevention in AF, including vitamin K antagonists (VKA) such as warfarin, newer agents such as dabigatran, rivaroxaban and apixaban and antiplatelet agents including aspirin and clopidogrel.
In recent years, nonpharmacologic therapies have been gaining acceptance as alternative stroke prevention strategies. They encompass exclusion of the left atrial appendage (LAA) from systemic circulation by surgical ligation or excision, percutaneous ligation and endovascular implantation of a left atrial occlusion device. Reductions in bleeding risk and long-term compliance issues bundled with comparable stroke prevention benefits have made these interventions increasingly attractive.2
While physicians are faced with a constantly expanding list of suitable treatment algorithms, healthcare funding remains limited. Comparative cost economic analyses of these interventions are therefore critical in optimizing resource allocation. They serve as an indispensable tool in the identification of neglected opportunities and redirection of resources to more efficient treatment strategies. It is predicted that the total number of life years saved by healthcare intervention could be doubled if proper reallocation of resources were to take effect.
In this review we seek to discourse the cost economics analysis of LAA exclusion over available therapeutic alternatives (warfarin and the new oral anticoagulants (NOACs)).
Worldwide Epidemiology of AF: A Cost Perspective
Awareness of the magnitude of the AF problem is warranted in understanding its cost economics analyses. AF constitutes a significant public health impediment, with an estimated share of 1% of the National Health Service budget in the United Kingdom3 and 16 to 26 billion dollars in annual United States expenses.3,4 Several regional studies suggest a rising prevalence and incidence of AF.2,5-8 In the United States, it is estimated that the number of adults with AF will more than double by the year 2050.9 Because the frequency of AF increases with advancing age, these secular trends may be explained in part by the demographic transition to an inverted age pyramid.10 Alternatively, an increase in AF incidence after age adjustment has been demonstrated, likely a reflection of fluctuating comorbidities and cardiovascular risk factors, in addition to miscellaneous contributors such as lifestyle changes.11
Chung et al10 conducted a comparative assessment of the burden of AF from 1980 to 2010 based on available epidemiological data from the 21 Global Burden of Disease (GBD) regions. The estimated global prevalence of AF in 2010 was 33.5 million. Burden associated with AF, measured as disability-adjusted life-years, increased by 18.8% (95% UI, 15.8–19.3) in men and 18.9% (95% UI, 15.8–23.5) in women from 1990 to 2010. Mortality associated with AF was higher in women and increased by 2-fold (95% UI, 2.0–2.2) in men and 1.9-fold (95% UI, 1.8–2.0) in women during the same time period. Overall, the data depicted strong evidence of progressive increases in overall burden, incidence, prevalence, and AF-associated mortality with significant public health implications.
Though the specific impact of stroke on mortality and disability was not scrutinized in this study, it remains a well-established contributor that influences outcomes of patients with AF.12 A substantial proportion of the mortality in AF patients is attributable to ischemic strokes, which account for 10% of early deaths and 7% of late deaths following AF diagnosis.11 The risk of ischemic stroke occurrence is increased 4-5 fold in patients with atrial fibrillation.11,12 Furthermore, AF is a known risk factor for stroke severity, recurrence and mortality.13 Roger et al approximated the annual cost of stroke care to be $40.9 billion.14
Cost Economic Evaluations: Cost-Effectiveness and Cost–Benefit Analysis
Cost-effectiveness analysis (CEA) is often employed in the evaluation of healthcare services. Typically cost effectiveness is quantified as the relationship between the cost associated with health gain given a certain measure (years of life, premature births averted, sight-years gained, etc.) divided by the health gain measure itself. Of course, healthcare benefit is not a black or white phenomenon. Aversion of death is no longer the only goal of healthcare providers. Alternate measures have been proposed to differentiate between a year of life in perfect health and a year of life with some degree of health impairment. One of the most commonly utilized outcome measures for this purpose is the quality-adjusted life year (QALY.15 This may be reported as discounted or undiscounted QALY, the former representing an adjustment that factors in the devaluation of a given outcome with time. The logic behind it is that any benefit is considered to be at its peak value to the patient in the immediate setting, with a predictable depreciation over time as adverse events result in declining quality of life. CEA uses a numerical indicator named “Incremental cost-effectiveness ratio” (ICER) which translates as the additional cost of extending a particular intervention divided by the additional health gain that would result1. Costs are usually described in monetary units while benefits/effects in health status are measured in terms of QALYs gained or lost. Though the numerical value may fluctuate from one nation to another, in the US, it is accepted that spending $50,000 per QALY is considered cost-effective expenditure. Conversely, a therapy that leads to an increase in cost with a decrease in QALY is deemed counterproductive.
Another commonly employed mean of investigation is Cost Benefit Analysis (CBA). It is a systematic process that serves to calculate and contrast the benefits and costs of an intervention. It involves comparing the total expected cost of each option against the total expected benefit, to see whether the benefits outweigh the costs, and by how much. In CBA, benefits and costs are expressed in monetary terms, and are adjusted for the time value of money, so that all flows of benefits and flows of project costs over time are expressed on a common basis in terms of their “net present value”.
Several decision-analytical models are utilized in economic evaluations, the Markov model most commonly chosen to address sophisticated health problems. This type of modelling permits presentation and analysis of probabilistic processes over time. It is usually used to simulate disease progression, and is particularly suitable for diseases that are chronic and recursive in nature, such as AF.
Cost-Effectiveness Analyses of NOAC for Stroke Prevention in AF
NOACs have pharmacological advantages over conventional anticoagulants that generally result in clinical benefit, as evidenced by various trials in a range of clinical settings.16-19,18-22 Unfortunately, these new drugs remain more expensive than VKA, thereby imposing a greater cost burden on health systems. Despite this, studies20-22 have consistently shown that the NOAC are cost-effective for stroke prevention in AF patients as compared to the more widely utilized conventional anticoagulants, particularly warfarin. For instance, Limone et al in a systematic review of economic models of NOACs vs Warfarin reported that ICERs vs Warfarin range between $3,547-$86000 for Dabigatran 150mg, $20,713 -$150,000 for Dabigatran 110mg, $23,065- $57,470 in Rivaroxaban and $11,400- $25,059 in Apixaban, concluding that all agents are ultimately cost effective. Of the available NOACs, apixaban has been shown to be the most cost-effective followed by dabigatran and rivaroxaban.23 All three agents exhibit a negative incremental cost and therefore produce savings.24
In brief, these new agents represent a paradigm shift in anticoagulant therapy for stroke prevention in AF. This will have to be taken into account when pharmacologic therapies are compared to non-pharmacologic alternatives.
Cost-Effectiveness Analyses of Left Atrial Catheter Ablation (LACA) for Stroke Prevention in AF
LACA is more and more commonly employed in the treatment of AF. Solid evidence supports LACA as an effective tool for AF symptom suppression, but only observational data support stroke prevention.25,26 A large multicentre randomized clinical trial (CABANA) is under way, specifically designed to address the impact of LACA in stroke and other outcomes. Therefore, though not specifically aiming for stroke prevention, this treatment modality could theoretically decrease stroke incidence by virtue of rhythm control. A decision-analytic model was designed to assess the cost effectiveness of LACA in low and moderate stroke risk patients.27 Costs and outcome measures were derived from the literature and Medicare data. The study concluded that LACA could be cost-effective in AF patients at moderate risk for stroke, but remains ineffective in low-risk patients.
A CBA is warranted in further analysing the advantages of LACA as compared to LAA exclusion and pharmacotherapeutic stroke prevention modalities. Such a model would take into account the broader benefit spectrum of LACA as pertains to heart failure progression, symptomatic relief and more.
Evaluation of Net Clinical Benefit of Left Atrial Appendage Closure for Stroke Prevention in AF
Current international guidelines propose the consideration of LAA closure in patients at high risk of stroke, who have contraindications to anticoagulation, are at high bleeding risk or prefer an alternative means of prevention.28-32 In a clinical setting, 61.8% of patients implanted with a Watchman device were considered ineligible for anticoagulation therapy based on their comorbidities, bleeding history/risk, and compliance issues.28 Of these, 38.7% had prior major bleeding or predisposition to bleeding (HASBLED score >2). Unfortunately, available cost-effectiveness data derives from large trials, such as PROTECT AF, which sought to compare LAAC outcomes to Warfarin therapy, and included only patients eligible to Warfarin.33 As such, conclusions from these analyses cannot be applied directly and reliably to LAAC in clinical practice.
Gangireddy et al34 conducted a post-hoc analysis utilizing patients who underwent Watchman device closure as compared to those maintained on anticoagulation. A total of 707 patients in the PROTECT AF trial and 566 patients from the Continued Access PROTECT AF (CAP) registry were included. Net clinical benefit (NCB) of LAA closure was estimated based on incidence of ischemic stroke, intracranial haemorrhage, major bleed, pericardial effusion and death. Each adverse outcome was weighted according to its relative impact in disability and death. NCB was 1.74%/year in the PROTECT AF trial and 4.97%/year in the CAP registry, both in favour of LAAC over anticoagulation. Furthermore, greater benefit was documented in patients with CHADS2 score of 2 as compared to those with a CHADS2 score of 1. A temporal analysis showed a linear benefit curve for anticoagulation with time, as opposed to a bimodal curve for LAAC. Early procedure related strokes and pericardial effusions adversely influenced the early benefit subsequent to LAAC. However, a later decline of these complications bundled with decreasing incidence of intracranial haemorrhage and death lead to an improvement in NCB as compared to Warfarin in the long run.
Economic Evaluation of Percutaneous Left Atrial Appendage Occlusion, NOAC and Warfarin for Stroke Prevention in Patients with Nonvalvular AF
To date, there are no trials that directly compare NOAC with LAA exclusion devices from an economic perspective. Therefore, we will have to derive this endpoint from broader warfarin-controlled trials as it remains the established standard.
Singh et al35 evaluated the quality-adjusted survival and costs associated with warfarin, dabigatran, or LAA occlusion strategies in patients with non-valvular AF at risk of stroke. A Markov model was developed that simulated 10,000 individual patient iterations in order to assess the projected costs and outcomes, estimating probabilities of different potential outcomes based on published data. Three primary treatment modalities were incorporated: (1) Dose-adjusted warfarin with a target international normalized ratio (INR) of 2.0 to 3.0, (2) Dabigatran, and (3) LAA occlusion. Outcomes of interest were life expectancy (measured in years), QALYs, costs and the ICER. For each therapeutic approach, 5 health states were possible: (1) No significant events, (2) myocardial infarction (MI), (3) stroke, (4) bleed, or (5) death.
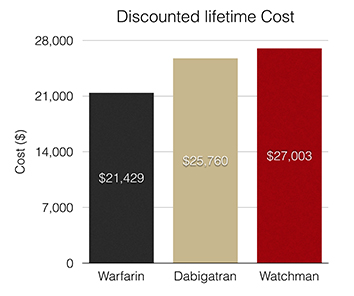
At 4.55 years, warfarin therapy exhibited the lowest discounted quality-adjusted life years, followed by dabigatran at 4.64 and LAA occlusion at 4.68 (Figure 1). The average discounted lifetime cost was $21,429 for patients on warfarin therapy, $25,760 in the dabigatran arm, and $27,003 for LAA occlusion patients (Figure 2). Compared with warfarin, the ICER for LAA occlusion was $41,565 while that of dabigatran was $46,560. This meant that dabigatran imposed a higher financial weight per added unit of effectiveness as compared to LAA occlusion.
Figure 1. Relative impact of available stroke prevention interventions on life expectancy and quality adjusted life years in patients with NVAF as derived from Singh et al, 2013
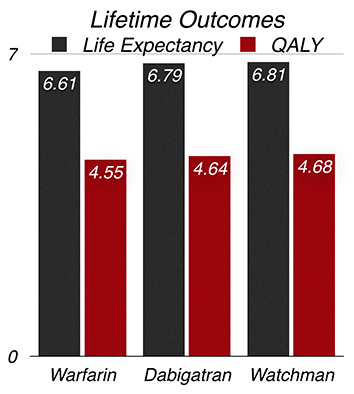
Figure 2. Relative discounted lifetime costs associated with various stroke prevention interventions in patients with NVAF as derived from Singh et al, 2013
Subsequently, the study concluded that LAA occlusion and dabigatran are both cost-effective as compared to warfarin therapy. More importantly, it affirmed that based on current evidence, a strategy of LAA occlusion is more cost effective than dabigatran therapy.
The same group more recently published a similarly designed Markov model (Micieli et al), this time to assess interventions in new onset NVAF.36 In addition to the previously studied interventions, it incorporated Rivaroxaban and Apixaban. The present study adopted a base case consisting of patients with new onset NVAF presenting to the ED. In contrast, the group’s previous base case35 (Singh et al, 2013) consisted of NVAF patients presenting to outpatient care with stroke risk factors similar to those in the RE-LY and PROTECT AF trials. The new study again looked at QALY’s and discounted lifetime cost. Warfarin again had the lowest QALY (5.13), followed by Dabigatran (5.18), Rivaroxaban (5.21), LAAO (5.21) and Apixaban (5.25). Similarly, Warfarin again had the lowest discounted lifetime cost ($15,776) followed by Rivaroxaban ($18,280), Dabigatran ($20,794), LAAO ($21,789) and Apixaban ($28,167). Overall, the study related that Apixaban is the most cost effective intervention for new onset NVAF.
The study by Singh et al is the first comparison of these novel therapies for stroke prevention in NVAF. Although a direct comparison of LAA exclusion and dabigatran would be ideal, such a study would require a large patient population with long-term follow-up to demonstrate noninferiority35 and is therefore unlikely to be available to us in the foreseeable future.
Singh et al employed a population aged at 76 years, a factor that sheds controversy on the applicability of these results to younger patient populations. Moreover, it also remains to be seen whether these calculation may be applicable to other jurisdictions with different models of healthcare delivery and funding.
The LAA exclusion leg in this study is modelled closely after, and derives data from the PROTECT AF trial.37 Follow-up data from the trial published recently also showed non-inferiority for the composite endpoint but affirmed more primary safety events in the LAA occlusion group than in the warfarin group.38 Furthermore, the recently published PREVAIL trial reported significantly improved procedural safety parameters compared to PROTECT AF. Pericardial effusions requiring surgical repair decreased from 1.6% to 0.4% (p=0.027), and those requiring pericardiocentesis decreased from 2.9% to 1.5% (p=0.36). Therefore, it seems plausible to infer that ICERs derived from this trial could demonstrate an even larger cost-effectiveness in the LAA occlusion arm.
Most relevantly, Singh et al assumed the price of the Watchman to be ~$8500 in both studies described above. Current prices of the Watchman device in the US are $12500 - $18000. These differences would significantly alter all cost-effectiveness analyses.
A CBA of LAA Closure versus Warfarin for Stroke Prevention in AF
Reddy et al39 constructed a cost benefit model, established using clinical data from the PROTECT AF trial. Adverse outcomes for LAA closure were estimated from PROTECT-AF data and warfarin outcomes were derived from the literature. Costs encompassed the cost of treatment and procedural complications. Benefits were defined as the savings accrued through reduction in stroke and mortality. The model showed an initial disadvantage to LAA closure, with the first 5-year cumulative cost benefit of -$2,300 when each additional life year is valued at $10,000. At year 6 however, the tables were turned as a positive cost benefit of $750 was evident, with further benefits accumulating each life year thereafter. The following year, the group constructed a Markov model comparing clinical outcomes, quality of life and total costs of LAAC vs Warfarin, once again derived from PROTECT AF data.40 They reaffirmed that LAAC is cost effective at 6 years and dominant at 10 years, at which time it becomes less expensive and more effective than Warfarin (Table 1)
.
Table 1. Results of a cost utility analysis of LAAC vs Warfarin conducted by Reddy et al40
| Time (Years) |
6 |
10 |
Lifetime |
| Incremental quality-adjusted life expectancy (years) |
0.16 |
0.4 |
1.3 |
| Incremental Cost per QALY ($) |
37,713 |
Dominant |
Dominant |
Similarly, a budget impact model was constructed to project the 10 year cost-effectiveness of LAA closure (modelled using the PROTECT AFIB trial) as compared to warfarin and Dabigatran (modelled using the RE-LY trial).41 The cost/benefit of LAA closure decreased with additional life years, becoming less expensive than Dabigatran at 8 years, and only 10% more expensive than Warfarin at 10 years (Figure 3).
Figure 3. Total cumulative cost of LAA occlusion vs Warfarin at year 1 and year 10. Cost of LAA occlusion is significantly higher initially, but costs level off at 10 years post-intervention
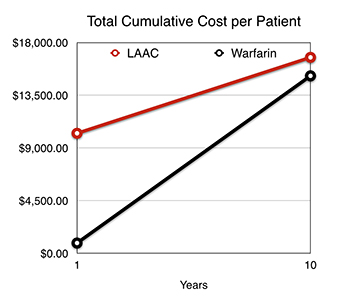
Both studies showed that the majority of costs associated with LAA closure are borne early, mainly in the first year. However, in the long term, this modality becomes increasingly cost-effective and provides an opportunity for chronic healthcare savings. This information may provide the framework for physicians in assigning treatment strategies based on predicted life expectancy. From an economic standpoint, LAA closure may be ill advised in patients with a very low predicted life expectancy (<2-3 years). This is because a high expense will be met immediately, and patients are unlikely to benefit adequately from it within their lifetime.
Applicability of These Results to Other LAA Exclusion Devices.
At present, there is no CEA or CBA data pertaining to non-Watchman endocardial and epicardial (Lariat) LAA exclusion devices. With less established clinical efficacy parameters and different safety outcomes, it seems unlikely that the available results could be extrapolated to them.
The Lariat is associated with a lower rate of leaks at 1 year compared to Watchman.42 On the other hand, manipulation of the LAA with the Lariat device is both endocardial and intrapericardial. This dual access approach widens the range of possible complications (ventricular puncture, epigastric vessel laceration, hemopericardium, pericarditis, and incomplete ligation). A retrospective, multicenter study of consecutive patients undergoing LAA ligation with the Lariat device43 reported major complication occurrence in 15 patients (9.7%), and procedural success limited by bleeding. Such factors are likely to negatively impact the CEA and CBA.
Initial attempts to reduce stroke risks in patients with AF were made by cardiac surgeons performing excision, suture closure, or stapling of the LAA. These procedures have been performed for many decades in thousands of patients undergoing cardiac surgery for other conditions and, to a lesser extent, as standalone surgical procedures. Surprisingly, there is no data available indicating benefits in patients undergoing these procedures.38 Indeed, the only randomized study on surgical LAA exclusion, published years ago, failed to show a reduction in stroke events in the treatment group.1,44
It is clear that cost-effectiveness data extrapolated from Watchman trials cannot be generalized to all LAA exclusion strategies. Additional clinical efficacy data gathered over a significant follow-up period will be critical in establishing reliable cost effectiveness analyses for these intervention modalities.
Limitations in Available Data
Available LAA exclusion cost-effectiveness data have been extrapolated from the PROTECT-AF population. Given that CEA compares different treatments and their differences, it is unlikely that using data from PREVAIL would yield a more favourable result for the Watchman device, considering the low rates of stroke in the control population of this trial.
As mentioned, one of the most prominent limitations to present cost-effectiveness analyses is the absence of a direct comparison between LAA exclusion and NOACs. These novel agents are gaining widespread acceptance, and are likely to become more affordable with time, factors that will surely sway CEAs in their favour. Similarly, many patients have absolute contraindication to oral anticoagulants, and so the alternative for these patients would be the receipt of no therapy at all. An ICER comparing LAA closure to non-therapeutic controls does not exist to date, but could be particularly useful for decision-making within this subset of patients.
Little attention has been given to LACA as a stroke prevention modality. Its utilization in the moderate to high stroke-risk population was shown to be cost-effective, and may present a reasonable alternative to LAA exclusion whose long-term clinical repercussions remain unclear. Furthermore, this modality may be used in patients with contraindication to anticoagulation, making a CBA of the two modalities more precise.
To date, cost-effectiveness data on LAA exclusion remain scarce. The limited studies available are heavily routed in Watchman clinical outcomes studies, and cannot be generalized to alternative LAA exclusion modalities. Available analyses however, have shown a cost-effective advantage to LAA exclusion, more prominently so in the long run.