Atrioesophageal Fistula: A Review
Krishna Kumar Mohanan Nair, MD1, Asaf Danon, MD, MSc2, Ajitkumar Valaparambil, MD1, Jacob S. Koruth,MD3, Sheldon M. Singh, MD4
1Department of Cardiology, Sree Chitra Tirunal Institute of Medical Sciences &
Technology, Thiruvananthapuram, Kerala, India
.2Department of Cardiology, Lady Davis Carmel Medical Center, Haifa, Israel.
.3Helmsley Cardiac Arrhythmia Service – Mount Sinai Medical Center, Mount Sinai School of Medicine, New York, NY, USA
.4Schulich Heart Program, Sunnybrook Health Sciences Centre, University of Toronto, Toronto, Ontario Canada
.
Catheter ablation of atrial ablation (AF) has become an important therapy in recent years. As with all evolving techniques, unexpected complication may occur. Atrioesophageal fistula is a very rare complication of AF catheter ablation. Described for the first time in two very experienced centers in 2004, this complication is the most dreadful and lethal among all the others related to AF catheter ablation. Its clinical presentation is extremely variable. Rapid diagnosis and surgical therapy may prevent death. This review article will summarize the risk factors, diagnosis, treatment and possible preventive strategies for this condition.
Key Words : Catheter Ablation, Complications, Atrial Fibrillation.
Correspondence to: Krishna Kumar Mohanan Nair, MD
Department of Cardiology
Sree Chitra Tirunal Institute of Medical Science and Technology
Thiruvananthapuram, Kerala, India
Non-pharmacologic therapy for atrial fibrillation (AF) is increasing in popularity and can be performed with catheter based and surgical approaches. The most commonly employed strategy for ablation of AF presently involves creation of circumferential lesions around the pulmonary vein ostia or antra with or without the placement of additional ablation lesions within the left atrium (i.e. linear lesions in the left atrial roof, mitral isthmus, or ablation of sites with complex fractionated atrial electrograms).1,2,3
AF ablation carries a small risk of complications with the most serious being atrioesophageal fistula (AEF). Although the incidence is less than 0.1%, it is usually fatal.4,5,6 Esophageal perforation or fistula was reported in 31 patients (0.016%) in the Global Survey of Esophageal and Gastric Injury in Atrial Fibrillation study. Symptom onset for esophageal perforation or fistula was reported on average 19.3 days after the ablation procedure but could appear as short as 6 days and as long as 59 days post ablation.6 Esophageal injury has been observed most frequently with percutaneous radiofrequency ablation, although it has also been reported with other energy sources including cryoablation,7 high-intensity focused ultrasound,8 and even surgical ablation.9
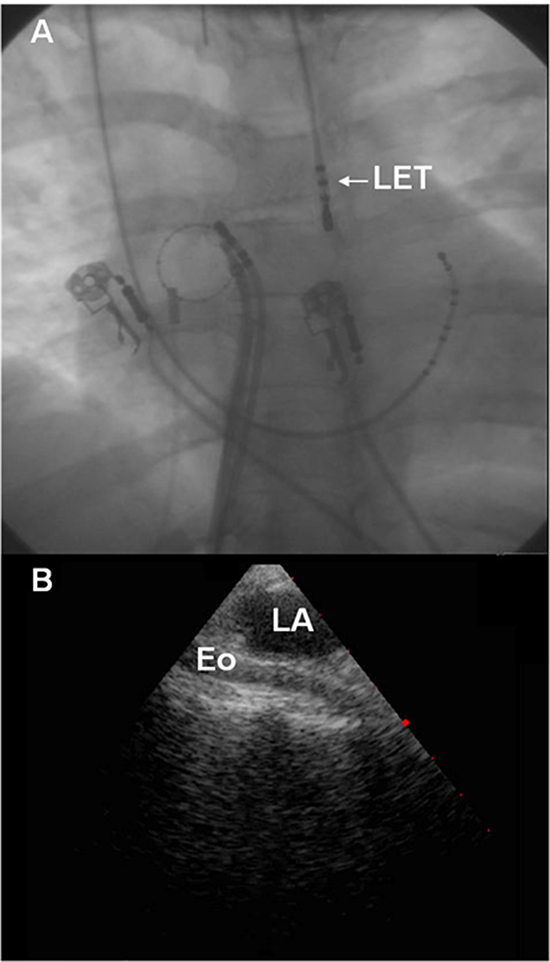
The esophagus lies in close proximity to the left atrium placing it at risk for injury during catheter ablation procedures (Figure 1). As AEF typically presents many days after ablation, direct mechanical insult during the index procedures is unlikely to be the primary culprit for fistula formation. Current theories of esophageal injury and AEF formation implicate adverse healing secondary to thermal injury to the esophagus during the index ablation procedure. The insult is believed to start at the esophageal side and extend into the mediastinum, the pericardium and then the left atrium.10 Epithelial intestinal tissue is highly susceptible to radiofrequency (RF)-induced thermal injury. Heat damage results from thermal conduction in the tissue rather than direct power application. Heat may affect esophageal endothelial cells directly, or may damage anterior esophageal arteries causing ischemia and ulceration of the mucosal layers. Morphological changes of periesophageal connective tissue and the posterior wall of the LA can be seen on endosonography after AF ablation, even in the absence of endoscopic epithelial damage.11 The delayed appearance of AEF favors esophageal artery ischemia as the primary mechanism of injury. In addition, pre-existing esophagitis due to gastroesophageal reflux may exacerbate esophageal injury perhaps by interfering with the usual repair mechanisms after esophageal injury.12 After esophageal tissue necrosis develops, mediastinitis and fistula formation occurs resulting in a communication between the esophageal lumen and the pericardium and subsequently with the left atrial blood pool via the oblique sinus.10
Figure 1. Proximity of the LA and Esophagus. A) Fluoroscopy demonstrating an esophageal temperature probe (LET). As noted, the esophagus frequently lies immediately posterior to the LA. B) Intra-cardiac Echocardiography demonstrating the relationship between the LA and esophagus.
Risk Factors For Atrioesophageal Fistula
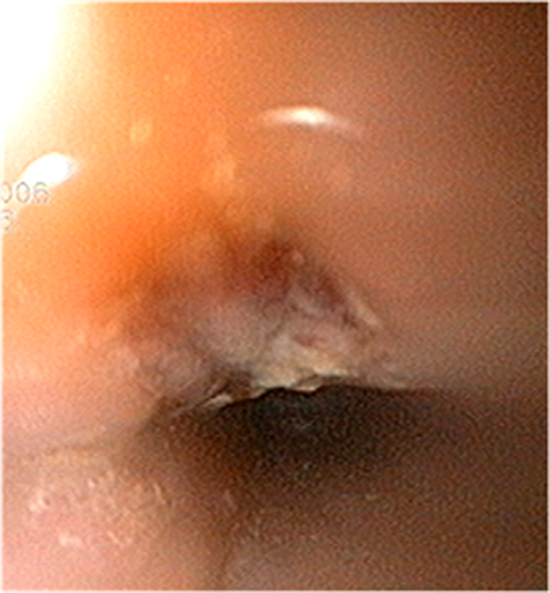
Owing to the low incidence of AEF, esophageal endoscopic studies have been used to screen for asymptomatic epithelial injuries after ablation, with esophageal ulcerations (ESULs) serving as potential precursors of fistula formation (Figure 2). Several studies have examined predisposing risk factors for esophageal injury. Patients with persistent AF may be at higher risk due to a larger left atrial size which makes the relationship between the left atrium and esophagus more intimate. Multivariate analysis of a cohort of 260 patients undergoing AF ablation highlighted that the distance between the left atrium and esophagus was an independent predictor of ESUL.13 Patients with persistent AF were more likely to have LA enlargement with compression of the esophagus between the left atrium and the spinal cord, potentially decreasing the distance between the left atrium and esophagus and thus the risk for esophageal injury. Interestingly, Yamasaki and colleagues14 highlighted that, the distance between the left atrium and esophagus may also be an issue in individuals with a low body mass. Rather than direct compression of the left atrium to the esophagus seen in persistent AF patients, in this case the injury may be related to a shorter distance with less intervening tissue between the left atrium and the esophagus.14
Figure 2. Endoscopy demonstrating an esophageal ulcer, a possible precursor to AEF.
Extensive ablation on the posterior wall, such as may occur during persistent AF ablation, may predispose to esophageal heating particularly when higher powers are administered in the vicinity of the posterior wall. An increased incidence of ESUL was noted with higher power settings15 and the use of a deflectable sheath (which may improve contact and subsequent heat transfer to the esophagus) when ablating on the posterior wall of the left atrium.16
General anesthesia may also increase the risk of esophageal injury.17 The mechanism of this is unclear but may be related to decreased esophageal peristalsis and swallowing during anesthesia which might prevent physiological cooling. Additionally, it is possible that injury may be a result of the higher use of oro- or naso-gastric tubes in procedures performed under general anaesthesia. It is possible that these tubes may result in mechanical fixation of the esophagus against the LA. As is apparent, multiple factors play a role in esophageal ulcer formation. However, risk factors for the progression of an esophageal ulcer to AEF remain unknown and will be difficult to discern given the rarity of AEF.
Measures To Minimize The Risk Of Esophageal Injury
As it has been suggested that gastroesophageal reflux may play a role in aggravating the initial esophageal insult and hinder appropriate healing thereby promoting the development of AEF, prophylactic proton-pump inhibitors (PPIs) have been recommended for patients undergoing AF ablation.12 Adequately powered clinical trials to establish the efficacy of proton pump inhibitors to reduce AEF may never be feasible given the low incidence of AEF.
Esophageal Temperature Monitoring
As it is difficult to predict the extent of heat transferred to the esophagus during catheter ablation procedures, it has been proposed that real-time luminal esophageal temperature monitoring may provide some assessment of the extent of heat transferred to the esophagus during ablation. A temperature probe placed in the esophagus at the level of the ablation catheter may allow one to detect increases in luminal esophageal temperature and may alert the operator to excessive heat transfer (Figure 1).
In a retrospective study of patients undergoing AF ablation Singh et al.18 demonstrated that patients were less likely to experience esophageal injury after catheter ablation when using luminal esophageal temperature monitoring. Their practice was to interrupt RF applications when the luminal esophageal temperature increased to 38.5°C. In this series only 6% of patients with ablation guided by esophageal temperature monitoring developed ESUL as opposed to 36% of those without monitoring. A more recent prospective study which employed a triple-thermocouple esophageal temperature monitor and limited esophageal temperature to 40°C also hinted at a low incidence of esophageal injury with only 1.6% of 184 patients showing signs of ESUL.19
Leite et al20 attempted to define an acceptable esophageal temperature rise prior to stopping RF applications.20 In this study power on the posterior wall was limited to 25 watts and terminated when the temperature increased more than 2°C from baseline. Using this strategy, no patients demonstrated esophageal thermal injury on follow-up endoscopy. Based on the totality of this work it has been suggested that careful esophageal temperature monitoring with interrupting RF application when esophageal temperature increases may minimize the risk of esophageal injury and subsequent AEF. However, limitations inherent to luminal esophageal temperature monitoring may not always prevent esophageal injury and AEF formation.
Frequently reposition the esophageal temperature probe and mismatch of the esophageal diameter relative to that of the temperature probe may result in incorrect positioning of the esophageal temperature probe thereby limiting its ability to provide accurate local temperature readings.21 Moreover, due to the phenomenon of thermal latency, the esophageal temperature may continue to rise even after RF is interrupted, resulting in temperature overshoot in a significant number of patients. Finally, esophageal luminal temperature may be significantly lower than esophageal mural temperature and thereby not reflect the extent of heat transfer to the outer and mid walls of the esophagus.22 Consequently, injury may occur after ablation even when the luminal esophageal temperature is assiduously monitored. Operators must be aware of these limitations as AEF has been reported when esophageal temperature did not rise during ablation23 clearly highlighting that esophageal temperature monitoring alone is insufficient to completely prevent esophageal thermal injuries. Other methods to actively protect the esophagus during AF ablation must be identified.
In addition to the limitation of accurate esophageal monitoring, recent work has highlighted the effects of radiofrequency ablation near metallic devices. Nguyen et al.24 highlighted esophageal temperature probes may function as “lightning rods”, attracting electrical current from the ablation catheter and potentiating heat transfer to the esophagus. Further research is needed to confirm whether esophageal temperature probes indeed exacerbate heat transfer and subsequent esophageal injury. Consistency of this finding would call into question current practices of luminal esophageal temperature monitoring during AF ablation.
Mechanical Deflection Of Esophagus
Mechanical techniques to move the esophagus away from the tip of the ablation catheter have been devised with the hope of preventing thermal injury to the esophagus. This approach is possible as the thoracic esophagus is not fixed in position by true ligaments or other significant fibrous attachments to surrounding structures. Chugh et al.25 demonstrated the feasibility of displacing the esophagus by deflecting a transesophageal (TEE) probe placed within the esophagus during ablation. They found that it was possible to move the esophagus on average 2 cm in 10 of 12 (83%) patients. To avoid mechanical complications and shunting of RF energy towards the TEE probe, the probe was removed in all patients after the esophagus was deviated. Unfortunately the esophagus remained displaced after removal of the endoscope in only 22% of patients making this approach suboptimal.
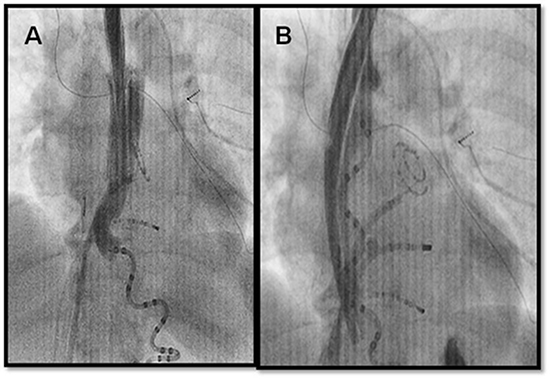
A recent study by Koruth et al.26 utilized an endotracheal stylet within a thoracic chest tube to deflect the esophagus away from the area of energy delivery in 20 patients undergoing AF ablation (Figure 3). Unlike Chugh’s work,25 the stylet and chest tube remained in the esophagus to allow for sustained deviation during ablation. Leftward and rightward deflection averaged 2.8 cm each which was maintained during ablation at the posterior wall. Post-procedural endoscopy demonstrated ulceration in one patient (5%) and evidence of trauma from esophageal instrumentation without clinical consequence in 12 patients (63%). This technique did not require participation of an endoscopist during the ablation procedure as it was performed by the anaesthesiologist participating in the AF procedure. It may well be that endoluminal esophageal displacement may become an effective method of protecting the esophagus during ablation of AF.
Figure 3. Deviation of the esophagus. A) Fluoroscopy with barium contrast to highlight the baseline position of the esophagus posterior to the left atrium. Of note is the presence of a multi-electrode temperature monitor. B) Using a chest tube and stylet, the esophagus is deviated to the right and away from the left atrium and region where ablation is required. Barium highlights the course of the esophagus.
Thermal Insulation Of Esophagus
Instrumentation of the pericardial space and introduction of a balloon catheter between the LA and esophagus to move the esophagus away from the LA is an alternative approach to reduce heat transfer to the esophagus during AF ablation.27,28 While esophageal temperature rises are limited with the introduction of a pericardial balloon, this approach does add significant complexity to an already complex AF ablation procedure.
AEF typically occurs 1 to 4 weeks after the catheter ablation procedure, although earlier and later onsets have been reported.29 The majority of signs and symptoms of AEF are not specific and may include fever, fatigue, malaise, chest discomfort, nausea, vomiting, dysphagia, odynophagia, hematemesis, melena and dyspnea. A high index of suspicion is recommended in patients with constitutional symptoms after AF ablation.
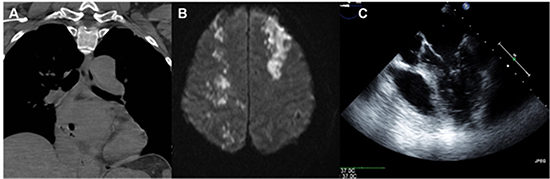
When entertaining the diagnosis of AEF a white blood cell count should be obtained as this is an early and sensitive laboratory marker of an AEF.30 In addition, imaging should be performed emergently (Figure 4). Computed tomographic (CT) scan of the chest with intravenous contrast is considered by many as the test of choice as it can rapidly and safely demonstrate findings suggestive of AEF such as pneumomediastinum or pneumopericardium. This test can be considered diagnostic if intravenous contrast enters the esophagus or mediastinum from the left atrium. Transthoracic echocardiography may demonstrate air in the left heart, pericardium or the presence of a pericardial effusion. Esophageal instrumentation with endoscopy and transesophageal echocardiography are not recommended as they theoretically may worsen the situation by increasing fistula size and also increase the risk of air embolism secondary to increased esophageal pressure with instrumentation and insufflation.31
Figure 4. Imaging to aid with the diagnosis of AEF. A) CT chest demonstrating air in the LA, B) MRI of the brain demonstrating infarctions in multiple territories, c) Echocardiogram demonstrating air in the left heart.
Early recognition is important, as patients often develop endocarditis with septic emboli leading to neurological manifestations such as altered mental status, seizures, and coma within hours of symptom onset.30
Although the prevalence of AEF post-AF ablation is low, the fatality rate is high and reported between 67% and 100%.32 The high case-fatality rate has traditionally been attributed to the lack of recognition and late presentation of this complication. The prognosis of patients who survive to obtain an accurate diagnosis and receive corrective surgical intervention is variable.
Available therapeutic options for AEF include surgical repair of the fistula (combined left atrial and esophageal repair) via thoracotomy, esophageal stenting and conservative management with aggressive chest tube drainage and treatment of sepsis. Of these three approaches, conservative treatment of esophageal fistula remains controversial, as it requires frequent radiologic assessments and is associated with very high mortality rate. Data on stenting versus surgical treatment of AEF are conflicting and at the present there is no consensus on the most effective treatment strategy for AEF. In our opinion esophageal stenting may be useful only if esophageal perforation is present without fistula formation to the left atrium.33,34 Our preference is, if possible, early aggressive surgical repair. Our group reported on the outcomes of 29 patients undergoing AEF repair.34 The report suggested that surgical esophageal repair with placement of tissue between the esophagus and left atrium may result in lower morbidity and mortality.34 Attention to proper left atrial and esophageal repair is critical and may be best achieved with the use of cardiopulmonary bypass. Furthermore, surgical and medical treatment of the associated mediastinitis is important. We recommend a multidisciplinary approach to care including cardiac-thoracic surgeons, infectious disease, neurology, and critical care physicians as well as allied health care professionals such as dieticians, physiotherapists and occupational therapists.
Due to the rarity of the complication, it is not clear if earlier detection and earlier repair will result in improved clinical outcomes; however this strategy does make sense. Given this, patient education on the signs and symptoms of AEF is of paramount importance to allow patients to present to medical attention sooner to facilitate earlier repair prior to the onset of mediastinitis or stroke. Additionally, education of primary care and emergency room physicians of this complication is also important to avoid misdiagnosis, and ensure esophageal manipulation is minimized. A recent approach by Canadian centers performing AF ablation is to provide patients who have undergone an AF ablation procedure a pocket card (Figure 5) which they present to their primary care physician with the onset of vague symptoms. The interaction when the patient receives this card educates them on the symptoms of AEF with the hope of earlier presentation. Furthermore, the instructions on the card ensure primary care physicians do not instrument the esophagus and encourages collaboration with the primary Electrophysiologist to ensure that the diagnosis is made as soon as possible. It is hoped that this strategy may minimize the morbidity and mortality associated with this complication.
Figure 5. Patient pocket card

AEF is uncommon but has been reported with all approaches to AF ablation, without clearly identifiable predisposing factors. Despite our knowledge of this complication, outcomes with AEF related to AF ablation remain poor. Ongoing assessment of prevention strategies, as well as patient and physician education to recognize this complication should be encouraged as, given the growth in AF ablation procedures, AEF will continue to occur.