Ablation of Atrial Fibrillation in Combination with Left Atrial Appendage Occlusion in A Single Procedure. Rationale and Technique
Ignacio García-Bolao, Naiara Calvo, Alfonso Macias, Joaquin Barba, Nahikari Salterain, Pablo Ramos, Gabriel Ballesteros, Renzo Neglia
Clinica Universidad de Navarra. Pamplona, Spain.
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia, and is associated with a fivefold increase in the risk of ischemic stroke and systemic embolism.
Left atrial appendage (LAA) is the source of thrombi in up to 90% of patients with nonvalvular atrial fibrillation (AF). Although thromboembolic prophylaxis by means of oral anticoagulants (OAC) has been shown to be very effective (OAC), they also confer an inevitably risk of serious bleeding.
Catheter ablation (CA) is an effective treatment for symptomatic AF but its role in stroke prevention remains unproved. Recently, LAA percutaneous occlusion has been demonstrated to be equivalent to OACs in reducing thromboembolic events.
The aim of this review is to describe the rationale, feasibility, outcomes and technique of a combined procedure of AFCA and percutaneous LAAO, two percutaneous interventions that share some procedural issues and technical requirements, in patients with symptomatic drug-refractory AF, high risk of stroke, and contraindications to OACs.
Key Words : Atrial Fibrillation Ablation, Left Atrial Appendage Closure, Thromboembolism.
Correspondence to: Ignacio García-Bolao.
Department of Cardiology and Cardiac Surgery.
Clínica Universidad de Navarra.
Avda Pio XII 36, 31008 Pamplona. Spain.
Non-valvular atrial fibrillation (AF) is associated with a fivefold increase in the risk of ischemic stroke and systemic embolism that causes increased mortality and morbidity and higher medical expenses.1 Hence, assessment of thromboembolic risk and the use of adequate prophylactic anticoagulation is mandatory in the proper clinical management of AF.
Although anti-vitamin K drugs or the more recently introduced factor II/Xa inhibitors can significantly reduce the risk of stroke in at-risk patients with AF, these oral anticoagulants (OAC) medications are associated with severe hemorrhagic adverse effects2 On the other hand, atrial fibrillation catheter ablation (AFCA) is an effective rhythm control strategy for patients with symptomatic, drug-refractory AF but its role in stroke prevention remains unproved.3
These challenges have led to interest in mechanical exclusion of the left atrial appendage (LAA), that has been shown to be the source of thrombi in up to 90% of the patients with non-valvular AF, as an interventional, ¨local¨, method to prevent thromboembolism in AF. Devices for percutaneous occlusion have shown efficacy and safety in achieving this goal when OAC is contraindicated or declined by the patient.4
Combining AFCA and LAAO, two percutaneous interventions that share some procedural issues and technical requirements, could reduce the incidence of stroke in selected high-risk patients while simultaneously relieving AF symptoms in a single session. The aim of this review is to describe the rationale, feasibility, outcomes and technique of a combined procedure of AFCA and percutaneous LAAO.
Limitations of Anticoagulation and Rhythm Control Strategies to Prevent Thromboembolism
Although thromboembolic prophylaxis by means of OAC was shown to be very effective, leading to a 60% relative risk reduction of stroke compared to placebo,2 vitamin K antagonists, principally warfarin, have some significant deficiencies such as slow onset of action, narrow therapeutic windows, need for regular blood sampling to monitor the international normalized ratio, marked inter-individual variations in drug metabolism, overlap with parenteral anticoagulants and multiple drug and food interactions, all of which lead to an incomplete protection or an increased risk of bleeding.5 For these reasons, as many as 65% of patients with indications for such treatment do not receive it, while the international normalized ratio (INR) is estimated to be out of range in a further 19% of patients.6
Recent clinical trials have found that newer, target-specific oral anticoagulants (NOACs) such as direct thrombin inhibitors (dabigatran) and Xa inhibitors (rivaroxaban, apixaban and edoxaban) which do not require monitoring and have fewer drug interactions, offer efficacy and safety equivalent to, or better than, those of warfarin for reducing the risk of stroke in patients with non-valvular AF.7-10 But like warfarin, and due to the inherent nature of anticoagulation, they also confer an inevitably risk of serious bleeding. Moreover, although the inclusion of NOACs in the secondary prevention of systemic embolism has increased the safety of chronic anticoagulant therapy in patients with NVAF, roughly 20% of patients resign from these new agents within 2 years of therapy due to complications or poor tolerance.11 For example, in the RELY trial,7 10% of patients receiving dabigatran and 17% of those receiving warfarin stopped the treatment within 1 to 2 years. In the ROCKET-AF study,8 24% of those treated with rivaroxaban and 22% of those treated with warfarin stopped treatment during the study, in the ARISTOTLE trial,9 25% of patients discontinued apixaban and 28% discontinued warfarin and during the ENGAGE study,10 34% of patients stopped warfarin and 19% interrupted edoxaban.
Apart from OAC, different strategies of maintenance of sinus rhythm with antiarrhythmic drugs or catheter ablation have been studied as another means to reduce the incidence of stroke in atrial fibrillation patients. Although it seems logical that the risk of systemic embolism may be lessened if the atrial fibrillation burden can be reduced or eliminated by rhythm control (antiarrhythmic drugs or AFCA), many clinical trials have failed to demonstrate a difference between rhythm control with antiarrhythmic drugs in reducing rates of stroke or systemic embolism12 probably due to the low effectiveness for maintaining sinus rhythm or the high rate of discontinuation of antiarrhythmic drugs. Regarding AFCA, and although low stroke risks were reported by observational studies in patients who maintained sinus rhythm after ablation despite OAC cessation, most current guidelines.3,13 recommend that systemic anticoagulation should be continued indefinitely in patients with a high risk for stroke, due to several facts. First of all, recurrences (not only symptomatic but also silent) of AF are common both early and late following AF ablation. Secondly the ablation procedure itself destroys a portion of the atria and, finally, the impact of this on stroke risk is uncertain and it has not been addressed by large randomized prospective trials designed to assess the safety of stopping anticoagulation after AF catheter ablation. For all these reasons, nowadays, AFCA is considered a symptomatic treatment and the consensus is that it should not be indicated with the sole purpose of stopping anticoagulation.3,13,14
Left Atrial Appendage Closure for Stroke Prevention
The LAA has long been recognized as the site of clot formation in most patients with nonvalvular AF and in fact, it has been shown that 90% of thrombus in patients with AF form in this cul-de-sac structure.15 Thus, LAAO to preventing thromboembolism has important theoretical basis in patients with AF.
Due to the aforementioned concerns with anticoagulation, interventional alternatives for the prevention of thromboembolism in patients with NVAF, such as the exclusion of the left atrial appendage (surgically or percutaneously using different dedicated devices) have been explored (Figure 1). The main body of scientific evidence comes from the PROTECT-AF cohort, the most relevant randomized clinical trial that has compared both strategies. In the PROTECT-AF trial, 707 patients from fifty-nine centers in the USA and Europe were prospectively randomized in a 2:1 ratio in an unblinded fashion to LAAO with the Watchman device versus standard warfarin therapy.16 The trial was designed to examine the efficacy and safety of percutaneous closure of the LAA in patients with nonvalvular AF (not contraindicated for warfarin) and to assess noninferiority of the WATCHMAN LAA occluder device to standard warfarin therapy, which was the control arm. The first publication,16 showed that the efficacy of percutaneous closure of the LAA with WATCHMAN device was non-inferior to that of warfarin therapy. Importantly, when follow-up was extended from 600 to 2621 patient-years (3.8 years),4 LAAO reduced the relative risk of the primary end point (the composite of stroke, systemic embolism, and cardiovascular death) by 40% (1.5% absolute reduction) compared with the warfarin control arm. Furthermore, the device-based strategy was associated with a 60% relative risk (1.4% absolute reduction) of cardiovascular death and 34% relative reduction (5.7% absolute reduction) in all-cause death, strongly suggesting for the first time a survival benefit for the Watchman group when compared with the control warfarin. Longer follow-up results, up to 5 years, were recently communicated (Reddy et al, data not published) showing similar results. It should be noted that the mean follow-up in this trial exceeded by far that of most contemporary stroke prophylaxis trials,7-10 such as RELY (2.0 years), ROCKET-AF (1.9 years), ARISTOTLE (1.8 years) and ENGAGE (2.8 years)
Figure 1. Transoesophageal echocardiography images during the combined ablation procedure and LAAO Left panel: three-dimensional TEE views after an ACP device were released Right panel: two-dimensional TEE images during the deployment of a Watchman device. Optimal position and compression was confirmed with no residual leaks and a tug test was performed demonstrating simultaneous movement of the device and LAA before releasing the device
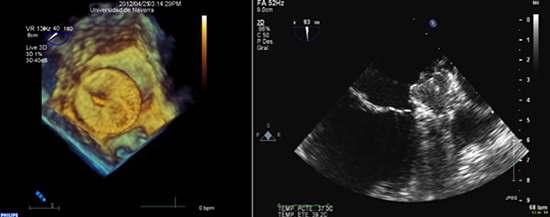
Although in the PROTECT-AF there was an initial higher rate of adverse safety events in the intervention group than in the control group,17 the positive effect of increased operator experience and overcome of the learning curve was clearly demonstrated in the CAP registry with shorter implant time, higher implant success and warfarin discontinuation rate, and lower complication rates.15 The PROTECT-AF trial also found that, regardless of whether the enrolled patients had received prior warfarin therapy, the LAA closure significantly improved the quality of life of patients.18
The ASAP registry focused on the effects of LAAO in those patients who cannot tolerate warfarin even in the short term. In 150 patients followed for a mean of 14.4 (± 8.6), the authors found a a 77% reduction from the expected stroke rate of 7.3% based on the CHADS2 scores of the patient cohort.19
The Watchman device received the FDA approval in 2015,20 “to reduce the risk of thromboembolism from the left atrial appendage (LAA) in patients with non-valvular atrial fibrillation who:
• Are at increased risk for stroke and systemic embolism based on CHADS2 or CHA2DS2- VASc scores and are recommended for anticoagulation therapy;
• Are deemed by their physicians to be suitable for warfarin; and
• Have an appropriate rationale to seek a non-pharmacologic alternative to warfarin, taking into account the safety and effectiveness of the device compared to warfarin“.
There are no available data coming from randomized trials related to other occlusion devices or LAA ligation techniques. Initial results with the ACP/Amulet occluder, described from some observational studies and multicentric registries are promising, indirectly comparable to some extent to the Watchman device, but to date there is no available head-to-head comparison of the Amplatzer devices with oral anticoagulation.21
Typical indications for percutaneous LAAO include patients with a high thromboembolic risk (CHA2DS2-VASc score of .2) but with a contraindication to systemic anticoagulation. This subset of patients represent the most accepted clinical indication for LAA occlusion, albeit by having to extrapolate the results of the PROTECT AF study to that specific cohort, that was specifically excluded from this trial. In addition, the results of the ASAP registry would support this indication.19 According to the 2012 ESC Guidelines for the management of atrial fibrillation this indication has a class IIb recommendation.3
Other recommended indications for the use of LAA occluders, based in an expert consensus statement published recently22 are summarized in (Table 1).
Table 1. Suggested indications for left atrial appendage occlusion22
| 1 |
As alternative to oral anticoagulation when oral anticoagulation is possible |
| • |
Patient refusal of (N)OAC despite adequate information and physician advice |
| 2 |
As replacement for anticoagulation when anticoagulation is not possible |
| 2a |
Patients with a contraindication to anticoagulation |
| • |
Patients with a high thromboembolic risk (CHA2DS2-VASc score of ≥ 2) but contraindication to (N)OAC |
| 2b |
Patients with an increased bleeding risk under systemic anticoagulation |
| • |
HAS-BLED score ≥ 3 |
| • |
Need for a prolonged triple anticoagulation therapy (e.g. recent coronary stents) |
| • |
Increased bleeding risk not reflected by the HAS-BLED score (e.g. thrombopenia, cancer, or risk of tumour-associated bleeding in case of systemic OAC) |
| • |
Severe renal failure as contraindication to NOAC |
| 3 |
As a complement to anticoagulation |
| • |
Patients with embolic events despite adequate OAC provided no other plausible cause |
| 4 |
As adjunct to ablation of atrial fibrillation |
| • |
Patients with a significant risk of thromboembolic events (CHA2DS2-VASc score of ≥ 2) undergoing an ablation procedure to treat symptomatic atrial fibrillation, who, in addition, have a strict or relative contraindication to (N)OACs |
(N)OAC: (Novel) oral anticoagulant
Rationale and Possible Indications for the Combination of Laao and AF Ablation
In view of the above, the combination of LAA occlusion with catheter ablation might be a comprehensive way to improve the symptoms of AF while at the same time reducing the incidence of stroke in selected high-risk patients in a single session. Theoretically speaking, and assuming that the abscence of randomized data and/or cost-effectiveness studies supporting this strategy do not allow a broad recommendation, this hybrid procedure would cover the full clinical spectrum of AF in terms of antiarrhythmic and symptomatic effects (CA) as well as an anti-embolic intervention (LAAO) for selected patients.
Provided that the patient has a formal indication for both procedures, and specially if they have high stroke or bleeding risk and an anticipated reduced efficacy of CA alone, the combination of PV isolation with LAA closure in a single session could reduce the need and risks of a repeated left atrial intervention, a new transseptal puncture, perhaps general anaesthesia, and probably a new anticoagulation perioperative period should LAAO become desirable during follow-up.
Based on the expert consensus, in single-center observational series and in personal communications, the EHRA/EAPCI document on catheter-based LAA occlusion22 suggests that patients with a significant risk of thromboembolic events (CHA2DS2-VASc score ≥2) undergoing an AFCA procedure, who also have a strict or relative contraindication to OACs, might be acceptable candidates for the combination of LAA occlusion and AF ablation in a single procedure.
Results of Combining Laao and AF Ablation in a Single Session
So far, few data on the combination of LAA occlusion and AF ablation in a single session have been published (Table 2).
Table 2.
|
Type |
n |
AF type |
CHADS2-VASC |
HASBLED |
Technique |
Procedure time (min) |
LAAO success 3-m (%) |
1-yr OAC freedom(%) |
1-yr AT freedom (%) |
Complications |
| Swaans et al23 |
Observational |
30 |
Px/Ps |
3 (1-5) |
2 (1-3) |
PVAC ± MASC Watchman |
97 (75-115) |
97 |
77 |
70 |
1 late dislodgement 1 tongue hematoma 2 groin hematoma |
| Calvo et al24 |
Observational |
35 |
Px/Ps/LSPs |
3.1±1.1 |
3.1±1 |
RF PVI ± roof line Watchman/ACP |
160±33 |
100 |
97 |
78 |
3 pericardial effusions |
| Romanov et al25 |
Randomized |
89 |
Px/Ps |
2.2±0.6 |
3.5±0.8 |
RF PVI Watchman |
189 ± 29 |
87 |
79 |
59 |
2 groin hematoma |
The first report of this strategy comes from a series of 30 consecutive patients with documented paroxysmal, or (longstanding) persistent, non-valvular AF with a CHADS2 score ≥1 or (relative) contraindication for OAC.23 The authors performed pulmonary vein isolation (PVI) with the phased multipolar ablation system (PVAC)® in all the patients and additional complex-fractionated atrial electrograms ablation with the MASC and MAAC in 8 cases immediately followed by LAAO with the Watchman device. The median CHADS2 and HAS-BLED scores were 2.5 and 2, respectively. Twenty-three patients (77%) had a history of stroke, of whom 9 (30%) had a stroke under oral anticoagulation. Eight patients (27%) had a relative contraindication for VKA that was due to bleeding or failure to achieve an adequate international normalized ratio, and 2 patients had both. The median total procedure time was 97.3 minutes (38 minutes for LAAO). A median of 1.5 devices per patient were required to reach an optimal LAAO and at the end of the procedure, 3 patients had minimal residual flow (flow ≤5 mm). There were only 3 minor perioperative complications. At the 12-month follow up, 70% of the patients were free from atrial arrhythmias, 13% of the patients underwent a redo procedure, 23% of the patients did not discontinued Warfarin (1 due to late device embolization, 1 for dense spontaneous contrast in the left atrium, 1 due to pulmonary embolism, and in 4 patients, due to their treating cardiologist’s preference on the basis of recurrent or persistent AF). During follow-up none of the patients had thrombus formation on the surface of the device and no thromboembolic events had occurred. Three patients had a severe non-procedural bleeding event.
We reported our experience on 35 consecutive patients with symptomatic drug-refractory AF, a CHADS2 score of ≥1, a CHA2DS2-VASc score ≥2 and relative or absolute contraindications for OACs, or who refused OAC therapy despite adequate information.24 Patients underwent a combined procedure of PVI ± roof line and LAAO with the Watchman or the ACP device, depending on the LAA anatomy. Median score was 3 on both CHA2DS2-VASc and HAS-BLED. Persistent or long-standing persistent AF was present in 71% of the patients. Nine percent of the patients had a prior stroke under OAC, and 48% had bleeding complications. The mean total combined procedure time was 160.5 ± 33.75 min, while the mean subsequent LAAO procedure time was 42.05 ± 11 min. A Watchman device was implanted in 29 (82%) patients and an ACP in 6 (18%) patients. A median of 1.3 devices per patient was used in this series. The periprocedural complications included three cases of severe pericardial effusion successfully treated by percutaneous pericardiocentesis (1 presumably due to the PVI procedure and 2 to the LAAO procedure). There were no device embolization events during follow-up. One patient died 17 days after the procedure, while on OAC and aspirin, due to an intracerebral hematoma associated to extremely high INR. At 3-month follow-up, all 35 patients (100%) met the criteria for successful sealing of the LAA. At a mean follow-up of 13 months (3–75), 78% of patients were free of arrhythmia recurrences and 97% discontinued OAC. There was one case of transient ischemic attack at 2 years post-procedure. The transesophageal echocardiograpy (TEE) did not reveal LA thrombus and there was complete closure of the LAA. This patient was placed on clopidogrel for secondary prevention. In our series, the observed ischaemic stroke rate was 2.6% per year representing 42.3% fewer events than expected according to the predicted ischemic stroke rate of this cohort taking from historical series.24
Finally, Romanov et al25 recently published a trial on 89 patients with paroxysmal or persistent AF and high thromboembolic and bleeding risk, that were randomized to either PVI or PVI + LAAO with the Watchman occluder. The aim of the study was to assess the impact of LAAO added to PVI in terms of the antiarrhythmic response of PVI. Ninety-eight percent of the patients received and implantable loop recorder. Briefly, The closure device was successfully implanted after PVI in 39 (87 %) of the 45 patients assigned to the intervention group, there were no statistical differences between both groups in terms of procedure-related complications, and LAAO was not observed to influence the success of PVI (evaluated by time to the first recurrence of any atrial tachyarrhythmia and the AF burden) after the blanking period.
Taken together, this results suggest that the combination of LAAO with different PVI techniques in a single session can be performed successfully and safely, do not interfere with repeat PV isolation, and do not seem to influence the long-term success of PVI in patients with symptomatic refractory AF.
Technical Challenges of The Combined Procedure
The completion of combined CA and LAAO in a single session involves some technical modifications compared to the standalone procedures that have to be taken into account. Regarding the pre-procedural assessment, and although it is not mandatory, we perform routinely a multi-slice cardiac CT scan in all the patients. A 3D reconstruction of the left atrium is useful to assess the morphologic features of the LAA (type, measurements, presence of challenging anatomies, etc). Although the final decision on device size is based on information collected with both intraoperative TEE and/or fluoroscopy, CT scan allows to choose in advance the type of the occluder, hints to the device size and is helpful to rule out the presence of thrombi, therefore avoiding the need of the preoperative TEE. Another additional advantage of the CT scan for the combined procedure is its subsequent use for image integration with the non-fluoroscopy navigation system during the AFCA procedure.
Regarding the type of the device, in our institution the ACP/Amulet devices are usually indicated for appendages shorter than wide or with very complex anatomies, while the Watchman device is implanted in the rest of the patients. Although there is no definitive evidence, the physical structure of the Watchman occluder, without a disk covering the pulmonary ridge, and the lower incidence of late embolization whith this device, could facilitate a hypothetical redo procedure.
The LAAO procedure tipically requires continuous intraoperative TEE guidance and therefore, general anesthesia. Although we perform our standard AFCA under simple conscious sedoanalgesia, and we do not use TEE guidance for the transseptal punctures, during the combined procedures we prefer to perform both interventions under general anesthesia from the beginning. First of all, TEE is extremely useful to titrate the location of the transeptal puncture, since an inferoposterior access and the avoidance of a PFO entrance is essential. Secondly, from the logistic point of view and due to the need TEE guidance during transseptal puncture and LAAO we feel more comfortable with the patient under general anesthesia throughout the entire procedure.
Post-procedural anticoagulation with warfarin is recommended for the Watchman device to avoid thrombus formation on the device until completion of endothelization, provided there are no contraindications to anticoagulation. However, in the ASAP registry, patients received clopidogrel for 6 months and ASA indefinitely without OAC, and the ischaemic stroke rate was only 1.7% compared with 2.2% in the PROTECT AF device group. The postprocedural anticoagulation strategy for the ACP device banks on the good record regarding low thrombogenicity of the Amplatzer device family, and indicates in its instructions for use DAT only without an oral anticoagulant. Therefore, a standalone LAAO can be managed without postoperative anticoagulation. However, after a combined procedure, patients should receive systemic anticoagulation for two months, according to the 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation.26 For these reason, whenever possible, we use warfarin or a dose-adjusted regimen of low-molecular weight heparin during this period and we perform the follow-up TEE after two months, shifting to (dual) antiplatelet therapy upon its result. Note that due to the need of a short period (1-3 months) of anticoagulation after a left linear atrial ablation, caution should be taken before indicating a combined procedure in patients with an absolutely strict contraindication for short-term oral anticoagulation.
Finally, and needless to mention, the multidisciplinary nature of this combined intervention requires the availability of an appropriate team with specific training and experience in both AFCA and LAAO procedures, including an anesthesiologist, an experienced echocardiographer, and nursing and technical staff who are familiar with every procedural step.22
The combination of AFCA and percutaneous LAAO in a single procedure is a feasible strategy in patients with symptomatic drug-refractory AF, high risk of stroke, and strict or relative contraindication to OACs. This strategy will undoubtedly undergo further scrutiny in future randomized trials and cost-effectiveness studies but is highly attractive for its potential as a combined antiarrhythmic and antithrombotic intervention in high-risk patients.